full article:
Accidental bowel leakage is rarely disclosed1. It can range from occasional loss of flatus, through staining of underwear with mucus or stool, to faecal incontinence, which the International Continence Society defines as the involuntary loss of liquid or solid stool that is a social or hygienic problem2. The impact of faecal incontinence on quality of life can be debilitating and embarrassing. Stringent coping strategies often alienate the sufferer from friends and family3. There is little awareness of it in the general community. Even sufferers confuse it with diarrhoea, faecal urgency, irritable bowel syndrome or inflammatory bowel disease4.
Risk factors resulting in the development of faecal incontinence include congenital anorectal abnormalities; neurological or spinal damage; obstetric or anal trauma; anal or rectal cancers; inflammatory bowel disease; reconstructive bowel surgery; psychological problems; abdominal/pelvic irradiation; infections; reactions to medications, drugs or diet; rectal prolapse, anal fistula or haemorrhoids; immobility; increasing age; chronic constipation; obesity and poor management of diarrhoea or loose stool; and idiopathic causes5-7.
Internationally, the prevalence of faecal incontinence in community-dwelling adults ranges from 0% to 15.2%8. Australian studies have estimated prevalence between 8%9 and 11%10,11 in community-dwelling adults, with telephone interviews being used in a South Australian study9 and two postal surveys being conducted in Sydney, New South Wales10,11. Studies undertaken at the Townsville Hospital urogynaecology and colorectal outpatient clinics reported up to 26% of patients having the condition, with a considerable effect on their quality of life3,12,13.
Currently up to 72% of nursing home residents suffer with faecal incontinence14. The proportion of Australians over the age of 65 is expected to increase from 13.4% of the population in 2007 to 25.3% of the population by 2047 and the percentage of those over the age of 85 will likely rise from 1.7% to 5.6%15. Many Australians wish to age in their homes rather than move into specialised care16. Increased demand for community-based incontinence services will be due to not only the ageing population15 but also the fact that younger cohorts (baby boomers, X and Y generations) are more assertive and expect to have their needs satisfied17. World War II (age 85-90 in 2012) and post-war cohorts (67-84) are known as the silent generation and are stoic and reserved, respect clinicians and are more likely to comply with their recommendations. This general attitude may be reflected in their reticence to disclose embarrassing issues such as faecal incontinence and reluctance to seek treatment for this condition.
Baby boomers (47-66) are sceptical, questioning and expect pharmacological solutions to their age-related problems17. They are more likely to try novel treatments such as anal implants18, sacral nerve or percutaneous tibial nerve stimulation19 for their faecal incontinence, with the hope of finding a 'magic pill' or panacea17. With compulsory superannuation they are prepared to pay for healthcare services into their old age15. Generation X (36-46) treasure independence, are focused on their quality of life and work to fund their more balanced lifestyle. Their choice of health service providers are likely to be an economic decision and they will require more personalised support, shared decision-making and greater self-management20. The Y generation (18-36) are tolerant team players who value mentorship and have grown up with the internet and frequent technological change. They value excitement and instant gratification through email, messaging and social media and may adopt healthcare services via social media17; they may prefer self-management of faecal incontinence using telephone and tablet applications. For this and other age-related health problems the Ottawa Charter action areas of building healthy public policy, creating supportive environments, strengthening community action, developing personal skills and reorienting health services provide a useful framework to develop prevention, early intervention and treatment protocols21.
This study was undertaken to investigate personal demographics, health/risk factors, bowel habits, nutrition intake (fibre and fluid) and physical activity in relation to faecal incontinence. Here the authors report the prevalence of faecal incontinence in community-dwelling adults and assess disclosure of accidental bowel leakage in regional and rural areas of North/Far North Queensland, Australia.
A sample of 3620 subjects was compiled from the 2006-2007 Cairns and Townsville White Pages® telephone directories (Cairns: 1112 urban, 481 rural, 226 remote; Townsville: 1049 urban, 432 rural, 320 remote) using systematic random sampling. The first non-business name and address was selected from each column between 90-100 mm and 190-200 mm from the top of the page. The sample addresses were updated from the 2007-2008 printed and online White Pages® directory prior to distribution. Where a subject no longer appeared in the directory the next alphabetically listed private address was chosen as a replacement.
