Introduction
In children under 6 years of age, early childhood caries (ECC) is a condition characterized by the presence of one or more decayed, missing, or filled primary teeth1. Among young Canadian Inuit children, the prevalence of ECC has been reported to range from 50 to 97%, but recent statistics are not available2-5. Overall, caries are a considerable problem among Canadian Aboriginal children in contrast to the general population who experience much lower rates of ECC6-10.
Early childhood caries has a complex etiology. It is affected by the amount of cariogenic bacteria, the presence of cariogenic foods, and the susceptibility of host tooth11,12. Other environmental and behavioural factors such as oral hygiene habits, socioeconomic status, enamel hypoplasia, fluoride exposure, previous caries experience, access to dental care and other dietary factors also impact caries development10-12. Early childhood caries may lead to pain, improper speech development, reduced ability to chew, low weight-for-age, lower self-esteem and sleep disturbances; however, the evidence for many of these associations is only weak or moderate6,13.
Historical evidence suggests that dental caries were rare among Alaska Natives in the early 20th Century14-17. An Arctic nutrition transition, particularly the reduced intake of traditional food and an increased consumption of high-sugar foods, likely contributed to widespread ECC in Inuit communities18. Various studies as early as the 1970s have reported that commercially available market food makes up a significant proportion of the Inuit diet, especially among young children19-21. In addition, studies among Canadian Inuit adults and Dene/Métis adults and children show that diets higher in market foods are also higher in simple carbohydrates22,23. Among Dene/Métis children and Inuit women, percent energy from sucrose was reported to be 18-19% and 12-17%, respectively22. It is likely that the Arctic nutrition transition has been exacerbated by other oral health risk factors common in these regions, including the practice of giving bottles to children at bedtime, lack of fluoridated water, inadequate access to dental health care, and lack of education about preventative measures7,24,25.
Smaller studies reported that children in Nunavut experience a high prevalence of tooth decay and dental caries. The main objective of this study was to provide the prevalence and correlates of parental-reported oral health of Inuit preschoolers, with a focus on dietary and socioeconomic risk factors.
Methods
Study population and ethics
The data presented in the current report were from the Nunavut Inuit Child Health Survey, and details of the methodology have been presented elsewhere26. In brief, 16 of the 25 communities in Nunavut were selected to participate in the survey, representing all three regions of the territory. Communities were selected based on region, population size, latitude, and logistic feasibility due to flight routes and financial costs relative to the number of age-appropriate children. Inuit children, ages 3 to 5 years, were randomly selected to participate in the survey. Recruiters were instructed to make three attempts to reach each household with a 3-5 year old. Written informed consent was obtained from the child's primary caregiver, the majority of which were either a biological parent (68.4%) or an adoptive parent (21.6%). Of the 537 households successfully contacted, 75 (11.6%) refused on initial contact and 74 (13.8%) accepted but later cancelled or did not show for the interview, providing a participation rate of 72.3% (388) over the 2 years of data collection.
Certification of Ethical Acceptability for Research Involving Human Subjects was obtained from the McGill Faculty of Medicine Institutional Review Board. A Scientific Research License was obtained from the Nunavut Research Institute.
Participatory research methods
Participatory research methods were implemented through all stages of the survey work. The Nunavut Inuit Child Health Survey was developed by a steering committee made up of representatives from partner Inuit, community, and territorial organizations and McGill University and University of Toronto. Research agreements were signed between communities and the research centers before commencing research.
Questionnaires
After giving written, informed consent, interviews were conducted with parents and primary caregivers about their child's health history, home environment and dietary habits. Because the questionnaires were long, to minimize research burden the oral health history was limited to two questions:
- How would you rate your child's oral and dental health - very good, good, fair, poor?
- Does your child have any decayed, extracted or filled baby teeth?
