Introduction
Gestational diabetes mellitus (GDM) is defined as carbohydrate intolerance first occurring during pregnancy1. For most women, diabetes is temporary; however, an episode of GDM confers an approximately seven-fold increased risk of developing type 2 diabetes mellitus (T2DM)2.
Two major diabetes prevention studies demonstrated that lifestyle interventions, which promote healthy eating, increase physical activity and modest weight loss of 5-7%, can delay or prevent the onset of T2DM in individuals with impaired glucose tolerance3,4. Given the increasing prevalence of diabetes5, there is an urgent need to identify and target high risk populations and develop affordable, effective risk reduction and diabetes prevention strategies.
Women with previous GDM are an at-risk group likely to benefit from diabetes prevention interventions. Improving women's awareness and knowledge of the benefits of risk reduction activities may provide the opportunity and incentive to undertake lifestyle measures to reduce their risk for diabetes. However, mothers with young children face many barriers to engaging in healthy lifestyle programs, including time constraints, multiple commitments, tiredness and resuming work6,7.
Several recent studies reported the nature and prevalence of postpartum diabetes risk behaviours in women with previous GDM but few studies included rural women. A retrospective study of health behaviour in 121 Danish women with previous GDM showed participants with a weight above the healthy weight range (BMI >25 kg/m2) were more likely to gain than lose weight (33% vs 18%; p<0.05)8. Most expressed some degree of concern about developing diabetes, but there was no significant association between concern and weight loss or activity level either before or after pregnancy.
In a study of 226 Australian women with previous GDM, 33% met activity guidelines of at least 150 min of activity per week, and 51% were overweight or obese6. Most frequently cited barriers to activity were lack of child care and time constraints. Women reported feeling least confident to be active when tired or encountering time constraints. Self-efficacy and social support were positively correlated with physical activity. A study of 53 rural women with previous GDM in the Goulburn Murray area of northern Victoria demonstrated 44% meeting activity guidelines with 66% overweight or obese9. The majority knew how to prevent T2DM but only a small number were undertaking lifestyle modifications to reduce their diabetes risk9. Together, these studies suggest a large proportion of women with previous GDM do not engage in diabetes prevention behaviour despite being aware of the risks.
The current study aimed to examine the prevalence of and readiness to adopt diabetes risk reduction behaviours in rural postpartum women with previous GDM. The Stages of Change (SOC) theory, widely used to examine the lifestyle behaviours relevant to diabetes prevention10,11,was used as the conceptual framework12.
Methods
Goulburn Valley Health (GVH) is one of 5 regional referral hospitals in Victoria and is the major regional referral site for specialist care of women with GDM. The region encompasses a large rural centre (population 25 000-99 000, Rural, Remote & Metropolitan Areas [RRMA] level 3) and smaller towns (population <10 000, RRMA levels 3-5).
The study population was identified from a GVH medical record database and included all women diagnosed with GDM between July 2001 and December 2005 (n = 210). Participants were eligible if they had GDM in the previous 5 years, were over 18 years of age, not currently pregnant, did not have T2DM and were able to speak English. Two hundred and ten women were mailed a questionnaire and stamped return-addressed envelope followed by a reminder letter 10 days later. Three weeks after the second mail-out telephone contact was made with those participants who had not responded, and the questionnaire was resent to interested participants.
The survey instrument contained questions about demographic details, SOC for physical activity, dietary fat reduction and weight loss, physical activity level, and dietary fat intake. Physical activity was measured using a modified version of the Active Australia Questionnaire (AAQ), which is reliable and valid when compared with other widely used measures of physical activity13. Respondents documented the number of times and the total time they engaged in walking, moderate or vigorous physical activity during the previous 7 days.
A food frequency questionnaire (FFQ) was used to collect dietary intake data. The FFQ, a brief food behaviour tool that measures self-reported fat and fibre intake behaviour, has been shown to be both reliable and valid14. The FFQ contains 20 questions such as 'How many days a week do you eat fried food with a batter or breadcrumb coating?'. An additional question was added relating to the frequency of consuming high fat snacks, making a total of 21 questions, of which 14 related to fat (fat score). Weight and height were self-reported.
The SOC tool is used to measure SOC for dietary fat intake15,16 and weight loss17. Participants selected the response that best represented their perspective of the behaviour in question from a series of 6 options. The SOC tool was also used for activity, substituting the words being physically active for losing weight or reducing fat respectively. The SOC was coded from 1 ('I haven't given the matter of physical activity/weight loss/dietary fat any thought at all'; pre-contemplation) to 5 ('I have been physically active/losing weight/reducing dietary fat for more than six months'; maintenance).