The self-administered questionnaire (Appendix 1), a study information sheet, an incentive leaflet (return of the completed survey provided an opportunity to win a $250 or $50 voucher), an opaque incentive response envelope, and a reply-paid envelope were mailed to everyone on the database. The survey contained 62 questions in five sections including personal demographics (11) with additional questions about female obstetric history (8), health/risk factors (6), bowel habits (32), nutrition (fibre and fluid intake) (2) and physical activity (3).
Demographic questions included those related to age, gender, cohabitation, residence type, postcode, education level, country of birth, height, weight and indigenous status. Female obstetric history questions included menopausal status, number of natural and caesarean births, use of forceps/vacuum, episiotomy, post-tear stitches, hysterectomy and hormone replacement therapy. Health or risk factor questions included participant-perceived general health as well as factors, previously identified, as potentially causal for this condition5-7 (Appendix 1).
Participants were asked for information about year and results of a colonoscopy. Questions about bowel habits included those related to frequency of defecation; stool type22; urgency; difficulty in emptying bowels; constipation and straining; accidental anal leakage and its effect on quality of life; and coping strategies used. Nutrition questions related to fluid and fibre intake. Activity questions related to normal daily activity, exercise and pelvic floor exercises.
The initial survey was mailed out in July 2007, followed by two follow-up surveys mailed to non-responders in September 2007 and January 2008. One hundred random non-responders were invited to answer the survey by phone in February 2008. All mail-outs contained reply-paid envelopes and covering letters explaining the purpose of the study. The follow-up mail-outs included a non-response/remove me from the database tool seeking reasons for non-response. Anonymity of replies was maintained. Core faecal incontinence questions were adapted from the Cleveland Clinic Florida Fecal Incontinence Score (Wexner) which correlates well with clinical presentation of faecal incontinence and has been referred to as 'a tool of choice' for assessing faecal incontinence in community-dwelling older adults23.
In this study, faecal incontinence was defined as accidental leakage of solid or liquid stool in the previous 12 months that was not caused by a virus, medication or contaminated food.
Statistics
Community prevalence and 95% confidence intervals (CI) of faecal incontinence and accidental bowel leakage were determined. Numerical data are given as mean value and standard deviation or median value and interquartile range (IQR), depending on the distribution. Comparisons between characteristics were conducted using χ2 tests and χ2 tests for trend, nonparametric Wilcoxon tests, and student's t-tests. Statistical analyses were conducted using Statistical Package for the Social Sciences Windows v17 (SPSS Inc., http://www.spss.com). Throughout the analysis p<0.05 was considered statistically significant. The sample size calculation was based on the Sydney, New South Wales community postal surveys (faecal incontinence prevalence = 11%)10,11. The authors expected the North/Far North Queensland prevalence to be greater. Choosing 12.1% (10% higher) with a confidence level of ± 2% (ie true level to be between 10.1% and 14.1%), the sample size calculation for each of the Townsville, Cairns and rural settings was 1022. Systematic random sampling generated 3620 private names for the database.
Ethics approval
James Cook University Human Research Ethics Committee granted ethical approval (H2630).
Of the 3620 surveys mailed out, 432 were returned to sender as 'no longer at this address' (18 of these were reported deceased); nine completed surveys were invalid (one under-age, two nursing home residents, six living outside North/Far North Queensland). Fourteen of the 100 random numbers telephoned were inactive phone numbers. Of the remaining 3165 potential participants, 95 gave reasons for not wishing to participate; 1523 questionnaires were returned giving a response rate of 48.1%.
Valid responses were from 628 (41.0%) men and 891 (58.5%) women. Male respondents were older than female respondents (mean age: 57.3 (95% CI: 56.2-58.5) vs 52.3 (51.3-53.4) years, p<0.001) and had a marginally higher mean body mass index (27.6 (27.3-28.0) vs 26.7 (26.3-27.1), p=0.001). Although more women (11.3%) than men (7.8%) resided alone (p<0.001), there were no significant differences between the genders with regard to type or location of residence, level of education, occupation type, country of birth or indigenous status.