Caregiver's rating of a child's oral health has been used successfully in a previous study with young children27. The answers to these questions were given by the parents and caregivers and no objective clinical assessments were made. When caregivers reported that their child had 'decayed, extracted or filled baby teeth', children were classified as having reported-caries experience (RCE). Finally, caregivers were asked if their child was taking any nutritional supplements such as fluoride, vitamin D or multivitamin and multimineral supplements. Brands were recorded to allow ingredients to be determined.
Dietary intake questionnaires
Each caregiver was asked to complete a qualitative food frequency questionnaire (FFQ) for their child. The FFQ was designed to capture past-month information about common traditional foods that are available in the three regions of Nunavut. It also captured past-month frequency of milk and various high-sugar food consumption. One 24 hour dietary recall was also conducted for each child participant by training interviewers on a four-stage, multiple pass interviewing technique. Food model kits were used to estimate portion sizes. A second repeat 24 hour dietary recall was collected on a 20% sub-sample of participants. Food frequency information was entered using EpiInfo (CDC, USA; http://www.cdc.gov/epiinfo/) and data were double verified. Twenty-four hour dietary recall information was entered using CANDAT (Godin London Inc; London, ON, Canada; http://www.candat.ca/) and was double verified.
Statistical analyses
Weighted-prevalence rates and 95% confidence intervals (CI) for RCE as well as for caregiver-reported oral health ratings were calculated. From the qualitative FFQ, the percent of children who consumed traditional food, milk and various high-sugar food/beverages was determined. The mean number of times per day these foods were eaten was also calculated. Milk and sugar intake in grams per day was also determined from the 24 hour recall. In univariate and multivariate analyses, the outcome variable of interest was whether or not the child had RCE. Frequencies of milk, high sugar food and traditional Inuit food intake were examined as risk factors using Student's t-test. Other factors such as nutritional supplement use, daycare attendance and socioeconomic variables, including type of housing (public vs private) and recipient of income support in the household were examined using a χ2 test; relative risks (RR) and 95% CI were calculated. When risk factors were significantly associated with RCE, they were included in a multiple logistic regression model to examine independent associations. For all analyses, a p-value ≤0.05 was considered significant. Twenty-four hour recall analyses and weighted prevalence estimates were performed in SAS v9.2 (SAS Institute Inc; Cary, NC, USA), while all other analyses were performed using Stata 10 (Stata Corp; TX, USA; http://www.stata.com/stata10/).
Results
Population characteristics
Fifty-three percent (204/388) of the participating children were female and the mean age was 4.4 ± 0.9 years. Cultural, socioeconomic, and health indicator characteristics have been presented elsewhere26. Unweighted and weighted prevalence estimates were similar. A weighted 69.1% (95% CI: 63.7-74.4%) of children had RCE (Table 1). No differences were found in the prevalence of RCE by region (χ2 p = 0.41). While 23% of children currently took any vitamin or supplement, very few children took a fluoride supplement (4.6%, 95% CI: 2.3-6.9%) or a vitamin D supplement (4.9%, 95% CI: 2.4-7.4%). Sixteen percent of children (95% CI: 12.3.-20.1%) were taking a multivitamin and multimineral supplement containing vitamin D and calcium but not fluoride. Very few parents rated their child's oral and dental health as 'very good': 9.5% (95% CI: 6.0-13.0%); whereas, the majority rated their child's health as 'good': 44.5% (95% CI: 38.9-50.1%), 'fair': 29.5% (95% CI:24.3-34.7%), or 'poor': 16.6 (95% CI:12.3-20.9%). The percentage of children with RCE varied by parental-reported oral health rating category (χ2 = 92.57, p <0.001) (Fig1). Children reported to have 'very good' oral health had no RCE, but there was no significant difference in the percentage of children with RCE between the 'good' and 'fair' categories, while more children had RCE in the 'poor' oral health category.
