Sri Lanka is a middle income country with a well-established public health infrastructure developed more than four decades ago. Of the 20.6 million population in this island nation, 80% live in rural areas1. A well organized maternal and child health (MCH) program has operated since 1968, with significant gains in reducing both maternal and infant mortality rates2. The Family Health Bureau functioning under the Sri Lanka Ministry of Health is the focal point for this program which consists of maternal health, child health, women's health and family planning. Antenatal care is one of the key elements of the MCH component, provided through the streamlined infrastructure of the Ministry of Health and provincial services which comprise a wide network of medical institutions and Medical Officer of Health (MOH) areas3. Pregnant women are eligible to receive antenatal care free of charge in the public health system, irrespective of their socioeconomic status and geographic location, and have a minimum of five contacts with the primary health team during pregnancy. This service model achieves a 98% antenatal care coverage2.
Oral health care has recently been incorporated into the existing national MCH program, with practice guidelines aiming to promote the oral health status of mothers and their children by providing comprehensive care during the prenatal and antenatal periods4. The specific objectives of this program are to:
- reduce the incidence of complications of poor oral health during pregnancy
- control early childhood caries (ECC) by reducing the risk of transmission of cariogenic bacteria from mother to baby
- reduce the incidence of adverse pregnancy outcomes attributed to poor oral health
- educate pregnant women on preventive measures for dental caries in young children.
All pregnant women who visit antenatal clinics are referred to the nearest public dental clinic for oral health screening and necessary care. Oral health education sessions are also delivered to pregnant women with health education programs on lactation management, nutrition and other MCH health topics using the antenatal clinics as health promotion settings. Oral health education sessions provide a unique opportunity to make the women aware of the importance good oral health during pregnancy and to motivate them to attend for an oral screening and have their oral health problems addressed. This important initiative was promulgated by evidence of a high burden of oral disease among Sri Lankan antenatal women5,6.
Pregnancy is an important milestone in the life-course of a female with the dual factors of pregnancy affecting oral health and oral health affecting the pregnancy outcome. Pregnant women are vulnerable to common oral diseases such as periodontal disease and dental caries; if oral health is not well maintained during this period there are implications for oral health in the woman's subsequent life7,8. Changes in dietary habits and oral hygiene practices can increase the risk of tooth decay during pregnancy8. For example, a study conducted in Iraq showed significant difference in decayed teeth among pregnant and single females9. The increased production of oestrogen and progesterone during pregnancy may give rise to the development of gingivitis and periodontitis attributed to increased vascular permeability and possible tissue oedema10,11. Evidence shows that periodontal disease is associated with gestational diabetes mellitus (GDM) which contributes to maternal and infant morbidity as well as the risk of the mother developing type 2 diabetes mellitus subsequently12,13. Moreover, maternal periodontal disease may increase the risk of adverse pregnancy outcomes such as pre-eclampsia, pre-term births, retarded fetal growth and low birth weight14,15.
A plausible mechanism suggested for such associations is chronic periodontal infections that serve as reservoirs for Gram-negative anaerobic bacteria, their toxins as well as inflammatory mediators such as PGE2 and TNF-α, C-reactive proteins (CRP) posing potential threats to the foetal-placental unit. Sustained elevated levels of CRP and interleukins can act as insulin antagonists, interfere with insulin signalling and causing glucose intolerance and GDM16.
Nevertheless, there have been very limited investigations into oral health status and pregnancy outcomes. A prospective follow-up study conducted in a rural central mountainous locale of Sri Lanka suggested a possible association between maternal periodontal disease and pre-term low birth weight6. Studies have shown that the transmission of cariogenic bacteria from mother to infant is increased when the mother has poor oral health with untreated dental caries17. Hence, active dental caries among antenatal women has been identified as one of the most important predictors for caries among toddlers18.
