Prostate cancer is second to lung cancer in incidence worldwide, and it is the third most common cause of cancer deaths in developed countries (age-standardised rates in 20081). Understanding geographical patterns of prostate cancer incidence and mortality is important in order to detect any disparities in access to diagnostic and treatment services. In particular the physical distances to access specialist services are believed to disadvantage rural patients2,3.
In general, rural communities have higher proportions of disadvantaged groups, being typically older, poorer, experiencing a higher burden of chronic diseases, and some having a different ethnic composition compared with urban communities4-7. All of these factors may impact on a man's ability and willingness to travel potentially long distances for cancer screening or treatment. In addition, the availability of particular modalities of treatment may vary depending on patient's age and general health status8,9. In the case of prostate cancer, although the specific aetiology of the disease is not known, age, family history and African ancestry have been identified as risk factors10,11.
Internationally, discrepant patterns have been observed for cancer incidence and mortality when rural and urban populations are compared. Although some evidence suggests higher incidence of and lower mortality from cancers in urban areas12,13, the observations vary considerably by type of cancer as well as by country14-16. Furthermore, results may vary due to inconsistent definitions of the concepts of urban and rural residence.
The aim of this study was to undertake a systematic review of the literature in order to determine whether there are differences in prostate cancer incidence and mortality between rural and urban men. It was hypothesised that rural men in general are less likely to be screened for prostate cancer, more likely to be diagnosed with advanced cancer, will have worse access to curative treatment and a higher mortality rate than their urban counterparts.
A systematic review of the literature was performed in order to explore whether there are differences in: (i) incidence of prostate cancer; and (ii) mortality from prostate cancer between urban and rural populations.
Search strategy
Three electronic databases - Medline, CINAHL and Embase - were searched in July 2011 using key phrases 'prostate cancer incidence rural ' and 'prostate cancer mortality rural', inclusive of the following MeSH terms: 'prostatic neoplasms'; 'prostatic' and 'neoplasms'; 'prostate' and 'cancer'; 'prostate cancer'; 'incidence'; 'mortality'; 'survival'; and 'rural'. For CINAHL and Embase searches, MeSH terms 'rural population' and 'rural areas' were also included. The search was limited to peer-reviewed studies published in English from 1 January 1980 onwards and involving human subjects. Furthermore, the following inclusion criteria were applied:
- population: cohort (population-based study) of adult men
- intervention or exposure: prostate cancer (grouped cancers, ie urological cancer excluded)
- comparison: rural versus urban
- outcome: incidence, mortality (relative risk, hazard ratio or standardised mortality or incidence ratio and confidence intervals with p values must be stated).
In total, 139 articles were listed following the 'incidence' search and 53 following the 'mortality' search of Medline. Fifty-one articles (one of these a duplicate) were included in both lists. Therefore, these articles were only assessed once. Subsequently, titles and abstracts were assessed by applying the inclusion criteria resulting in the exclusion of 59 articles from the 'incidence' list and 33 articles from the 'mortality' list. Consequently, the full-texts of 49 articles were retrieved and evaluated for suitability. As a result 13 articles were included in this review. The inclusion of articles was reviewed by two researchers (ZO, RL). The 36 articles were excluded because statistical information was missing (n=15), prostate cancer (7) or rural/urban residence (7) was not specifically mentioned (8), the study was not based on a cohort (3), or the outcome was other than mortality or incidence (3).
The search of CINAHL resulted in 76 articles, none of which met the inclusion criteria. This was primarily because their focus has been on different treatment options and their outcomes, rather than on general prostate cancer incidence and mortality. Sixty-seven articles were identified during the Embase search; 30 articles of these matched those from the Medline search. The remaining 37 articles deviated from the main topic and did not meet the inclusion criteria.
Reference lists of included manuscripts were hand-searched to identify further publications, resulting in the inclusion of 7 articles that had not been identified by database search. In addition, based on the secondary literature search, 5 'grey literature' reports from four countries (New Zealand, Australia, England, and Canada) were accessed.
Assessment of the scientific quality of studies was undertaken with respect to a number of methodological issues such as sample size, study design, presentation of results, and statistical findings.