Total minutes spent being active each week for each participant was calculated as the sum of the minutes spent walking plus the minutes spent in moderately intense activity plus a factor of twice the minutes spent in vigorous activity18. Sufficient physical activity was categorised as meeting activity guidelines (150 min per week over ≥5 sessions), and BMI was calculated from reported weight/height2. The FFQ scores were reported as total fat score (FS; maximum score = 70); higher score representing more fat-reducing behaviours.
Data were coded and analysed using the Statistical Package for the Social Sciences v14.0 (SPSS; Chicago, IL, USA; www.spss.com). Descriptive statistics were used to describe study participants and variables. Variables were categorised as number of children (1 child, ≥2 children), income (≤$47,000, >$47,000), employment (work and/or study, home duties), rurality (RRMA 3 or RRMA 4 & 5) and BMI (<25 or ≥25 kg/m2). Categorical data were reported as a proportion, with continuous variables described using mean and standard deviation or median and interquartile range (IQR; 25th, 75th percentile) according to their distribution. Chi-square test or Fisher's exact tests were used to analyse categorical variables. A Kruskal Wallis test compared the continuous variables (BMI, activity and fat score) to SOC categories. A p-value below 0.05 was considered significant (two-tailed tests). Approval to conduct the study was obtained from the GVH and University of Melbourne Human Review Ethics Committees.
Results
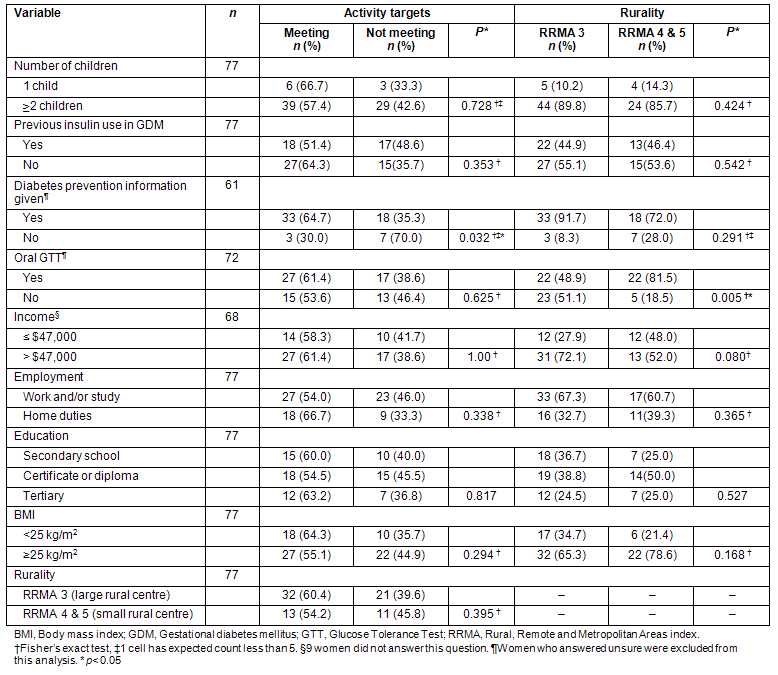
Eighty-four women consented to participate, seven were excluded; four had T2DM and three were pregnant, giving a final sample of 77 women. The socio-demographic characteristics of the participants are given (Table 1).
Women reported being active for an average of 315.4 ± 297.2 min (range 0-1500 per week). Fifty-eight percent achieved recommended activity targets for health benefits. Mean FS was 46.1 ± 7.73 and the mean BMI for the total sample was 29.6 kg/m2 (± 7.30); 63.6% of women above the healthy weight range (BMI ≥25 kg/m2). Of the women with a BMI ≥25 kg/m2, 22.1 % were overweight (BMI 25-29 kg/m2) and 41.5% were obese (BMI ≥ 30 kg/m2). Sixty-one percent of women had a postpartum oral glucose tolerance test (OGTT), and 45% required insulin during their last episode of GDM.
Women were classified into one of 5 motivational stages for the three target behaviours: activity, weight loss and dietary fat reduction (Table 2). Due to the small number of women in the pre-contemplation stage for all three behaviours, the five stages were collapsed into four by combining those in pre-contemplation and contemplation. Most were in preparation stages for activity and weight loss; whereas, for reducing dietary fat, the largest proportion was in the maintenance stage.