Therefore, improving the oral health status of pregnant women could prevent the complications of dental diseases during pregnancy, may reduce adverse pregnancy outcomes, the maternal and infant morbidity associated with GDM and has the potential to reduce the incidence of ECC, which is a public health problem in many countries, including Sri Lanka. For example 65.5% of 5 year-old children suffer from tooth decay19. In addition, a recent study reported 32.2% ECC prevalence among 1 to 2 year olds in Sri Lanka, of which 95% was unattended20. Such issues underpin the program objectives of the national oral health program for pregnant women.
Against this backdrop, the purpose of this article was to report the oral health status of antenatal women based on the findings of two recent studies conducted among rural and urban antenatal women in the Western Province of Sri Lanka. Also highlighted is the need for providing oral health care to this group under the recently introduced national program by making services more accessible and acceptable to pregnant women.
Data for the present study were obtained from two consecutive studies. The first included 459 pregnant women in their second trimester (between the 13th and 28th weeks of their period of amenorrhoea [POA]) from a rural setting of the Western Province, Sri Lanka. Second study included 348 pregnant women in their third trimester (29th to 37th weeks POA) from an urban setting in the same province.
In Sri Lanka the urban-rural definition is based exclusively on administrative boundaries. 'Urban areas' are defined as those areas consisting of municipal councils; whereas, for 'rural areas' the 'Pradeshiya Sabha' is the administrative unit21. This definition was used in the last Population and Housing Census conducted in 200121; however, it has the inherent limitation of underestimating urbanization, which does not reflect the actual situation, and this highlights the need to compile a new statistical urban definition for Sri Lanka based on multivariate analysis of the variables related to urbanization22.
The rural setting was located in the 'Divulapitiya MOH area' situated in the Gampaha District of the Western Province, which covers an area of 193 km2 with an estimated mid-year population of 143 215 (as reported by the MOH Office) and a population density of 782 per km2 . The majority ethnic group is Sinhalese and agriculture is the main source of income. The urban setting was located in the 'Pilyandala MOH area' with an estimated mid-year population of 162 255 (as reported by the MOH Office). The total land area is 47 km2 with a population density of 3350 per km2, over four-fold higher than that of the rural setting, a characteristic feature of the urban-rural population distribution in Sri Lanka.
For both studies, sample size calculations were based on a formula method of estimating a population proportion with specified absolute precision23, as well as adjustment for non-response. Sample size calculation for the rural study was as follows:
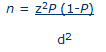
n = Sample size
z = Standard normal deviate of 1.96 for a confidence level set at 95%
P= anticipated population proportion for dental caries/ periodontal disease
d = absolute precision required on either side of the proportion.
Prevalence of dental caries in pregnant women considered to be 65%, based on a study conducted in Mexico24 which was used for the calculation. Therefore, for the rural study sample size determination was as follows:
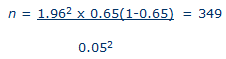
With the addition of 10% non-response rate, the minimum sample size would be 384. However, the final sample consisted of 459 women for practical reasons.
For the urban study the caries prevalence rate of 92% from the rural study was used for the sample size calculation as follows:
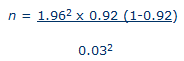
n = 314.15
To compensate 10% non respondents,
Sample size = 314 x 110%
= 345.4
Hence, the minimum sample size was 346 but the final sample consisted of 348 urban pregnant women.
Both groups were selected using a cluster sampling method with probability proportionate to size (PPS) technique25 depending on the cumulative population of pregnant women in the respective Public Health Midwife (PHM) areas. Clusters consisted of randomly selected pre-determined numbers (12-14 from a given clinic) of antenatal women from lists of clinic attendees who belonged to the stipulated period of gestation of a given PHM area. Written, informed consent was obtained from the selected participants. Participants were approached via their respective PHM with the prior approval of the relevant MOH who is in-charge of the antenatal care services delivered in PHM areas under his/her purview.
