Context
Advanced abdominal pregnancy (AAP) is defined as a pregnancy of over 20 weeks' gestation with a foetus living, or showing signs of having once lived and developed, in the mother's abdominal cavity1-4. Often undiagnosed prior to operative intervention, and prone to dramatic complications, advanced abdominal pregnancy presents special challenges to the physician working in remote areas with limited resources for diagnosis and management.
Kolofata health district covers an 8502 km rural zone in the Far North Province of Cameroon. Its population of 115 274 consists primarily of subsistence level farmers and herders. Illiteracy is very high and fewer than 5% of women can read or write. Health care is provided by six primary care health centers and the 100 bed district hospital. The hospital receives approximately 26 000 patients a year in consultation.
We present a case of AAP received in the Kolofata District Hospital, and follow with a review of 163 others reported throughout the world since 1946.
Issue
Case report
A 30 year-old woman, gravida 5 with four living children and no history of abortion, presented to her local health centre with amenorrhea of 9 months' duration and a chief complaint of abdominal pain and absent foetal movements for 7 days. The patient's third and youngest living child was 12 years old. The birth of her fourth child, who died at 10 months of probable pneumonia, had been followed by a 10 year period of infertility before this fifth pregnancy. There was no history of pain prior to this episode, and she had had no vaginal bleeding during the pregnancy. Three previous antenatal visits were made to the health center. Each visit consisted of an interview and clinical examination by a nurse or midwife, who noted an apparently normal pregnancy. Ultrasound examination, unavailable in rural health centers in Cameroon, was not performed.
Detecting a transverse lie and no foetal heartbeat, the consulting nurse referred the patient to our hospital.
On examination she was in no evident pain or distress. She weighed 62 kg and had a blood pressure of 110/70 mmHg. Her conjunctivae were pale (hematocrit 26%; normal range 38-46%). Her abdominal circumference was 89 cm. An abdominal mass presumed to be the uterine fundus was palpated at 31 cm above the symphysis pubis (Fig1). The foetal presentation was not clear. No foetal heart sounds were heard. Ultrasound examination revealed a partially decomposed full term male foetus lying obliquely, the head in the mother's right upper quadrant, and a large globular placenta of abnormal echogenicity: multiple fluid-filled spaces in the placenta gave an unusual picture not unlike that of molar tissue. The mother's bladder was empty but a mass suggestive of a possibly empty uterus adjacent to the foetus was detected. A diagnosis of possible mixed-molar or abdominal pregnancy was made. To rule out the latter, the ultrasound examination was repeated several hours later with the mother's urinary bladder full. This time, the non-gravid uterus was clearly visible posterior to the bladder and distal to the externally implanted placenta and foetus. The patient was taken to the operating room.
An exploratory laparotomy under ketamine anaesthesia was performed through a sub-umbilical median incision that extended 3 cm above the umbilicus. The thickened peritoneum was entered and a partially macerated male foetus lying in a prone and slightly oblique position was revealed (Fig2). No putrid odour was noted. The foetal head was in contact with the maternal liver, stomach and transverse colon. The breech was astride the placenta, the maternal surface of which cupped the external wall of the uterine fundus. The placenta, enveloped in membranes and coated with meconium, was friable. Placental tissue penetrated the mesentery posteriorly and the uterus inferiorly. The foetus was removed without difficulty. Extraction of the placenta incited a profuse haemorrhage which was controlled by prolonged manual pressure, sutures and a pedicle graft of the omentum. Although most of the placenta was removed, placental debris adherent to surrounding structures was left in place. The fallopian tubes, ovaries and uterus were macroscopically normal. Intestinal motility and the mesenteric arterial pulse were monitored throughout the procedure and remained normal. A peritoneal drain was inserted before closure. Blood loss was estimated to be 500 mL. No transfusion was given. The baby weighed 3000 g and had no evident deformity (Fig3). The mother was given heparin for 24 hours and ceftriaxone for 3 days, followed by penicillin and gentamycin for 4 days. She was discharged well on post-operative day 8 and was in excellent health when last seen for routine follow up 3 months later.
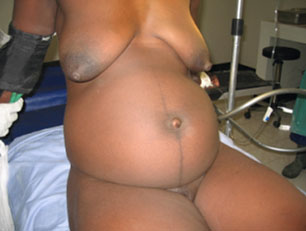
Figure 1: Normal external appearance of woman carrying an abdominal pregnancy at term.