Table 1: Weighted prevalence of reported caries experience and other related oral health indicators among Inuit preschoolers: Nunavut Inuit Child Health Survey, 2007-2008
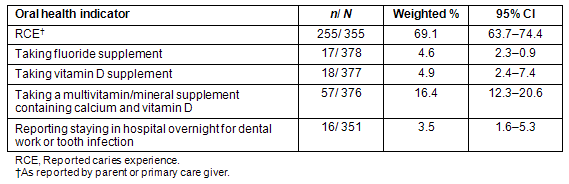
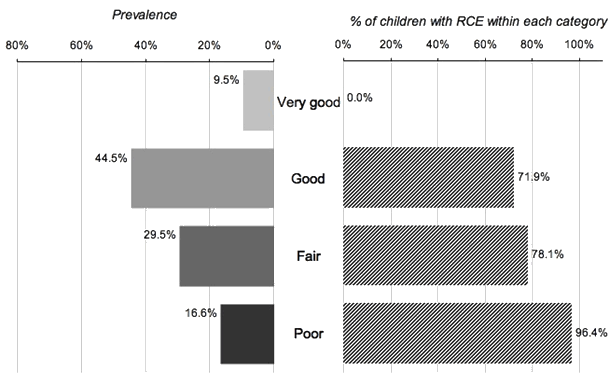
Figure 1: Weighted prevalence of Inuit preschoolers' oral/dental health rating, and within each category, the percentage of children with reported caries experience, as reported by parent or primary care giver. Nunavut Inuit Child Health Survey, 2007-2008; (n = 354).
Food frequency information
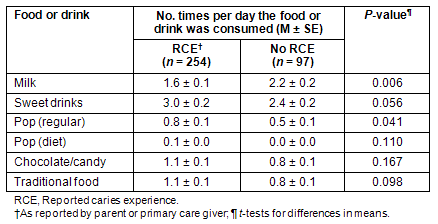
Using the qualitative FFQ information, 88.7% (378/380) of caregivers reported that their child drank milk in the past month. Among consumers, the mean frequency of intake was 2.0 times per day. Many children also drank sweet drinks (88.7%, 337/380) on average 3.2 times per day, while 68.2% (259/380) drank regular soda pop on average 1.0 times per day. A few children drank diet soda pop (4.2%, 16/380) on average 0.9 times per day. Finally, 93.2% consumed chocolate or candy and mean intake frequency was 1.1 times per day. When asked what their child's preferred brand of breakfast cereal was, 72.6% (276/380) reported a high-sugar brand.
Univariate analyses revealed that children with RCE drank milk less often and soda pop more often than children with no RCE (Table 2). Children with RCE drank milk 1.6 ± 0.1 times per day, whereas children without RCE drank milk 2.2 ± 0.2 times per day (p ≤0.01). Children with RCE drank regular soda pop 0.8 ± 0.1 times per day compared with children without RCE who drank soda pop 0.5 ± 0.1 times per day (p ≤0.05). Children with RCE consumed slightly higher sweet drinks (3.0 ± 0.2 times per day) than children with no RCE (2.4 ± 0.2) but this difference only approached statistical significance (p = 0.06). No differences were seen in chocolate or candy consumption (p = 0.17) or in overall traditional food intake (p = 0.09). However, when all high-sugar foods from the FFQ were combined, children with RCE consumed high-sugar foods more often than children without RCE (4.9 ± 0.3 vs 3.7 ± 0.3 times/day, respectively, p ≤0.01).
Table 2: Differences in mean frequency of milk, high-sugar food and traditional food intake among preschoolers with and without reported caries experience. Nunavut Inuit Child Health Survey, 2007-2008
Twenty-four hour dietary recall information
Milk intake was also higher among children without RCE than with RCE, based on data from the 24 hour recall, which provided quantitative information. Children without RCE drank 325.6 ± 44.8 g/day of milk, whereas children with RCE drank 225.9 ± 17.0 g/day (p = 0.01). However, there were no differences in past-day sugar intake between the two groups (RCE: 109.3 ± 5.3 g/day, no RCE: 110.0 ± 6.6 g/day, p = 0.944).