The prevalence (95% CI) of faecal incontinence was 12.7% (10.9-14.5%, Table 1). Overall, there was no significant difference between genders (men 12.6%, women 12.8%) or residence (regional centre or rurally). Prevalence increased significantly with age in men (18-39: 3/65, 4.6% (0.0-9.9%); 40-59: 31/251, 12.4% (8.3-16.4%]; >60: 38/252, 15.1% (10.6-19.5%], p=0.034) but not in women, although it was similar for both genders over 40 years of age (Fig1).
Of those with faecal incontinence, 54 (34.8%) reported urge incontinence, 19 (12.3%) passive incontinence and 44 (28.4%) both passive and urge incontinence. Only 28.1% of incontinent participants could always differentiate between flatus and stool compared with 53.5% of continent respondents (p<0.001, Table 2). Nocturnal bowel leakage was a problem for 41 (26.3%) of incontinent respondents. While 59 (36.6%) incontinent respondents had problems modifying their diet to control their bowel, 49 (32.7%) did not know which medications would control their bowels and 56 (34.1%) felt they had no control over their bowels; only 11 (7.1%) had sought treatment for this condition. More than 38% (64) of respondents with faecal incontinence reported having first degree relatives with 'bowel problems'.
Most incontinent participants (130/174, 74.7%) reported incontinent episodes occurring less than once per month, 32 (18.4%) more than once a month, 10 (5.7%) more than once a week, and 2 (1.1%) at least once per day (Table 3, Fig2). More than half the incontinent respondents (91, 53.2%) and 245 (18.6%) of continent respondents reported bowel leakage due to an acute illness. There were no statistically significant gender, age or rurality differences for acute illness-related accidental bowel leakage.
A comparison of respondents with and without incontinence showed that more with incontinence reported poor or very poor general health; more frequent, irregular, incomplete/fragmented defecation, and looser stools; inability to delay by 15 minutes or requiring aids to defecate; alternating diarrhoea and constipation; a history of constipation; and pad-wearing (all p<0.001, Table 2). In addition, more incontinent respondents reported that their bowel function negatively affected their daily activities such as work, sports, housework/gardening, social activities, travel, relationships and sex life (all p<0.001). More than half the incontinent respondents (82/162) reported difficulty finding public toilets.
Embedded within the survey were a number of questions relating to accidental bowel leakage in addition to the Wexner score5. Table 1 presents the disclosure of accidental bowel leakage with the most severe element counted for each respondent. Those who did not report faecal incontinence, but disclosed accidental bowel leakage due to an acute illness, are reported separately in Table 1 (n=245, 97 male) even if they also reported other accidental bowel leakage items. Stool-related accidental bowel leakage (18.2%) included faecal incontinence, soiling with flatus (not including possible mucus-related soiling with flatus, n=3, one male), and soiling with urgency. This was not statistically different for gender or location/rurality of respondents' residences but was significantly higher for the over-40 age groups (p=0.040). Possible stool-related accidental bowel leakage (3.0%) included leakage of mucus; staining underwear, if it was considered a problem in the month prior to completing the survey; accidental bowel leakage that was found bothersome; and passive accidental bowel leakage. Of the remaining 1109/1513 respondents who completed these questions 346 (22.9%) reported they could not always differentiate between flatus and stool, 69 (4.6%) reported accidental leakage of flatus and only 532 (35.2%) respondents reported no concerns about accidental bowel leakage, and no accidental bowel leakage.
Types of accidental anal leakage are presented in Table 4 and did not differ across regions. Leakage rates of solid and liquid stool, mucus and accidental flatus were similar for both genders although staining of underwear was significantly higher in men (26.8%) than in women (17.3%, p<0.001) and for older respondents (<39: 15.9%; 40-59: 28.8%; >60: 33.7%, p<0.001).