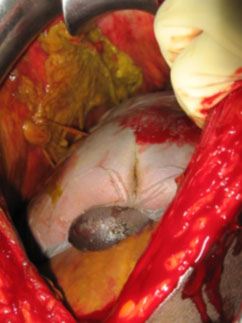
Figure 2: Breech of partially macerated male foetus lying prone astride the placenta, outside the intact uterus.
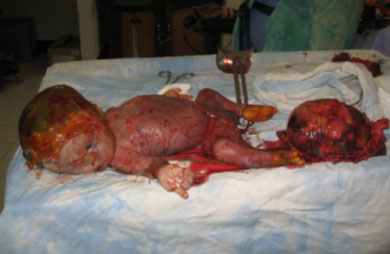
Figure 3: Newborn and placenta.
Review of published cases
A journal search and PubMed review was completed. The journal search consisted of articles published pre-1950 (not listed in PubMed) compiled by a manual search of old journals for relevant papers, guided by references listed in papers post-1950 found through PubMed. Altogether, 163 cases of AAP described in 22 reports from 13 countries since 1946 (Table 1 and Table 2) suggested that while patterns emerge, there is no typical case of AAP, and controversies remain concerning optimal management.
Table 1: Characteristics of 163 cases of advanced abdominal pregnancy reported from 13 countries since 19461-22
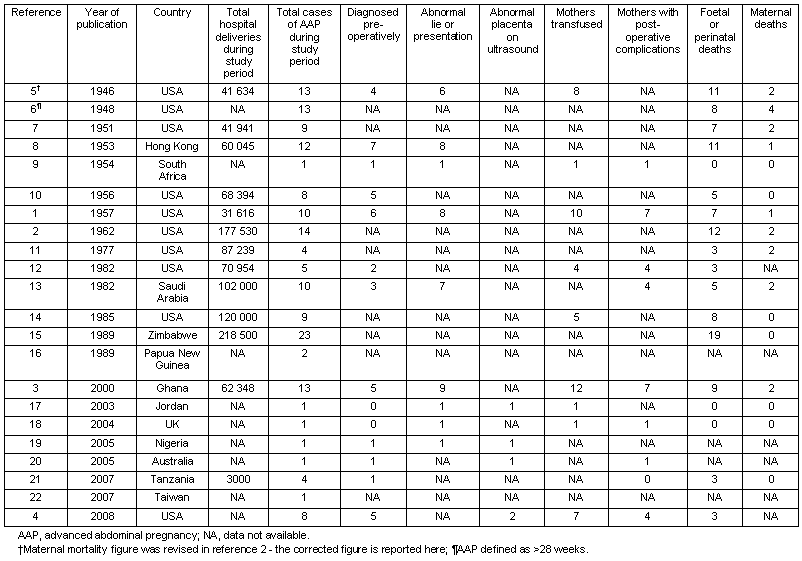
Table 2: Summary of 163 reported cases of advanced abdominal pregnancy1-15,17-21
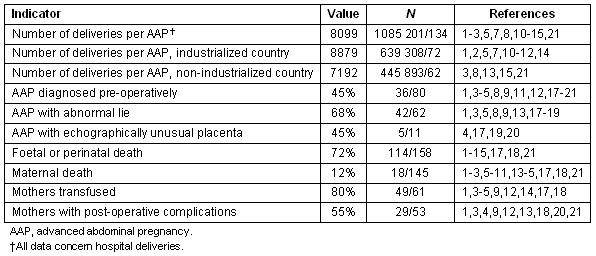
Data: Because complete descriptions are not uniformly available in all reported cases and case series, the denominator varies for each of the indicators listed. Calculations are necessarily based only on data explicitly stated in the article. In articles where foetal lie, for example, is not mentioned, the cases presented in that paper are considered in neither the numerator nor the denominator of the indicator 'AAP with abnormal lie'.
Incidence: In the series reviewed here, one AAP occurred for every 8099 hospital deliveries. The incidence was 19% higher in non-industrialized countries than in industrialized countries.