Definition of rural and urban populations
One of the major limitations of this review is the inconsistent definition of 'rurality', the use of different geopolitical units (ie cantons, municipalities, minor civil units), and different definitions of residence (usual address vs address at diagnosis or death). The concept of rurality or remoteness may vary considerably among countries17-20. In addition, demographic and socioeconomic factors may differ between rural and urban groups within and among countries21-23. These different characteristics may significantly contribute to differences in cancer incidence and mortality.
Studies included in this review used various measures to define rural and urban populations, including settlement size, population density, accessibility to services (ie distances or travel times to service centres), economic activity (ie employment opportunities) or a combination of these.
For the purpose of this study, definitions of rural and urban populations were directly adopted from the individual articles. Although several studies discussed intermediate categories, such as suburban groups, only comparisons between 'extreme' rural/urban groups have been considered in this review.
Due to these limitations, instead of direct comparisons of incidence and mortality among the studies, general trends were described within and among studies. A meta-analysis could not be undertaken due to the inconsistent definition of rurality and lack of standardisation in data presentation.
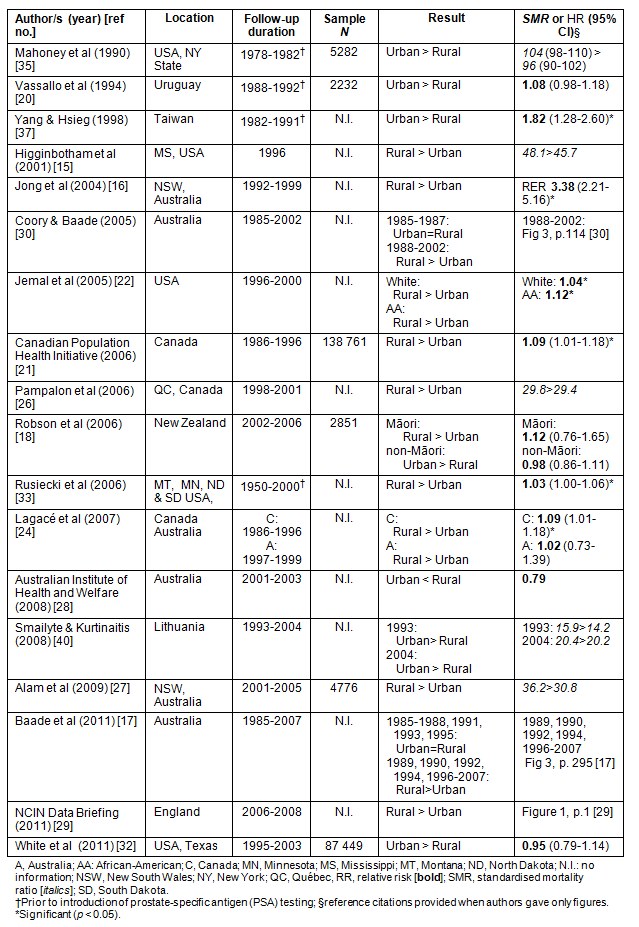
The majority of studies were identified from Medline. However, seven manuscripts were included from secondary literature15,19,20,22-26, and five reports were not published in peer-reviewed journals but met the inclusion criteria18,21,27-29. A total of 16 population-based studies discussed rural/urban differences in incidence of prostate cancer, while 18 studies examined data on mortality. Nine publications included both aspects but were analysed separately15,17,18,21,22,27-30. The key characteristics of studies discussed in this review are presented in Tables 1 (incidence) and 2 (mortality).
Data in all included studies were sourced from state or national cancer registries and official census and death records databases. Several studies included information on case completeness of registries as a measure of quality of the data, which in each case was greater than 85%19,22,31,32.
As per inclusion criteria, all studies were based on data from adult men, although some studies included only men over the age of 35 years17,22,30,33.
The number of new cases of prostate cancer (based on varying follow-up periods) ranged from 75931 to 24 33327 men; however, eight studies did not include this information. Six studies originated in the USA, six in Europe (Denmark, England, the Netherlands, Lithuania, Spain and Bulgaria), five in Australia, three in Canada, two in Central/South America (Costa Rica and Uruguay), and one each in New Zealand, Asia (Taiwan) and Africa (Egypt).