Dental caries experience and periodontal status was assessed using WHO's Oral Health Surveys Basic Methods 199726. Two examinations were conducted by two examiners trained in WHO criteria and calibrated by the same dental public health specialist. Inter-examiner variability was assessed for both examiners with the specialist separately and reported to be good with Kappa coefficient values of 0.89 and 0.91, respectively, for dental caries and 0.88 and 0.86 for periodontal assessment. Between two examiners this value was 0.93 for dental caries and 0.83 for periodontal status assessment. Consequently, two pilot studies were conducted by two examiners which consisted of 25 antenatal women each. Each tenth participant was re-examined by examiners independently to assess inter-examiner variability which was 0.92 and 0.91 for dental caries. These scores were and 0.87 and 0.89 for periodontal status assessment. In both studies all antenatal women who were approached by the primary healthcare staff of the specific antenatal clinics agreed to participate in the study. The examinations were performed in a suitable room of the antenatal clinic under standardized conditions, stipulated by WHO Oral Health Surveys as under day-light. Dentition status was assessed using plane mouth mirrors and Community Periodontal Index of Treatment Needs (CPITN) probes. No root caries were detected. Sound tooth crown was recorded as 0=carious tooth, 1=decayed tooth, 2=filled with decay/temporary restoration, 3=filled with no decay, 4=missing due to caries, 5=missing due to other reasons such as periodontal disease, orthodontic extractions, 8=unerupted tooth. When assessing missing teeth the reason for the tooth loss was specifically requested. The Decayed, Missing and Filled Teeth (DMFT) index was computed for each individual as the sum of D+M+F: D=total number of teeth recorded as 1 and 2, M= total number of teeth scored as 4 for those who below 30 years and 4 and 5 for those above the age of 30 years27; F=total number of teeth scored as 326. Periodontal status was assessed using the CPITN26.
The data were collected using an interviewer-administered pre-tested questionnaire administered by trained interviewers, and the clinical oral examination described. The questionnaire included socio-demographic information, utilization of dental services and perception of the safety of receiving dental treatment during pregnancy, administered as a sub-question of perceived need for receiving dental care. Those who responded 'no' to the latter were then asked about their perception of the safety of receiving dental care during pregnancy, recorded as 'safe' or 'unsafe'. Data collection for the rural study preceded the urban study and for the urban sample a minor modification was made to the questionnaire regarding the perception of receiving dental care during pregnancy. This was assessed as a separate question worded 'Is it safe to receive dental care during pregnancy?' (response 'Yes' or 'No').
Statistical analyses were conducted using SPSS v13.0 (www.spss.com). Data cleaning was completed by checking the frequency distributions of each variable and performing cross-tabulations as needed to detect and rectify errors. Distributions of continuous variables were assessed for normality. Statistical analysis of the data consisted of computation of frequency and percentages of variables, descriptive statistics such as mean, standard deviation and 95% Confidence Intervals. Independent sample t-test was used to determine significant differences in mean ages DMFT and its components among rural and urban pregnant women. Chi-square test of statistical significance was used to compare the frequencies between the two groups. For the multiple logistic regression analyses, age was categorized as 18-25 years, 26-34 years ≥35 years; caries experience was dichotomized as DMFT=0 and DMFT>0; and periodontal status as CPITN=0 and CPITN>0. However, for periodontal status the only significant association was between age group and CPITN=0 versus CPITN>0 in bivariate analysis for the overall sample (p=0.001). In the multiple logistic regression analysis for caries, experience the backward selection option was used, applying p<0.05 as the criterion for retaining in the model. Hosmer-Lemeshow goodness of fit test was used to assess the fitness of model.
Ethics approval
Ethics approval for the studies was provided by the Ethics Review Committee, Faculties of Medicine, University of Colombo (#EC/08/076) and University of Kelaniya (#P84/07/2009).