Diagnosis: The diagnosis of AAP is difficult and is made on the basis of history, physical examination and imagery. Echographic evidence of a non-gravid uterus alongside a foetus is diagnostic, but this will be seen only if the operator purposefully seeks the uterus on ultrasound examination. A pre-operative diagnosis of AAP is missed more often than it is made: only 45% of cases described here were diagnosed pre-operatively. The insertion of a balloon catheter in the uterus during ultrasound may help clarify an ambiguous image15. In any case, a high index of suspicion is crucial, and this should be triggered by any of the following clues:
- a history of bleeding or excessive abdominal pain during the first trimester
- a history of previous abortion or pelvic surgery
- a history of infertility
- bleeding or non-labour abdominal pain during the third trimester
- maternal declaration of cessation of foetal movements
- perception on the part of the mother or the physician that 'something is not right'
- abnormal foetal lie
- displaced cervix or abdominal mass palpated apart from the foetus
- unusual echographic appearance of the placenta
- failed induction.
Management: Because perinatal death may result from either prematurity or prolonged gestation in a compromised environment, the decision about when to intervene in the case of a live baby should be made in consultation with the mother. Once the foetus has reached a viable age, there is little reason to delay delivery. Regardless of timing, the mother's own safety will be best assured by careful monitoring, foresight and pre-operative preparation.
The principal controversy concerning management of AAP is whether or not to remove the placenta. Because the abnormally implanted placenta's blood supply is diffuse and often unidentifiable, attempts to remove it can incite catastrophic haemorrhage. Measures taken to control this haemorrhage during surgery risk compromising the blood supply of other organs. A placenta left in situ might resorb spontaneously but if it does not, the risk of infection, necrosis, and the need for a second surgery is considerable. Most authors agree that the placenta should be removed provided its blood supply is identified and can be ligated without damaging other organs. If the blood supply cannot be identified and safely ligated, the placenta should be left in place and the patient followed for possible complications. The use of methotrexate to shrink a placenta left in situ has been largely discredited. Delaying removal of a stillbirth to give time for the placenta's blood supply to shrink remains controversial.
Outcome: Foetal and maternal complications in AAP are the rule. In the series presented here, foetal or perinatal mortality was 72%, and pressure deformities were common among survivors. Maternal post-operative complications, most notably haemorrhage and infection, occurred in more than half of all patients, and over three-quarters required blood transfusion. Maternal mortality was 12%.
Lessons learned
Conclusions
Advanced abdominal pregnancy is rare, but given an overall incidence of one AAP for 8099 hospital deliveries, a medical officer responsible for a district population of 100 000 might expect to encounter an AAP every few years if every pregnant woman near term presented for prenatal care or delivery.
The case reported here has many features of other cases reviewed: the mother, pregnant and at term following a ten-year period of secondary infertility, presented with abdominal pain, absent foetal movement and abnormal foetal lie. The unusual echographic appearance of the placenta was key to suspecting that the pregnancy was extraordinary, and this suspicion prompted the repeat ultrasound examination, this time with a filled bladder to secure a higher quality result. The second examination confirmed the non-gravid state of the uterus and permitted the diagnosis of AAP. Abnormal echographic appearance of the placenta has not been cited by other authors as diagnostically important in cases of AAP, but it was described in several cases reviewed here and should be considered helpful.
For the physician in a remote area, where capacity to respond to potentially disastrous surprises is limited by lack of personnel and other resources, the ability to diagnose AAP pre-operatively is especially important. Pre-operative diagnosis gives the medical team time to review management and possible complications of AAP. The patient and family can be briefed. Additional personnel, adequate supplies of blood and resuscitation equipment can be made available. For these reasons, whenever a pregnant woman at term presents with any of the diagnostic criteria listed above, a thorough investigation, complete with clinical and, if possible, ultrasonographic assessment of the uterus and the placenta, in addition to the foetus, should ensue to assure that the baby is indeed within the uterine cavity. Regardless of symptoms and signs appreciated retrospectively, if the possibility of abdominal pregnancy is not considered at presentation, abdominal pregnancy will not be diagnosed.
Ultrasound examination, still insufficiently accessible in many remote areas, is a valuable tool and should be part of the minimal standard of care for every pregnant woman.
The variable definition of AAP and the changing accessibility of imaging technology over time and place may introduce error when comparing case studies or drawing statistical conclusions. More than 90% of the cases presented here were reported prior to 1990, when ultrasound was rarely available in rural settings in the developing world. It may be assumed that many potential cases of AAP in resource-rich countries are diagnosed and terminated in early pregnancy, resulting in the higher incidence seen in non-industrialized countries. Finally, publication bias may have resulted in an underestimation of mortality, as the thrill of success in these unusual cases naturally tends to make one more eager to publish.