Socioeconomic characteristics, vitamin/supplement use, and daycare attendance were evaluated (Table 3). Children living in public housing were more likely to have RCE than those living in private housing (74% vs 61.5%, respectively, p <0.05), and children taking vitamins or supplements containing either vitamin D, calcium, or fluoride were less likely to have RCE than children not consuming these vitamins/supplements (60.0% vs 75.5%, respectively, p <0.01).
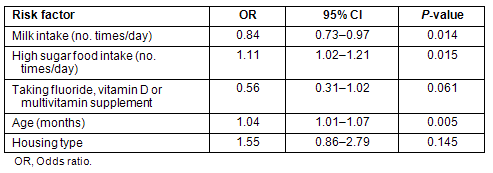
The multiple logistic regression model predicting an outcome of RCE contained frequency of milk intake, frequency of high sugar food intake, nutrition supplement use, child's age, and housing type (Table 4). In these analyses, taking a fluoride, vitamin D or multivitamin supplement was no longer significant. However, frequency of milk consumption remained significantly associated with RCE (OR = 0.84, 95% CI: 0.73-0.97) where an increase in milk consumption by 1 glass per day reduced the odds of having RCE by 16%. Frequency of high sugar food intake also remained significantly positively associated with the prevalence of RCE (OR = 1.11, 95% CI: 1.02-1.12) where an increase of one item per day of high-sugar food corresponded to an 11% increase in the odds of having RCE.
Table 3: Univariate analyses of risk factors for having reported caries experience among Inuit preschoolers: Nunavut Inuit Child Health Survey, 2007-2008
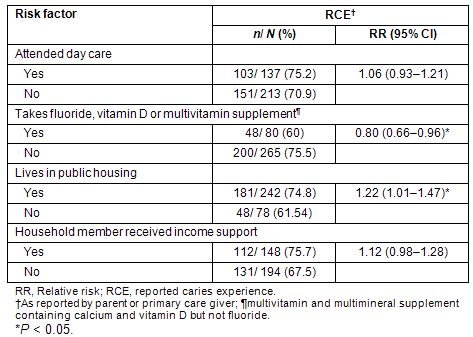
Table 4: Multiple logistic regression to examine risk factors for having reported caries experience, as reported by parent or primary care giver (n = 310)
Discussion
The current study identified a high prevalence of caries-experience among Inuit preschoolers (69.1%), as reported by parents and caregivers. Studies from the early 1990s in Nunavut report high rates of ECC among young Inuit children2,3,5. This is consistent with findings from other Inuit regions. Among Alaska Yup'ik (Natives), 77.4% of 2 to 5 year-olds had caries28. Among Inuit in Nunatsiavut Labrador, 97% of the population aged 5 to 22 years had dental caries, and among children 5 to 6 years, 68% were affected by severe tooth decay4. Our findings are also consistent with more recent findings from Canadian First Nations communities. Among 3-5 year-old Ontario First Nations' children, 74% had one or more caries29. Another randomized control trial conducted in First Nations communities in northern Ontario reported a baseline prevalence of caries among children aged from 6 months to 5 years of 69-73%25. In a study of 408 preschoolers in two disadvantaged and two First Nations communities in Northern and Southern Manitoba, there was a 53.7% prevalence of ECC, as determined by clinical examination30. In contrast, the prevalence of ECC among young children in two major Canadian urban areas ranged from 4.6 to 11%6,7. Among US pre-schoolers, the prevalence of ECC was 27.9% in 1999-200031. In northwestern Ontario, non-Aboriginal preschoolers experienced a 31.1-35.2% prevalence of ECC, whereas off-reserve Aboriginal preschoolers experienced rates of 73.8-81.6% and rates were even higher among on-reserve preschoolers9. Overall, caregiver reports have revealed that Inuit preschoolers across Nunavut experience high rates of caries, as is the case in many other Canadian Aboriginal communities, while the general population experience much lower rates.