Table 1: Respondents' disclosure of accidental bowel leakage
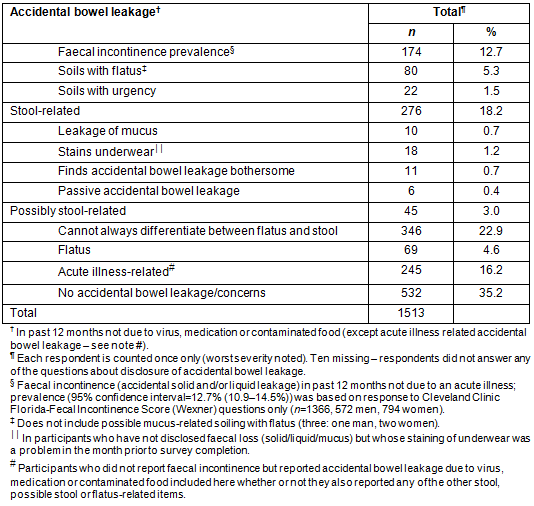
Table 2: Comparison of bowel habits and health in respondents with and without faecal incontinence
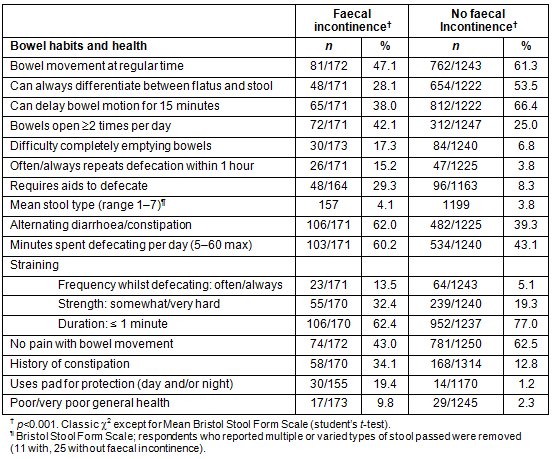
Table 3: Frequency of accidental faecal leakage in respondents?
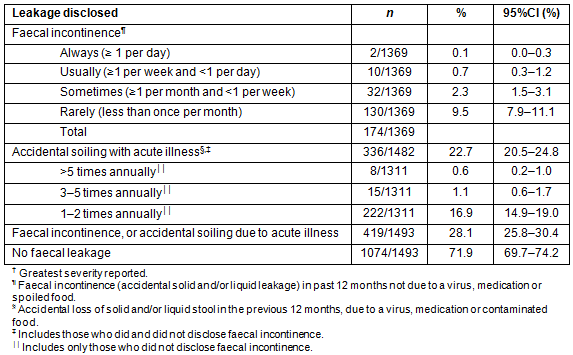
Table 4: Accidental anal leakage in respondents?
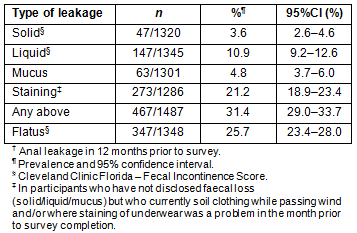
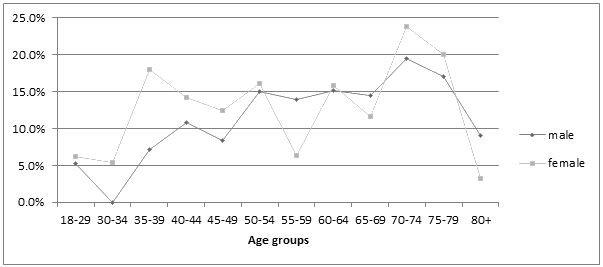
Figure 1: Proportion of respondents with faecal incontinence.
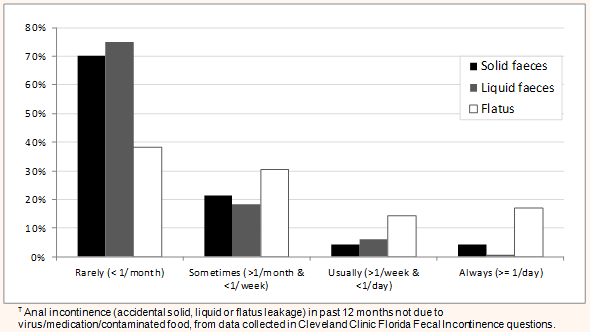
Figure 2: Frequency of anal leakage? in respondents with faecal incontinence
Discussion
The main finding of this study was that 12.7% of adult North/Far North Queensland community members reported faecal incontinence in the previous 12 months that was not due to an acute illness. This rate increased with age for men although, in total, there were no gender or locality differences. When soiling with flatus and urgency were included, stool-related accidental bowel leakage was substantially higher, at 18.2%.