References
1. Crawford JD, Ward JV. Advanced abdominal pregnancy. Obstetrics and Gynecology 1957; 10(5): 549-554.
2. Beacham WD, Hernquist WC, Beacham DW, Webster HD. Abdominal pregnancy at Charity Hospital in New Orleans. American Journal of Obstetrics and Gynecology 1962; 84(10): 1257-1266.
3. Opare-Addo HS, Deganus S. Advanced abdominal pregnancy: a study of 13 consecutive cases seen in 1993 and 1994 at Komfo Anokye Teaching Hospital, Kumasi, Ghana. African Journal of Reproductive Health 2000; 4(1): 28-39.
4. Worley KC, Hnat MD, Cunningham FG. Advanced extrauterine pregnancy: diagnostic and therapeutic challenges. American Journal of Obstetrics and Gynecology 2008; 198: 297.e1-297.e7.
5. Beacham WD, Beacham DW. Abdominal pregnancy - a review. Obstetrical and Gynecological Survey 1946; 1: 777-805.
6. Ware HH. Observations on thirteen cases of late extrauterine pregnancy. American Journal of Obstetrics and Gynecology 1948; 55: 561-582.
7. Cross JB, Lester WM, McCain JR. The diagnosis and management of abdominal pregnancy with a review of 19 cases. American Journal of Obstetrics and Gynecology 1951; 62(2): 303-311.
8. King G. Advanced extrauterine pregnancy. American Journal of Obstetrics and Gynecology 1953; 67(4): 712-740.
9. Villiers JN. Extra-uterine pregnancy at or near term: a case report with a review of the literature. South African Medical Journal 1954; 28(13): 254-260.
10. Yahia C, Montgomery G. Advanced extrauterine pregnancy: clinical aspects and review of eight cases. Obstetrics and Gynecology 1956; 8(1): 68-80.
11. Strafford JC, Ragan WD. Abdominal pregnancy: review of current management. Obstetrics and Gynecology 1977; 50(5): 548-552.
12. Delke I, Veridiano NP, Tancer ML. Abdominal pregnancy: review of current management and addition of 10 cases. Obstetrics and Gynecology 1982; 60(2): 200-204.
13. Rahman MS, Al-Suleiman SA, Rahman J, Al-Sibai MH. Advanced abdominal pregnancy - observations in 10 cases. Obstetrics and Gynecology 1982; 59(3): 366-372.
14. Hallatt JG, Grove JA. Abdominal pregnancy: a study of twenty-one consecutive cases. American Journal of Obstetrics and Gynecology 1985; 152(4): 444-449.
15. White RG. Advanced abdominal pregnancy - a review of twenty-three cases. Irish Journal of Medical Science 1989; 158(4): 77-78.
16. Alto W. Is there a greater incidence of abdominal pregnancy in developing countries? Report of 4 cases. Medical Journal of Australia 1989; 151(7): 412-414.
17. Badria L, Amarin Z, Jaradat A, Zahawi H, Gharalbeh A, Zobi A. Full-term viable abdominal pregnancy: a case report and review. Archives of Gynecology and Obstetrics 2003; 268: 340-342.
18. Ramachandran K, Kirk P. Massive hemorrhage in a previously undiagnosed abdominal pregnancy presenting for elective Cesarean delivery. Canadian Journal of Anaesthesia 2004; 51(1): 57-61.
19. Ekele BA, Ahmed Y, Nnadi D, Ishaku K. Abdominal pregnancy: ultrasound diagnosis aided by the balloon of a Foley catheter. Acta Obstetricia et Gynecologica Scandinavica 2005; 84(7): 701-702.
20. Roberts RV, Dickinson JE, Leung Y, Charles AK. Advanced abdominal pregnancy: still an occurrence in modern medicine. Australian and New Zealand Journal of Obstetrics and Gynaecology 2005; 45: 518-521.
21. Zeck W, Kelters I, Winter R, Lang U, Petru E. Lessons learned from four advanced abdominal pregnancies at an East African Health Center. Journal of Perinatal Medicine 2007; 35: 278-281.
22. Shaw S-W, Hsu J-J, Chueh H-Y, Han C-M, Chen F-C, Chang Y-L et al. Management of primary abdominal pregnancy: twelve years of experience in a medical centre. Acta Obstetricia et Gynecologica Scandinavica 2007; 86(9): 1058-1062.