The study periods of the articles ranged from 1978 to 2008. The prostate-specific antigen (PSA) test was introduced as a new method for prostate cancer diagnosis in the early 1990s. Consequently, the registration of new cancer cases has increased internationally34. In this review, eight articles (4 on incidence, 4 on mortality) were based on data collected prior to the widespread use of PSA tests19,20,25,31,33,35-37. Five studies used a mixture of data from both periods, which, in addition to varying follow-up periods, may result in conflicting interpretations17,21,24,30,38. For instance, Baade et al17 and Coory and Baade30 both based their research on an identical data set for a given period but Baade et al presented annual rates while Coory and Baade summarised the data into 3 year periods. As a consequence, each of these studies reported slightly different incidence and mortality patterns when comparing urban with rural men.
Table 1: Summary of studies on prostate cancer incidence in rural versus urban men (n = 16)15,17-19,21-23,25,27-31,36,38,39
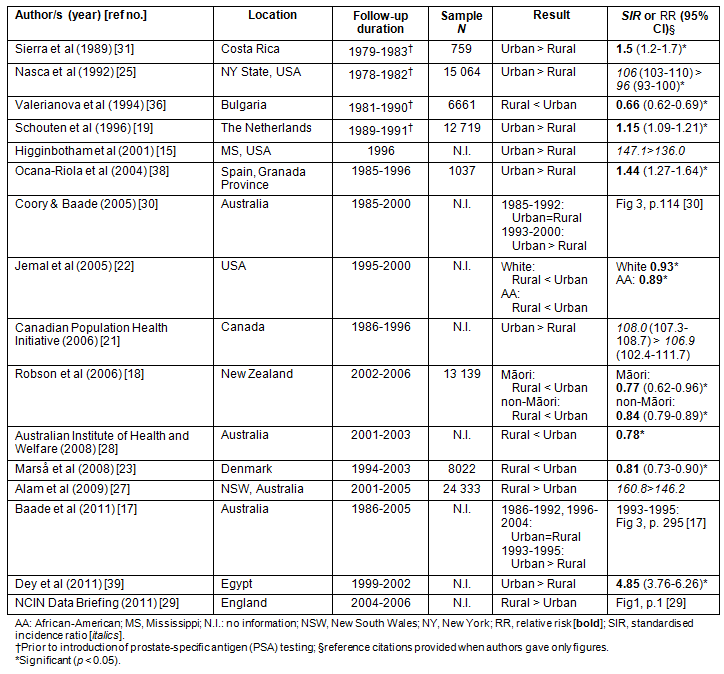
Table 2: Summary of studies on prostate cancer mortality in rural versus urban men (n = 18)15-18,20-22,24,26-30,32,33,35,37,40
Incidence
Twelve out of the 16 studies on incidence of prostate cancer reported higher rates for urban men, while a single study showed a reverse trend29. The remaining three studies showed no significant differences between rural and urban men but two observed a trend toward higher incidence in urban residents.
Four publications used data from a period prior to the PSA era, all of which reported significantly higher incidence rates in urban men19,24,25,36. Therefore, the introduction of PSA testing seems to have little effect on the incidence patterns when rural and urban residence is compared.
The National Cancer Intelligence Network (NCIN) study29, which reported higher incidence of prostate cancer in English villagers, analyses the most recent data from all the studies in this review (2004-2006). Therefore, this reverse trend might reflect a recent shift towards more diagnostic activity in rural areas or a 'saturation' of new cases in urban areas. A similar but not statistically significant trend has been observed in an Australian study based on data collected between 2001 and 200527.
Coory and Baade found no significant differences between rural and urban men in the period 1985-1992 (prior to widespread PSA testing), while they found significantly higher rates for urban residents for the years 1993 up to 200030. In the more recent study by Baade et al, a significantly higher incidence for urban men was reported for the years 1993-1995, but no difference was found for 1986-1992 and 1996-200417. This suggests an initial, more rapid uptake of screening in urban males with a later uptake in rural men so that over time the incidence has become roughly equal. It may also imply improved access to health care over time in the rural areas surveyed.