The prevalence of faecal incontinence is highly dependent on the definition applied10,24. This study's focused definition - 'accidental loss of solid and/or liquid stool in the previous twelve months, not due to a virus, medication or contaminated food' - has not, to the authors' knowledge, been used previously. Despite using this refined definition the rate of faecal incontinence in this study is higher than the two earlier Australian postal surveys10,11 although the severity (type and frequency) was similar. Using a broader definition of accidental stool leakage that did not exclude faecal incontinence resulting from an acute illness, the prevalence was much higher, at 28.1% (Table 3). Thus previous concerns of high levels of faecal incontinence in the North/Far North Queensland community are warranted3,12,13.
One strength of this survey was that it enabled respondents to disclose any type of accidental bowel leakage, whether they termed it faecal incontinence or not. In a recent New Zealand study, researchers combined three different measures - a bowel control problem, quality of life impairment and faecal incontinence ≥1/month - to better determine faecal incontinence prevalence in the community25. While the researchers reported 12.4% of respondents leaked solid or liquid stool at least once per month and 26.8% had impaired quality of life due to accidental bowel leakage, they calculated community prevalence at 13.2% using the overlap of at least two of three measures to redefine faecal incontinence. The aim of the present study too was to provide a more accurate estimate of those suffering with faecal incontinence. In this study, issues such as flatus and mucus were excluded. The New Zealand survey tool may have included these in the quality of life component26, thereby overstating the prevalence. Up to 50% of studies previously reviewed8,24 calculated prevalence using faecal incontinence occurring in the previous 12 months as done in the present study. As in those studies, the rate in this study may be higher than that of the New Zealand study and others using incontinence reported over shorter time frames.
A troubling result is that only 35% of the respondents had no bowel concerns. Further, many respondents with faecal incontinence felt they had no control over their bowels and didn't know how to manage their diet or which medications to take to mitigate the problem. As only 7% actually sought medical advice, fear, embarrassment and the stigma associated with faecal incontinence continues to hamper disclosure as previously identified27.
As more people age 'in place'16 the prevalence of faecal incontinence in the community is likely to increase toward that of aged-care facilities. Younger cohorts, which are less stoic, are unlikely to tolerate this condition and will demand treatment17,20. Ageing populations increase healthcare costs15, and increased demand for faecal incontinence treatment will further escalate these costs. The concepts of the Ottawa Charter for Health Promotion21 can be used to help reduce this added financial burden by encouraging prevention of constipation and obstetric damage, and ensuring benign clinical diagnoses are managed with conservative programs (potentially self-managed) including dietary management, pelvic floor exercises, defecation and relaxation techniques, and home biofeedback28.
Limitations of this study include a low response rate of 48.1% despite mailing two follow-up questionnaires. The average age of survey respondents was 54 years, 9 years older than the North/Far North Queensland adult population (45 years), which is marginally younger than the state (46 years) and national (46.5 years) averages29. Reasons for the low response rate could include lack of interest in the subject matter, particularly among younger survey recipients. Furthermore, younger people may be under-represented, not only because they may have chosen not to respond to the questionnaire, but also because they are more likely to only have a mobile telephone and thus not have received a survey because they were not listed in the telephone directory30. Older respondents may have self-selected due to a particular interest in the topic or because of a personal bowel issue24. Townsville, the largest city in North Queensland, has a highly mobile population with a 35% net transient population measure31. Of the sample of 100 non-respondents telephoned in an attempt to further increase the response rate, 14 telephones had been disconnected. If this were representative of the population, it could also partially explain the low response rate. However, using the alternative data collection method of telephone interviews may have yielded an even lower response rate based on experience from the South Australian study9. Similar response rates using this or analogous study enrolment methods were found in a cross-sectional study investigating accidents and injuries in North Queensland32 and a New Zealand faecal incontinence prevalence study using a random selection from the Canterbury electoral roll33. Thus the response rate may be a limitation of the method used to elicit information about a topic that is either 'sensitive' or does not interest a particular segment of the population.