There was a statistically significant difference in self-reported activity level, BMI, and FS over the four behaviour change stages (Table 2). The median value for physical activity and fat score increased steadily from pre-contemplation/contemplation to maintenance stage. Participants in maintenance recorded the highest level of physical activity. There was a statistically significant difference in BMI between women in the preparation stage compared with those in the pre-contemplation and action stages. The highest median BMI was recorded at the preparation stage and the lowest at pre-contemplation and action stages.
There was no significant association between any of the variables measured and the proportion of women meeting activity guidelines (Table 3) with the exception of women who remembered receiving diabetes prevention information and meeting activity targets (p<0.05). Additionally, there was no significant association between the rurality index (Table 3) and any of the variables measured. However, women living in a large rural area (RRMA 3) were significantly less likely to have a post-partum OGTT than women who lived in small rural areas (RRMA 4 & 5) (p<0.005).
Table 1: Socio-demographic characteristics of participants
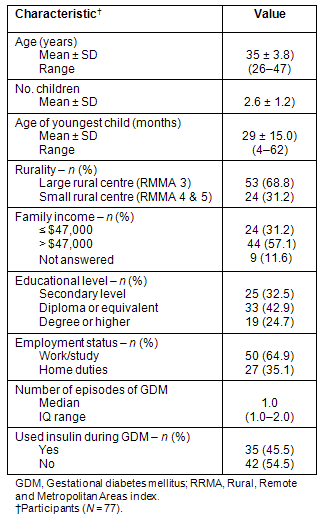
Table 2: Proportion of women at each stage of change (SOC) and relationship between SOC for activity, weight loss and reducing dietary fat
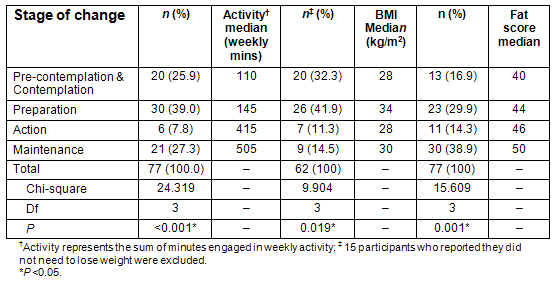
Table 3: Relationship between variables, meeting recommended activity targets and rurality index (RRMA)
Discussion
The study findings demonstrate that, although reporting a high level of readiness to change behaviour, a large proportion of rural postpartum women are overweight or obese despite being active. Fifty-eight percent of women reported undertaking sufficient physical activity for health benefits compared with 33.6% in a metropolitan population of women with GDM6, and 46% of women aged 18-23 years (with children) reported in the Australian Longitudinal Study of Women's Health19. It is possible that women with young children in rural areas find it easier to meet activity targets than adopting other diabetes risk reduction behaviours. In addition, the study reported a high proportion of women actively reducing their fat intake, a finding that was not reflected in the women's weight status.
The high proportion of obese women (44.4%) in the present study is a concern in terms of women's future health outcomes and their risk of developing T2DM. The total prevalence of overweight and obesity in the current study was higher than a metropolitan sample of women with previous GDM; overweight (63.1% vs 51.3%) or obese (41.5% vs 29.1%)6 but similar to that identified in a more general female population where 67.6% of women were overweight (40.8%) and obese (26.8%)20. The apparent disconnection in rural women between being more active but conversely more obese needs further exploration. This study demonstrates that being aware of the risk, being ready for change, being active and taking steps to reduce fat intake, may not lead to reduction of obesity, because most women failed to loose weight despite engaging in these behaviours. The significance of this association for weight loss programs for postpartum women is that obesity may be also associated with other factors, such as emotional wellbeing, as much as diet or lack of activity.
While not explored in this study, women are believed to be motivated to change by awareness of health risk in addition to dissatisfaction with body shape and size. Previous research indicates that women with previous GDM may not undertake lifestyle change to reduce their risk of developing diabetes despite understanding the risk8. Generally concern about future health does not translate into preventive health behaviours21. In the case of women with young children, immediate family priorities may override longer term health goals, especially when women feel well. This is an area for future research, investigating optimal methods, the timing of delivery of health promotion information and the requirements for ongoing support in this high risk population.
It was encouraging to find that women who remembered receiving diabetes prevention information in the postpartum period were significantly more likely to meet activity guidelines. It is, however, possible that women already aware of the association between GDM and diabetes risk may remember being given information. In the current study there was no baseline association between receiving diabetes prevention information and being a healthy weight, which was consistent with the authors' observation that achieving a healthy weight is more difficult than achieving regular activity for women with previous GDM.