Mortality
Eight out of 18 articles on mortality of men with prostate cancer reported a greater death rate for rural men16,17,21,22,24,29,30,33, one a reduced death rate37, and nine reported no difference. In six of these nine studies a trend towards higher death rate in rural men was observed15,18,24,26-28. The study by Yang and Hsieg, the only article to report a reduced death rate in rural men, did note that all men in the study region had good access to healthcare facilities37. Rural men may have travelled to urban centres for treatment, and subsequently have died there producing a misattribution bias.
Lagacé et al compared data on prostate cancer mortality in rural versus urban Canadian and Australian men due to their similar demographic and geographical characteristics24. However, the Canadian data spanned a period of 11 years (1986-1996), whereas Australian data covered only a 3 year period (1997-1999). Both studies showed a trend towards higher death rates in rural residents.
Two20,35 out of the three studies that showed a trend toward higher rates in urban men assigned rural/urban categories by residence at death. As a consequence some rural men with prostate cancer may have been counted as urban because they might have moved to urban centres to receive treatment. Mahoney et al acknowledged that their analysis did not account for migration, nor did it consider differing ethnic compositions of their rural and urban populations35. Furthermore, these two studies20,35 as well as the study by Yang and Hsieg37 were based on data collected prior to the widespread use of PSA testing.
Ethnicity
Although this review focused on population-based studies, differences in ethnic composition between urban and rural groups may considerably influence the overall prostate cancer incidence and mortality rates. Although the majority of studies in this review had not adjusted their rates for ethnicity, three studies presented results by ethnicity. Jemal et al found that the number of new cancer cases was higher in urban men, while mortality was higher in rural men, regardless of African or European ancestry22. Higginbotham et al, however, showed that there was no difference in incidence and mortality between urban and rural African American men in Mississippi15. For White American men, no difference was found in the number of new cases of prostate cancer but rural men showed a higher death rate. Robson et al reported that the incidence of prostate cancer was lower among rural Maori as well as rural non-Maori New Zealand men18. There was no difference in mortality between urban and rural residents of either Maori or non-Maori descent.
Discussion
Despite inconsistent definitions of rural/urban classification between the studies there seems to be a clear trend for an increased incidence of prostate cancer in urban men with 75% of articles reporting higher rates in these men compared with their rural counterparts. Lower rates of new prostate cancer cases in rural men have been repeatedly assigned to lower uptake of PSA screening in rural and remote areas16,17,21,22,30. Dey et al observed that in Egypt better detection possibilities in a cancer centre doubled the incidence rate39. However, even before the PSA era, diagnostic activity was more intensive in urban centres than in rural areas36.
Contrary to incidence, mortality patterns varied to some degree when rural and urban men with prostate cancer were compared. Although some studies identified a greater mortality rate for rural residents who face long distances in accessing cancer services16,21,22, others found no significant difference in death rates between rural and urban men with prostate cancer15,26,27,32,40. There is evidence, however, that after PSA testing was introduced death rates tended to be higher in rural men. Each of the four studies based on mixed data sets (those from slightly before and during PSA testing) showed a significant trend towards a higher death rate in rural men with prostate cancer. Based on these articles it seems that urban men particularly have benefited from early diagnosis during this period17,21,24,30.
Lower rates of PSA screening in rural areas may be associated with later stage at initial diagnosis for rural men, which may significantly impact their outcomes16,22. Jemal et al found that rural patients are more likely to present at a more advanced stage regardless of their ethnic background22. Similarly, Jong et al reported a reduction in excess risk of death (RER) for rural men with prostate cancer when the stage of disease at initial diagnosis was accounted for16, although their RER was still 2.5 times higher compared with men living in highly accessible areas of New South Wales, Australia. Conversely, Higginbotham et al found no difference between stage distribution among urban and rural residents in Mississippi, USA15. However, this finding corresponds with mortality patterns observed in this study, which did not differ between rural and urban men regardless of ethnic background.