There is a high level of untreated faecal incontinence in the North/Far North Queensland community which degrades the quality of life of those with this ailment. With a burgeoning ageing-in-place population the prevalence in this and other communities is likely to increase. Lifestyle education to prevent chronic constipation and encourage the development and maintenance of strong pelvic floor muscles among young people could reduce the prevalence of this condition. Faecal incontinence, its prevalence, prevention and treatments should be widely discussed to raise community awareness reducing the incidence and the destructive effect it has on the quality of so many lives. Eliminating the stigma of faecal incontinence should encourage those with the condition to overcome their embarrassment and seek early treatment. Where appropriate, conservative self-managed care is economically rational and can improve quality of life for all, not just those with access to the few specialist biofeedback clinics.
References
1. Whitehead WE. Diagnosing and managing fecal incontinence: if you don't ask, they won't tell. Gastroenterology 2005; 129: 6.
2. Norton C, Whitehead W, Bliss D, Metsola P, Tries J. Conservative treatment and pharmacological management of faecal incontinence in adults. In: A Abrams, L Cardozo, S Khoury, A Wein (Eds). Incontinence. Paris: International Continence Society, 2005; 1521-1564.
3. Bartlett L, Nowak M, Ho YH. Impact of fecal incontinence on quality of life. World Journal of Gastroenterology 2009; 15: 3276-3282.
4. Bliss DZ, Norton C, Vodusek DB. Raising awareness about fecal incontinence. Neurology and Urodynamics 2010; 29: 612-615.
5. Jorge JM, Wexner SD. Etiology and management of fecal incontinence. Diseases of the Colon and Rectum 1993; 36: 77-97.
6. Norton C, Chelvanayagam S. Causes of faecal incontinence. In: C Norton, S Chelvanayagam, (Eds). Bowel continence nursing. Beaconsfield, Buckinghamshire: Beaconsfield Publishers Ltd, 2004; 23-32.
7. Whitehead WE, Borrud L, Goode PS, Meikle S, Mueller ER, Tuteja A et al. Fecal incontinence in US adults: epidemiology and risk factors. Gastroenterology 2009; 137(2): 512-517.
8. Pretlove SJ, Radley S, Toozs-Hobson PM, Thompson PJ, Coomarasamy A, Khan KS. Prevalence of anal incontinence according to age and gender: a systematic review and meta-regression analysis. International Urogynecology Journal and Pelvic Floor Dysfunction 2006; 17: 407-417.
9. Hawthorne G. Measuring Incontinence in Australia. (Online). 2006. Available: http://www.bladderbowel.gov.au/assets/doc/ncms/Phase1-2InformationAndEvidence/14OutcomesMeasuresTrialSAHOS.pdf (Accessed 24 October 2012).
10. Kalantar JS, Howell S, Talley NJ. Prevalence of faecal incontinence and associated risk factors; an underdiagnosed problem in the Australian community? Medical Journal of Australia 2002; 176: 54-57.
11. Lam TCF, Kennedy ML, Chen FC, Lubowski D, Talley NJ. Prevalence of faecal incontinence: obstetric and constipation risk factors: a population based study. Colorectal Disease 1999; 1: 197-203.
12. Ho YH, Muller R, Veitch C, Rane A, Durrheim D. Faecal incontinence: an unrecognised epidemic in rural North Queensland? Results of a hospital-based outpatient study. Australian Journal of Rural Health 2005; 13: 28-34.
13. Bartlett L, Nowak M, Ho YH. Reasons for non-disclosure of faecal incontinence: a comparison between two survey methods. Techniques in Coloproctology 2007; 11: 251-257.
14. Australian Institute of Health and Welfare (AIHW). Australian incontinence data analysis and development. AIHW cat no. DIS 44, 2006; 119.
15. Productivity Commission (2008). Future demand for aged care services: trends in aged care services: some implications. Commission research paper. Canberra, ACT: Australian Government.