The mean age of the rural and urban of antenatal women was 27 ± 4.6 years and 29 ± 4.6 years, respectively (p=0.001). The ages ranged from 18 to 44 years for rural women and 18 to 41 years for urban groups. Of the 459 rural participants, 67.6% had an education level of up to General Certificate of Education: Ordinary Level (GCEO/L; includes 10 years of schooling and a public examination - the benchmark for average education level in Sri Lanka), and an additional 32.4% had completed above O/L level of education; and for the urban women this was 56.0% and 44.0%, respectively. Among both groups, the majority of pregnant women were housewives 71.5% (urban) and 73.8% (rural), respectively. The majority of rural and urban participants were either expecting their first or the second baby: 81.9% and 89.6% respectively; and rural and urban utilization of dental services within past 12 months was 64% and 40.7%, respectively. In all, 29% of rural antenatal women were not willing to receive dental care, irrespective of having oral health problems. Of these, 50% of rural women and 34.2% of all urban women perceived that receiving dental care during pregnancy was unsafe. All these differences were statistically significant (p<0.05) (Table 1).
The burden of dental caries among rural and urban antenatal women was high with a rural prevalence of 91.7%, and 81.3% for urban women. However, rural women had a significantly higher prevalence of dental caries than urban women (p=0.001). The mean DMFT among rural antenatal women were 5.4 ± 3, with 2.27 (± 2.31) decayed teeth, 1.25 (± 1.97) missing teeth, and 1.9 (± 1.89) filled teeth, indicating that average 5 teeth were affected among rural pregnant women with at least 2 of these teeth having untreated dental caries. Among the urban pregnant women, the mean DMFT was 3.69 (± 3.62) with 1.04 (± 2.15) decayed teeth, 1.07 (± 1.59) missing teeth and 1.59 (± 2.06) filled teeth, indicating 4 teeth were affected by dental caries, one of which had untreated dental caries. On average the rural women had as twice as many untreated dental caries compared with their urban counterparts (p=0.001). Rural women also had a significantly higher number of filled teeth (p=0.026). Rural women did not differ significantly from urban women in the number of missing teeth despite having a slightly higher score (p=0.155) (Table 2).
The prevalence of healthy periodontium was as low at 7% for both groups of women. Almost 60% of rural women presented with bleeding gums, while this was only so for 1.7% of the urban women. Similarly, the prevalence of calculus was 30.3% for rural women and 13.5% for urban women. The prevalence of shallow periodontal pockets (4-5 mm) was 3.5% for rural women but 73.0% for urban pregnant women (p<0.0001) (Table 2). Similarly, the prevalence of deep periodontal pockets was 4.3% for the urban group but negligible (0.4%) among the rural women.
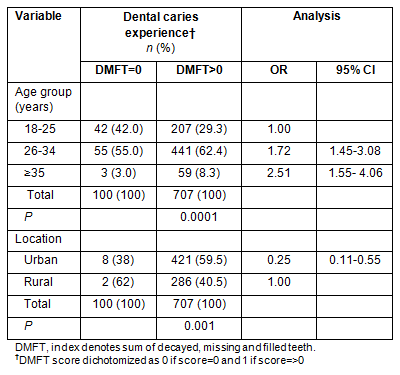
Results are shown for the binary logistic analysis of DMFT for the overall sample of 807 pregnant women (Table 3). The final model predicted 85.4% of the caries experience and the p-value for Hosmer-Lemeshow test of goodness of fit was 0.176 which indicated an acceptable fit of the model. In the final model significant predictors were age (adjusted OR [95% CI]: 2.51 [1.55-4.06], p=0.0001) and location (urban vs rural: adjusted OR [95% CI]: 0.25 [0.11-0.55], p=0.001).