In general, rural populations seem to have higher all-cause mortality rates mainly because they are on average older and have more co-morbidities compared with their urban counterparts7,17. Because prostate cancer is a disease of older men, the presence of several concurrent medical conditions may affect the decision-making process when types of treatment are discussed41,42.
The variation in trends may be partly attributed to differing definitions of rural/urban categories, particularly with earlier studies using population density and current residence at death as variables20,35, as opposed to more recent studies that account for parameters such as remoteness and economic activity in addition to population size16,26,32. Moreover, Coory and Baade30 and Baade et al17 showed that different lengths of follow up may result in differences in mortality patterns, even if the underlying data sets are identical.
Differences in ethnic background and socioeconomic status between rural and urban communities may play a role in urban-rural disparities in prostate cancer mortality. Recent research suggests that men of African ancestry are at a greater risk of developing prostate cancer and are also more likely to suffer from more aggressive tumours11,32. Mc Lafferty and Wang attributed high rates of late-stage diagnosis observed in the city of Chicago to a high proportion of economically disadvantaged groups in this urban centre43. Although physical distance does not present a barrier for urban residents, other factors such as low education level and poverty may have effects similar to remoteness on disadvantaged groups. It is therefore advisable to account for the heterogeneity of rural (or urban) populations in future analyses in order to gain better insight into the reasons for differences in cancer outcomes between rural and urban areas.
Limitations
A disadvantage for this study (and other reviews of rural inequalities) was the lack of uniform definition of rural-urban classification within and among countries. Some of the earlier studies offered only very vague descriptions of how they distinguished between urban and rural populations31,36,38, while more recent studies were based on classification schemes with precise descriptions, which involve geographical and economic aspects (eg Rural Urban Continuum Codes [RUCC] in USA or accessibility/remoteness index of Australia [ARIA]). However, authors often chose to pool several categories within these schemes, causing considerable heterogeneity, particularly within rural groups17,27. This effect is reinforced by the fact that the majority of studies used larger areas (eg counties or municipalities) as the basic geographical unit20,23,37. As a result the rural groups varied considerably with regard to physical remoteness as well as socioeconomic status (eg suburban residents with high income and excellent access to health care). However, using capital cities (or major cities) as the urban category20,23,28,31 results in socioeconomic and ethnic diversity, analogous to the pooled rural samples. Therefore, comparisons in this study were based on the most 'extreme' rural (very remote) and urban areas (capital cities) where such data were available.
This systematic review examined the current international literature on rural versus urban differences in prostate cancer incidence and mortality. The studies reviewed reported higher prostate cancer incidence among urban men but lower mortality rates. This may be explained in part by urban men's better access to health care, with increased screening and perhaps even over-detection of prostate cancer, while at the same time ultimately a reduction in mortality. However, mortality data showed heterogeneous trends, which may be due to inconsistencies in rural-urban classifications and lack of consideration for covariates, such as cancer stage, ethnicity, and socioeconomic and general health status.
Urban-rural disparities in prostate cancer incidence and mortality seem to arise not only from the obvious difference in physical distances to healthcare facilities between rural and urban residents, but also from differences in demographic and socioeconomic characteristics of these two groups that may influence access to and utilization of diagnostic and treatment services.
Future studies should examine these factors in order to assess their contribution to inequalities in outcomes between rural and urban populations. Further research would also be beneficial to ascertain to what extent differences in screening, diagnosis or treatment options affect outcomes for urban and rural men with prostate cancer, so that healthcare services can be improved and resources appropriately allocated.
Acknowledgements
This study was supported by Health Research Council of New Zealand (HRC Partnership Programme grant number 11/082, entitled 'The costs and complications of screening for prostate cancer'). The School of Population Health Board of Research is thanked for support in developing the manuscript.
References
1. Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. GLOBOCAN 2008 v1.2. IARC CancerBase no. 10. Lyon: International Agency for Research on Cancer, 2010. Available: http://globocan.iarc.fr (Accessed on 15 July 2011).
2. Campbell NC, Elliott AM, Sharp L, Ritchie LD, Cassidy J, Little J. Rural factors and survival from cancer: analysis of Scottish cancer registrations. British Journal of Cancer 2000; 82: 1863-1866.