It was noted that when parents classified their child's oral health as 'very good' no RCEs were reported, and that among those who rated their child's oral health as poor, 96.4% had RCEs. In contrast, the terms 'good' and 'fair' were associated with similar prevalence rates of RCEs. As the number of teeth affected by caries was not assessed, it is difficult to determine the validity of the terms 'good' and 'fair' using the prevalence of any RCE. In a small sample of US preschoolers, children whose mothers' gave them an oral-health rating of fair to poor had a significantly higher prevalence of ECC (66%) than those who were given a rating of good to excellent (29%)32. In a study evaluating attitudes toward caries in two First Nations communities and two disadvantaged communities in urban areas in Manitoba where the prevalence of ECC is high, 78.3% of caregivers whose children had ECC disagreed that rotten teeth could affect their child's health, whereas only 21.7% of caregivers whose children were caries-free disagreed30,33. These findings lead to an interesting hypothesis that a high prevalence of caries in a population may result in caregivers' easier acceptance of sub-standard oral health among preschoolers, which in turn would represent a challenge for dental health and nutritional educational campaigns.
Higher milk intake was independently and significantly associated with having no RCE. In other studies, evidence is available for a protective mechanism of milk for good oral health. In a pediatric population, children with caries, when compared with those without, had lower median intakes of milk at 2-3 years of age34. In another study, preschoolers from a Canadian Arctic region had significantly lower odds of severe tooth decay when they drank milk regularly7. Milk and milk product consumption was also protective against the development of root caries in an elderly Japanese population35. Vitamin D is important in bone mineralization and, although the mechanisms are still unclear, vitamin D was found to affect enamel and dentin mineral densities in a study comparing vitamin D receptor deficient and receptor positive mice36. Milk and milk products are also a source of casein and an in-vitro study found that natural casein phosphopeptides contained in yoghurt inhibited demineralization and promoted remineralization of dental enamel37. Casein phosphopeptides may be protective by stabilizing calcium phosphate on tooth surfaces. Thus, further research is warranted on the association of milk and/or its constituents with dental health.
While soda pop intake was significantly associated with RCE, sugar-sweetened beverages, chocolate and candy intake were not when considered separately. When all high-sugar foods were combined, however, there was a strong independent association between frequency of high-sugar food intake and RCE. It has been reported that children who consume more high -sugar beverages are more at risk for dental caries7,34. Sucrose helps stimulate cariogenic bacteria to colonize and adhere permanently to tooth surfaces via α-1-3-rich, water insoluble glucans, allowing acids formed by cariogenic foods easier access to tooth enamel11.
Historical literature on Alaska Natives suggests that the prevalence of caries in the early 1900s was low. For example, little evidence of caries was found in teeth from preserved Alaska Native human remains housed at the Smithsonian Institution14. Prevalence studies in the 1930s among Alaska Native adults and children also reported low rates of caries15-17. The historical data makes for an interesting comparison with today's high prevalence of poor dental health. One possible explanation for the discrepancies in oral health over time lies in the Arctic nutrition transition, where a high percentage of the diet is made up of carbohydrates, particularly in the form of high-sugar foods. It is possible that the historically high traditional food intake combined with low intake of high-sugar foods protected Inuit against dental caries in the past.
Prior to 1973, the residents of Nunavut did not have in-community access to dental care38. From the 2006 Aboriginal Children's Survey, only 57% of Inuit children under 6 years in Nunavut had received dental care in the past 12 months39. Nunavut has experienced one of the largest increases in dental expenditures in comparison with other regions of Canada, and in 2003 the Government of Canada reported that various oral health programs were in place that involved dietary counselling, oral health promotion, education at the prenatal level as well as in the classroom and well-baby clinics, oral hygiene instruction and the use of chemical sealants40-42. Preventative measures are particularly important because the isolation of the communities remains a deterrent to good dental care, including shipping of equipment and supplies and retaining qualified Inuit and non-Inuit dental professionals43. There is evidence that water fluoridation and fluoride varnishes may reduce the prevalence of ECC among Canadian Aboriginal preschoolers7,25. The identification of lower milk intake and higher intake of sugary foods as risk factors for RCE adds to evidence that dietary counselling is also an important component of prevention. Given the success in smoking education messages, where now the majority of homes with preschoolers have restrictions against smoking in the home, this provides some evidence that public health education can have a meaningful impact on health behaviours26 and that the same success could be achievable for oral health with renewed dental health education efforts developed and shaped by communities.