The findings support a predictive association between SOC and behaviour. The level of activity and fat score was highest for participants in action and maintenance stages. Excluding women in the healthy weight range, average BMI was highest for participants in the preparation stage for weight loss. The motivational SOC may be able to predict extent of diabetes risk reduction behaviours at a given time in this sample of rural women with a history of GDM. However, intent to change behaviour does not necessarily translate to consistent action over time and it is unclear whether reporting readiness to change predicts actual behaviour change.
Overall, 61% of women had a postpartum OGTT. Opportunities for early diagnosis and reducing insulin resistance are being missed by one-third of women in the current cohort. Current recommendations suggest all women who have GDM have an OGTT 6 weeks postpartum to determine whether glucose tolerance has returned to normal, repeated at regular intervals of 1-3 years, depending on the individual's likelihood of becoming pregnant and other diabetes risk factors1,22.
Unexpectedly, a higher proportion of women living in a small rural area reported having a postpartum OGTT, compared with women living in large rural areas (82.5% vs 48.9%, p< 0.05). Despite having to travel longer distances to access services23 women in small rural areas may consult the same doctor who is also likely to treat all family members and may, therefore, be more aware of family diabetes risk and the importance of screening. Research into this apparently paradoxical finding could provide useful information about service development in rural health services.
A number of limitations of the study need to be acknowledged. It is possible that women who agreed to participate in the study were already motivated to reduce their diabetes risk. The low participant response rate suggests the findings may not be representative of the larger population of rural women with GDM, and the study may be underpowered to detect significant associations among variables because of the small sample size. The significant association between receiving diabetes prevention information and activity should be interpreted with caution due to the small number in one of the groups.
Self-reported behaviours may not represent actual behaviours. Women with young children may perceive that the intensity of their activity to be higher than it actually is, given that in the course of caring for young children it could seem they are 'busy all day'. Finally, subjects in the study had previously attended the Diabetes Centre and knew the researcher, which may have influenced self-reported information.
Conclusion
Our study shows there is a gap between readiness to adopt diabetes risk reduction behaviours and healthy weight status. The postpartum period is an important period for a diabetes screening test; the findings suggest it is also an important period to promote lifestyle behaviour change. While the majority of women report being 'ready' to change behaviour, further studies are needed to determine whether intent can be translated to action and what factors assist the process. Lifestyle change is difficult and requires ongoing support to be sustained. Women with previous GDM are not easy to engage in health promotion and the challenge remains to design diabetes prevention interventions that do not add to the already busy life of a mother with young children.
Acknowledgements
This research was made possible by the assistance of the GVH Diabetes Centre Team and the willing participants. Wendy Swan received funding and support through the Commonwealth PHCRED Program
References
1. Hoffman L, Nolan C, Wilson JD, Oats JJN, Simmons, D. Gestational diabetes mellitus-management guidelines. The Australasian diabetes in pregnancy society. Medical Journal of Australia 1998; 169: 93-97.
2. Bellamy L, Casas J-P, Hingorani AD, Williams DR. Type 2 diabetes mellitus after gestational diabetes: a systematic review and meta-analysis. The Lancet 2009; 373: 1773-1779.
3. Diabetes Prevention Program. The Diabetes Prevention Program (DPP). Description of lifestyle intervention. Diabetes Care 2002; 25: 2165-2171.
4. Tuomilehto J, Lindström J, Eriksson J, Valle T, Hämäläinen H, Ilanne-Parikka P et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. New England Journal of Medicine 2001; 344: 1343-1350.
5. Cheung NW, Byth K. Population health significance of gestational diabetes. Diabetes Care 2003; 26(7): 2005-2009.
6. Smith BJ, Zehle K, Cheung NW, McLean M, Bauman AE. Postpartum physical activity and related psychosocial factors among women with recent gestational diabetes mellitus. Diabetes Care 2005; 28: 2650-2654.
7. Symons Downs D, Ulbrecht JS. Understanding exercise beliefs and behaviors in women with gestational diabetes mellitus. Diabetes Care 2006; 29(2): 236-240.
8. Stage E, Ronneby H, Damn P. Lifestyle change after gestational diabetes. Diabetes Research and Clinical Practice 2004; 63: 67-72.
9. Swan W, Kilmartin G, Liaw ST. Assessment of readiness to prevent type 2 diabetes in a population of rural women with a history of gestational diabetes. Rural Remote Health 2007 7(4): 802. (Online) 2007. Available: www.rrh.org.au (Accessed 14 October 2010).