3. Jones AP, Haynes R, Sauerzapf V, Crawford SM, Zhao H, Forman D. Travel times to health care and survival from cancers in Northern England. European Journal of Cancer 2008; 44: 269-274.
4. Bertolini P, Montanari M, Peragine V. Poverty and social exclusion in rural areas. Berlin: European Commission, 2008.
5. Weber B, Jensen L, Miller K, Mosley J, Fisher M. A critical review of rural poverty literature: Is there truly a rural effect? Institute for Research on Poverty Discussion Paper no. 1309-05, Madison, WI: University of Wisconsin-Madison, 2005.
6. Casey MM, Thiede Call K, Klinger JM. Are rural residents less likely to obtain recommended preventive healthcare services? American Journal of Preventive Medicine 2001; 21: 182-188.
7. Miller MK, Stokes CS, Clifford WB. A comparison of the rural-urban mortality differential for deaths from all causes, cardiovascular disease and cancer. Journal of Rural Health 1987; 3: 23-34.
8. Goins RT, Mitchell J. Health-related quality of life: Does rurality matter? Journal of Rural Health 1999; 15: 147-156.
9. Muus KJ, Baker-Demaray T, McDonald LR, Ludtke RL, Allery AJ, Bogart TA et al. Body mass index and cancer screening in older American Indian and Alaska Native men. Journal of Rural Health 2009; 25: 104-108.
10. Narain V, Cher ML, Wood DP. Prostate cancer diagnosis, staging and survival. Cancer and Metastasis Reviews 2002; 21: 17-27.
11. Powell IJ. Epidemiology and pathophysiology of prostate cancer in African-American men. Journal of Urology 2007; 177: 444-449.
12. Dalton SO, Schüz J, Engholm G, Johansen C, Kjaer SK, Steding-Jessen M et al. Social inequality in incidence of and survival from cancer in a population-based study in Denmark, 1994-2003: Summary of findings. European Journal of Cancer 2008; 44: 2074-2085.
13. Doll R. Urban and rural factors in the aetiology of cancer. International Journal of Cancer 1991; 47: 803-810.
14. Friis S, Storm HH. Urban-rural variation in cancer incidence in Denmark 1943-1987. European Journal of Cancer 1993; 29: 538-544.
15. Higginbotham JC, Moulder J, Currier M. Rural vs. urban aspects of cancer: First-year data from the Mississippi Central Cancer Registry. Family & Community Health 2001; 24: 1-9.
16. Jong KE, Smith DP, Yu XQ, O'Connell DL, Goldstein D, Armstrong BK. Remoteness of residence and survival from cancer in New South Wales. Medical Journal of Australia 2004; 180: 618-622.
17. Baade PD, Youlden DR, Coory MD, Gardiner RA, Chambers SK. Urban-rural differences in prostate cancer outcomes in Australia: what has changed? Medical Journal of Australia 2011; 194: 293-6.
18. Robson B, Purdie G, Cormack D. Unequal impact: Maori and Non-Maori cancer statistics 1996-2001. Wellington: Ministry of Health, 2006.
19. Schouten LJ, Meijer H, Huveneers JAM, Kiemeney LALM. Urban-rural differences in caner incidence in the Netherlands, 1989-1991. International Journal of Epidemiology 1996; 25: 729-736.
20. Vassallo A, De Stéfani E, Ronco A, Barrios E. Urbanization gradients and cancer mortality in Uruguay, 1988-1992. International Journal of Cancer 1994; 59: 345-350.
21. Canadian Population Health Initiative. How Healthy are Rural Canadians? Ottawa, ON: Canadian Institute for Health Information, 2006.
22. Jemal A, Ward E, Wu X, Martin HJ, McLaughlin CC, Thun MJ. Geographic patterns of prostate cancer mortality and variations in access to medical care in the United States. Cancer Epidemiology, Biomarkers & Prevention 2005; 14: 590-595.
23. Marså K, Johnsen N, Bidstrup PE, Johannesen-Henry CT, Friis S. Social inequality and incidence of and survival from male genital cancer in a population-based study in Denmark, 1994-2003. European Journal of Cancer 2008; 44: 2018-2029.