Limitations
The present study was limited by its cross-sectional design and reliance on parent/caregiver-reported oral health rather than clinically assessed oral health. While the question 'Does your child have any decayed, extracted, or filled primary teeth?' could also potentially include teeth which naturally exfoliated or teeth lost due to trauma, the parental reports cannot be validated with the current data. The strength of the study involves the use of both an FFQ and 24 hour recall to assess dietary habits. However, multiple 24 hour recalls would have strengthened the study by better characterizing usual intake. This was not conducted due to concerns of research burden.
Conclusions
Evidence of a high prevalence of reported caries experience was found among Inuit preschoolers in Nunavut. This helps to confirm the suspected severity of this health problem among young Inuit children. Evidence is reported that low milk and high-sugar food and beverage intake are independently associated with having RCE. This supports the need for continuation of established initiatives in Nunavut that aim to reduce the consumption of soda pop and sugar-sweetened beverages. Inuit-specific information can provide relevant data for communities and public health campaigns aimed at reducing the burden of early childhood caries.
Acknowledgements
Funding for this study was provided through the Government of Canada International Polar Year, Canadian Institutes for Health Research, Health Canada, and Indian and Northern Affairs Canada. The authors acknowledge the work of the field research team, including Christine Ekidlak, Laureen Angalik, Kathy Morgan and Nancy Faraj. The authors thank Dr Nelofar Sheikh and Louise Johnson-Down, and Zhirong Cao for their assistance in data management and dietary data analyses, and survey weighted analyses. Finally, the authors are grateful to the Nunavut Inuit Health Survey steering committee for their assistance with this project, and their guidance and feedback.
References
1. American Academy of Pediatric Dentistry. Definition of Early Childhood Caries. (Online) 2008. Availabble: http://www.aapd.org/media/policies.asp (Accessed 20 March 2010).
2. Albert R, Cantin R, Cross H, Castaldi C. Nursing caries and the Inuit children of the Keewatin. Journal of the Canadian Dental Association 1988; 54(10): 751-758.
3. Houde G, Gagnon P, St-Germain M. A descriptive study of early caries and oral health habits of Inuit pre-schoolers: preliminary results. Arctic Medical Research 1991; (Suppl): 683-684.
4. Zammit M, Torres A, Johnsen D, Hans M. The prevalence and patterns of dental caries in Labrador Inuit Youth. Journal of Public Health Dentistry 1994; 54(3): 132-138.
5. Young T, Moffatt M, O'Neil JD, Thika R, Mirdad S. The population survey as a tool for assessing family health in the Keewatin region, NWT, Canada. Arctic Medical Research 1995; 54(Suppl1): 77-85.
6. Weinstein P, Smith W, Fraser-Lee N, Shimono T, Tsubouchi J. Epidemiologic study of 19-month-old Edmonton, Alberta children: caries rates and risk factors. ASDC Journal of Dentistry for Children 1996; 63(6): 426-433.
7. Leake J, Jozzy S, Uswak G. Severe dental caries, impacts and determinants among children 2-6 years of age in Inuvik Region, Northwest Territories, Canada. Journal of the Canadian Dental Association 2008; 74(6): 519.
8. Schroth R, Harrison R, Lawrence H, Peressini S. Oral health and the Aboriginal child: a forum for community members , researchers and policy-makers. Journal of the Canadian Dental Association 2008; 74(5): 429-432.
9. Lawrence HP, Binguis D, Douglas J, McKeown L, Switzer B, Figueiredo R et al. Oral health inequalities between young Aboriginal and non-Aboriginal children living in Ontario, Canada. Community Dentistry and Oral Epidemiology 2009; 37: 495-508.