10. Glasgow RE, Bull SS, Piette JD, Steiner JF. Interactive Behaviour change technology. A partial solution to the competing demands of primary care. American Journal of Preventative Medicine 2004; 27(2S): 80-87.
11. Clark M, Hampson SE, Avery L, Simpson R. Effects of a tailored lifestyle self-management intervention in patients with type 2 diabetes. British Journal of Health Psychology 2004; 9: 365-379.
12. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. Journal of Consulting and Clinical Psychology 1983; 51(3): 390-395.
13. Timperio A, Salmon J, Crawford D. Validity and reliability of a physical activity recall instrument among overweight and non-overweight men and women. Journal of Science and Medicine in Sport 2003; 6(4): 477-491.
14. Wright JL, Scott JA. The Fat and Fibre Barometer, a short food behaviour questionnaire: reliability, relative validity and utility. Australian Journal of Nutrition and Dietetics 2000; 57(1): 33-39.
15. Lamb R, Joshi MS. The stage model and processes of change in dietary fat reduction. Journal of Human Nutrition and Dietetics 1996; 9(1): 43-53.
16. Lamb R, Joshi MS. Active but not consistent: dietary behaviour and the stages of change model. Psychology and Health 2004; 19(5): 543-559.
17. Macqueen CE, Brynes AE, Frost GS. Treating obesity: can the stages of change model help predict outcome measures? Journal of Human Nutrition and Dietetics 1999; 12: 229-236.
18. Armstrong T, Bauman A, Davies J. Physical activity patterns of Australian adults. Results of the 1999 national physical activity survey. Canberra, ACT: AIHW, 2000; 11.
19. Brown WJ, Mishra G, Lee C, Bauman A. Leisure time physical activity in australian women: relationship with well being and symptoms. Research Quarterly for Exercise and Sport 2000; 71(3): 206-216.
20. Barr E, Magliano D, Zimmet P, Polkinghorne K, Atkins RC, Dunstan DW et al. AusDiab 2005. The Australian Diabetes, Obesity and Lifestyle Study: Tracking the Accelerating Epidemic: Its causes and outcomes. Melbourne, VIC: International Diabetes Institute, 2006.
21. Feig DS, Chen E, Naylor CD. Self-perceived health status of women three to five years after the diagnosis of gestational diabetes: a survey of cases and matched controls. American Journal of Obstetrics and Gynecology 1998; 178: 386-393.
22. Oats JJ, McIntyre HD. Revision of guidelines for the management of gestational diabetes mellitus, Letter to the Editor. Medical Journal of Australia 2004; 181(6): 342.
23. Bourke L, Sheridan C, Russell U, Jones G, DeWitt D, Liaw ST et al. Developing a conceptual understanding of rural health practice. Australian Journal of Rural Health 2004; 12: 181-186.
Abstract
Introduction: For most women, gestational diabetes is temporary; however, an episode of gestational diabetes mellitus (GDM) confers an approximately seven-fold increased risk of developing type 2 diabetes mellitus. Objective: To examine readiness to adopt diabetes risk reduction behaviours and the prevalence of these behaviours among rural women with GDM during their last pregnancy.
Methods: The study design was a self-administered mailed questionnaire seeking information about demographics, stage of change, physical activity level and dietary fat intake. Setting: Regional outpatient context. Participants: Women with a single episode of GDM between 1 July 2001 and 31 December 2005 (n = 210). Main outcome measures: Stage of change for physical activity, weight loss and reducing dietary fat behaviour; meeting activity targets, body mass index (BMI) and dietary fat score.
Results: Eighty-four women returned completed questionnaires (40% response rate). Of the 77 women eligible (mean age 35 ± 3.8 years), 58% met recommended activity targets. Sixty-three percent of women were overweight or obese: mean BMI 29.6 kg/m2 (± 7.30). Women reported a high level of preparedness to engage in physical activity, weight loss and reduction of fat intake. Thirty-nine percent of women had not had any postpartum follow-up glucose screening. Women who remembered receiving diabetes prevention information were significantly more likely to meet physical activity targets (p<0.05).
Conclusions: Readiness to engage in behaviour change was high among this group of rural women for all three diabetes risk reduction behaviours measured. However, despite a high proportion of women meeting activity targets and reducing fat intake, the majority of women remained overweight or obese. Postpartum follow-up glucose testing needs to be improved and the impact of diabetes prevention information provided during pregnancy warrants further study.
Key words: diabetes prevention, obesity, physical activity, post partum, stage of change.
You might also be interested in:
2010 - Working with refugees - a manual for caseworkers and volunteers
2007 - Core drivers of quality: a remote health example from Australia