24. Lagacé C, Desmeules M, Pong RW, Heng D. Non-communicable disease and injury-related mortality in rural and urban places of residence. A comparison between Canada and Australia. Canadian Journal of Public Health 2007; 98: S62-69.
25. Nasca PC, Mahoney MC, Wolfgang PE. Population density and cancer incidence differentials in New York State, 1978-82. Cancer Causes and Control 1992; 3: 7-15.
26. Pampalon R, Martinez J, Hamel D. Does living in rural areas make a difference for health in Québec? Health & Place 2006; 12: 421-435.
27. Alam N, You H, Banks C, Baker D, Bishop J. Prostate Cancer in New South Wales. Sydney, NSW: Cancer Institute NSW, 2009.
28. Australian Institute of Health and Welfare. Rural, regional and remote health: indicators of health status and determinants of health. Rural Health Series no. 9. Canberra, ACT: AIHW, 2008.
29. NCIN Data Briefing. The effect of rurality on cancer incidence and mortality. (Online) no date. Available: www.ncin.org.uk/databriefings (Accessed 4 July 2011).
30. Coory MD, Baade PD. Urban-rural differences in prostate cancer mortality, radical prostatectomy and prostate-specific antigen testing in Australia. Medical Journal of Australia 2005; 182: 112-115.
31. Sierra R, Parkin DM, Leiva GM. Cancer in Costa Rica. Cancer Research 1989; 49: 717-724.
32. White A, Coker AL, Du XL, Eggleston KS, Williams M. Racial/ethnic disparities in survival among men diagnosed with prostate cancer in Texas. Cancer 2011; 117: 1080-1088.
33. Rusiecki JA, Kulldorff M, Nuckols JR, Song C, Ward MH. Geographically based investigation of prostate cancer mortality in four U.S. Northern Plain states. American Journal of Preventive Medicine 2006; 30: S101-108.
34. Baade PD, Youlden DR, Krnjacki LJ. International epidemiology of prostate cancer: Geographical distribution and secular trends. Molecular Nutrition and Food Research 2009; 53: 171-184.
35. Mahoney MC, LaBrie DS, Nasca PC, Wolfgang PE, Burnett WS. Population density and cancer mortality differentials in New York State, 1978-1982. International Journal of Epidemiology 1990; 19: 483-490.
36. Valerianova Z, Gill C, Duffy SW, Danon SE. Trends in incidence of various cancers in Bulgaria, 1981-1990. International Journal of Epidemiology 1994; 23: 1117-1126.
37. Yang CY, Hsieh YL. The relationship between population density and cancer mortality in Taiwan. Japanese Journal of Cancer Research 1998; 89: 355-360.
38. Ocaña-Riola R, Sánchez-Cantalejo C, Rosell J, Sánchez-Cantalejo E, Daponte A. Socio-economic level, farming activities and risk of cancer in small areas of Southern Spain. European Journal of Epidemiology 2004; 19: 643-650.
39. Dey S, Zhang Z, Hablas A, Seifeldein IA, Ramadan M, El-Hamzawy H et al. Geographic patterns of cancer in the population-based registry of Egypt: Possible links to environmental exposures. Cancer Epidemiology 2011; 35: 254-264.
40. Smailyte G, Kurtinaitis J. Cancer mortality differences among urban and rural residents in Lithuania. BMC Public Health 2008; 8: 56. (Online) 2008. Available: http://www.biomedcentral.com/1471-2458/8/56 (Accessed 3 April 2012).
41. Hall SE, Holman CD, Wisniewski ZS, Semmens J. Prostate cancer: socio-economic, geographical and private-health insurance effects on care and survival. BJU International 2005; 95: 51-58.
42. Syrigos KN, Karapanagiotu E, Harrington KJ. Prostate cancer in elderly. Anticancer Research 2005; 25: 4527-4534.
43. McLafferty S, Wang F. Rural reversal? Rural-urban disparities in late-stage cancer risk in Illinois. Cancer 2009; 115: 2755-2764.