10. Schroth R, Harrison R, Moffatt M. Oral health of Indigenous children and the influence of early childhood caries on childhood health and well-being. Pediatric Clinics of North America 2009; 56(6): 1481-1499.
11. Seow W. Biological mechanisms of early childhood caries. Community Dentistry and Oral Epidemiology 1998; 26(Suppl1): 8-27.
12. Reisine S, Douglass J. Psychosocial and behavioral issues in early childhood caries. Community Dentistry and Oral Epidemiology 1998; 26(Suppl1): 32-44.
13. Moynihan P, Peterson P. Diet, nutrition and the prevention of dental diseases. Public Health Nutrition 2004; 7(1A): 201-226.
14. Leigh R. Dental pathologies of the Eskimo. Dental Cosmos 1925; September: 884-889.
15. Rosebury T, Karshan M. Dental Caries among Eskimos of the Kuskokwim Area of Alaska. III. A dietary study of three Eskimo settlements. American Journal of Diseases in Children 1939; 57(6): 1343-1362.
16. Rosebury T, Waugh L. Dental caries among Eskimos of the Kuskokwim Area of Alaska. I. Clinical and bacteriological findings. American Journal of Diseases in Children 1939; 57(4): 871-893.
17. Siegel E, Waugh L, Karshan M. Dietary and metabolic studies of Eskimo children with and without dental caries. American Journal of Diseases in Children 1940; 59(1): 19-38.
18. Kuhnlein H, Receveur O, Soueida R, Egeland G. Arctic Indigenous Peoples experience the nutrition transition with changing dietary patterns and obesity. Journal of Nutrition 2004; 143: 1447-1453.
19. Kuhnlein H, Soueida R, Receveur O. Dietary nutrient profiles of Canadian Baffin Island Inuit differ by food source, season, and age. Journal of the American Dietetic Association 1996; 96(2): 155-162.
20. Moffat M, Young T. Nutritional patterns if Inuit in the Keewatin region of Canada. Arctic Medical Research 1994; 53: 298-300.
21. Verdier PC, Eaton RD. A study of the nutritional status of an Inuit population in Canadian High Arctic. Part 2: some dietary sources of vitamins A and C. Canadian Journal of Public Health 1987; 78(July/August): 236-239.
22. Kuhnlein H, Receveur O. Local cultural animal food contributes high levels of nutrients for Arctic Canadian Indigenous adults and children. Journal of Nutrition 2007; 137(4): 1110-1114.
23. Receveur O, Boulay M, Kuhnlein H. Decreasing traditional food use affects diet quality for adult Dene/Métis in 16 communities of the Canadian Northwest Territories. Journal of Nutrition 1997; 127: 2179-2186.
24. Tiberia M, Milnes A, Feigal R, Morley K, Richardson D, Croft W et al. Risk factors for early childhood caries in Canadian preschool children seeking care. Pediatric Dentistry 2007; 29(3): 201-208.
25. Lawrence H, Binguis D, Douglas J, McKeown L, Switzer B, Figueiredo R et al. A 2-year community randomized controlled trial of fluoride varnish to prevent early childhood caries in Aboriginal children. Community Dental and Oral Epidemiology 2008; 36: 503-516.
26. Egeland G, Faraj N, Osborne G. Cultural, Socioeconomic, and Health Indicators among Inuit Preschoolers: Nunavut Inuit Child Health Survey, 2007-2008. Rural and Remote Health 10: 1368. (Online) 2010. Available; www.rrh.org.au (Accessed 18 June 2010).
27. Schroth R, Moffatt M. Determinants of Early Childhood Caries (ECC) in a rural Manitoba community: a pilot study. Pediatric Dentistry 2005; 27(2): 114-120.
28. Indian Health Service. The 1999 Oral Health Survey of American Indian and Alaska Native Dental Patients: findings, regional differences and national comparisons. (Online) 2002. Washington, DC: Dept of Health and Human Services, Indian Health Service. Available: www.dental.ihs.gov/downloads/Oral_Health_1999_IHS_Survey.pdf (Accessed 18 April 2010).