16. Australian Institute of Health and Welfare (AIHW). The desire to age in place among older Australians volume 1 - reasons for staying or moving. AIHW bulletin. Cat. no. AUS 169, 2013.
17. Berkowitz EN, Schewe CD. Generational cohorts hold the key to understanding patients and health care providers: coming-of-age experiences influence health care behaviors for a lifetime. Health Marketing Quarterly 2011; 28: 190-204.
18. Bartlett L, Ho YH. PTQ? anal implants for the treatment of faecal incontinence. British Journal of Surgery 2009; 96: 1468-1475.
19. Maeda Y, O'Connell PR, Matzel KE, Laurberg S. Sacral nerve stimulation for fecal incontinence: at a crossroad and future challenges. Diseases of the Colon and Rectum 2012; 55: 621-624.
20. Keckley P. 2010 Survey of Health Care Consumers: key findings, strategic implications. (Online). 2010. Available: http://www.deloitte.com/assets/Dcom-UnitedStates/Local%20Assets/Documents/US_CHS_2010SurveyofHealthCareConsumers_050310.pdf (Accessed 5 September 2012).
21. World Health Organization (WHO). The Ottawa Charter for Health Promotion. (Online). http://www.who.int/healthpromotion/conferences/previous/ottawa/en (Accessed 28 October 2013)
22. Lewis SJ, Heaton KW. Stool form scale as a useful guide to intestinal transit time. Scandinavian Journal of Gastroenterology 1997; 32: 920-924.
23. Fallon A, Westaway J, Moloney C. A systematic review of psychometric evidence and expert opinion regarding the assessment of faecal incontinence in older community-dwelling adults. International Journal of Evidence-based Healthcare 2008; 6: 225-259.
24. Macmillan AK, Merrie AE, Marshall RJ, Parry BR. The prevalence of fecal incontinence in community-dwelling adults: a systematic review of the literature. Diseases of the Colon and Rectum 2004; 47: 1341-1349.
25. Sharma A, Marshall RJ, Macmillan AK, Merrie AE, Reid P, Bissett IP. Determining levels of fecal incontinence in the community: a New Zealand cross-sectional study. Diseases of the Colon and Rectum 2011; 54: 1381-1387.
26. Macmillan AK, Merrie AE, Marshall RJ, Parry BR. Design and validation of a comprehensive fecal incontinence questionnaire. Diseases of the Colon and Rectum 2008; 51: 1502-1522.
27. Bartlett L, Nowak M, Ho YH. Reasons for non-disclosure of faecal incontinence: a comparison between two survey methods. Techniques in Coloproctology 2007; 11: 251-257.
28. Bartlett LM, Sloots K, Nowak M, Ho YH. Biofeedback therapy for faecal incontinence: a rural and regional perspective. Rural Remote Health 11(1): 1630. (Online) 2011. Available: www.rrh.org.au (Accessed 24 October 2013).
29. Australian Bureau of Statistics. Population by age and sex, Australian states and territories, June 2010. ABS cat. no. 3201.0, 2010.
30. Call K, Davern M, Boudreaux M, Johnson P, Nelson J. Bias in telephone surveys that do not sample cell phones: uses and limits of poststratification adjustments. Medical Care 2011; 49: 355-364.
31. Cummings Economics. Comparative transient populations in Australian cities. (Online) 2011. Available: http://www.cummings.net.au/pdf/recent/J2276%20CE%20Note%20Comp%20Trans%20Pop'n%20Levels.pdf (Accessed 15 August 2011).
32. Carter A, Muller R. Practices, knowledge and perceptions influencing accident and injury in the Mackay/Whitsunday community. In: Reducing injuries in Mackay, North Queensland. Warwick, Queensland: Warwick Educational Publishing, 2002; 53-73.
33. Lynch AC, Dobbs BR, Keating J, Frizelle FA. The prevalence of faecal incontinence and constipation in a general New Zealand population; a postal survey. New Zealand Medical Journal 2001; 114: 474-477.
___________________________
Appendix 1: North Queensland Bowel Habit Project Survey