29. Peressini S, Leake J, Mayhall J, Maar M, Trudeau R. Prevalence of early childhood caries among First Nations children, District of Manitoulin, Ontario. International Journal of Paediatric Dentistry 2004; 14(2): 101-110.
30. Schroth R, Moore P, Brothwell D. Prevalence of early childhood caries in 4 Manitoba communities. Journal of the Canadian Dental Association 2005; 71(8): 567.
31. Beltrán-Aguilar E, Barker L, Canto M, Dye B, Gooch B, Griffin S et al. Surveillance for dental caries, dental sealants, tooth retention, edentulism, and enamel fluorosis - United States, 1988-1994 and 1999-2002. Morbidity and Mortality Weekly Report Surveillance Summaries 2005; 54(3): 1-43.
32. Roberts C, Warren J, Weber-Gasparoni K. Relationships between caregivers' responses to oral health screening questions and early childhood caries. Journal of Public Health Dentistry 2009; 69(4): 290-293.
33. Schroth R, Brothwell D, Moffatt M. Caregiver knowledge and attitudes of preschool oral health and early childhood caries (ECC). International Journal of Circumpolar Health 2007; 66(2): 153-168.
34. Marshall T, Levy S, Broffitt B, Warren J, Eichenberger-Gilmore J, Burns T et al. Dental caries and beverage consumption in young children. Pediatrics 2003; 112(3,pt1): e184-e191.
35. Yoshihara A, Watanabe R, Hanada N, Miyazaki H. A longitudinal study of the relationship between diet intake and dental caries and periodontal disease in elderly Japanese subjects. Gerodontology 2009; 26(2): 130-136.
36. Zhang X, Rahemtulla F, Zhang P, Beck P, Thomas H. Different enamel and dentin mineralization observed in VDR deficient mouse model. Archives of Oral Biology 2009; 54(4): 299-305.
37. Ferrazzano G, Catile T, Quarto M, Ingenito A, Chianese L, Addeo F. Protective effect of yogurt extract on dental enamel demineralization in vitro. Australian Dental Journal 2008; 53(4): 314-319.
38. Bedford W, Davey K. Indian and Inuit dental care in Canada: the past, the present, and the future. Journal of the Canadian Dental Association 1993; 59: 126-132.
39. Tait H. Aboriginal Peoples Survey, 2006: Inuit health and social conditions. (Online) 2008. Available: http://www.statcan.gc.ca/bsolc/olc-cel/olc-cel?catno=89-637-XWE2008001&lang=eng (Accessed 14 October 2009).
40. Government of Canada, Parliamentary Standing Committee on Health. First Nations and Inuit Dental Health. In: B Brown (Ed.). Fifth report of the standing committee on health. (Online) 2003. Available: http://www2.parl.gc.ca/CommitteeBusiness/ReportsResponses.aspx?Cmte=HEAL&Language=E&Mode=1&Parl=37&Ses=2 (Accessed 15 October 2009).
41. First Nations and Inuit Health Branch Non-Insured Health Benefits Directorate Program. Non-Insured Health Benefits Program Annual Report 2003/2004. Ottawa, Ontario: Program Analyses Division, First Nations and Inuit Health Branch Non-Insured Health Benefits Directorate, 2005; 1-106.
42. Health Canada First Nations and Inuit Health Branch. Government response to the 5th report of the Standing Committee on Health: First Nations and Inuit Dental Health. (Online) 2003. Available: http://www.hc-sc.gc.ca/fniah-spnia/pubs/nihb-ssna/_dent/2003-gvt-rps/index-eng.php (Accessed 15 October 2009).
43. Quiñonez C. A political economy of oral health services in Nunavut. In: Proceedings, 12th International Congress on Circumpolar Health; 10-14 September 2003; Nuuk, Greenland. International Journal of Circumpolar Health 2004; 63(Suppl2): 324-329.