Table 1: Distribution of rural and urban antenatal women by socio-demographic attributes,
utilization of dental services and misconceptions about receiving dental treatment during pregnancy
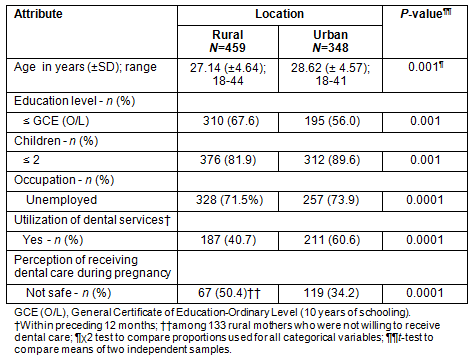
Table 2: Burden of oral disease among urban and rural antenatal women
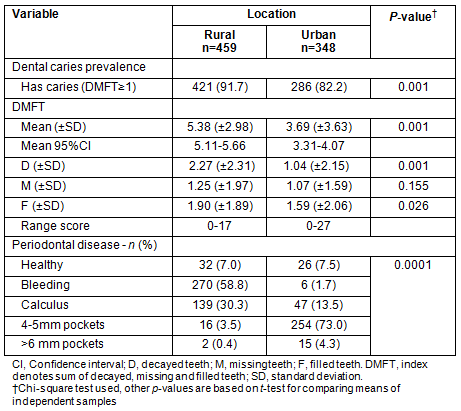
Table 3: Frequency distribution for logistic regression analyses
for dental caries experience: significant predictors of the final model
Discussion
As the findings revealed, antenatal women in Sri Lanka carried a high burden of oral disease with regard to dental caries and periodontal disease, and age was the significant independent predictor for both conditions while location (urban vs rural) was another significant predictor for caries experience. Furthermore, urban women were significantly more likely to use dental care services within the preceding 12 months. However, considerable proportions of antenatal women from both groups had not received dental care/dental screening over past 12 months. Similar studies conducted in developed countries such as the USA have revealed that most women do not access oral health care during pregnancy despite evidence that poor oral health can have an adverse impact on the health of the pregnant woman and her baby28. A recent study reported low utilization of dental care services by Malaysian antenatal women but that those who were aware of the association between poor maternal oral health and adverse pregnancy outcomes were more likely to use those services29.
Dental caries is an infectious and transmissible disease30. Babies are born without Streptococcus mutans, the predominant bacteria attributed to dental caries. There is evidence that women are the primary source of S. mutans colonization in their babies31, and contrary to earlier studies, S. mutans has been shown to colonize the mouths of babies prior to the eruption of deciduous teeth30. Untreated dental caries in women result in high salivary levels of S. mutans which could be transmitted to the baby30; therefore, treating caries and reducing the salivary levels of S. mutans is vital to reducing the incidence of dental caries among their toddlers. The rural women's negative perceptions of receiving dental treatment during pregnancy may have contributed to their lower rates of dental services utilization in the preceding 12 months and higher prevalence of untreated dental caries. Differences in the availability and accessibility of dental care services in urban and rural locales could also have been influential.
That urban women had a higher prevalence of 4-5 mm periodontal pockets despite higher access to dental care may be attributable to the hormonal changes at their stage of pregnancy increasing sensitivity to periodontal disease10. Alternatively, more urban women may have had scaling (professional removal calculus above and below gingivae), which could have facilitated a more accurate assessment of probing pocket depths. Moreover, the main clinical parameter accounted for in periodontal diagnosis by the CPITN index is probing pocket depths, and this may have given rise to an overestimation of prevalence measures32; however, a recent study reported underestimation of prevalence measures using the CPITN index33. Therefore, the periodontal status findings of both groups could have been influenced by inherent measurement issues.
Age was an important factor associated with periodontal status among the whole sample, while age and location were associated with dental caries experience among the women in the multiple logistic analyses (Table 3). This is plausible considering the cumulative oral disease experience with advancing age in addition to the risk posed by the many issues associated with pregnancy, such as hormonal changes, changes in dietary habits and oral hygiene practices. Not surprisingly, older pregnant women have a higher need for oral health care than their younger counterparts.
Limitations
The present findings should be interpreted with caution due to limitations. For example, the rural study was conducted prior to the introduction of the national oral health program for pregnant women and the urban study was conducted soon after its implementation. While dental care is deemed safe during pregnancy, the optimal period is considered to be during the second trimester with emergency services provided at any time34, and this was the reason for selecting the second trimester for the women in the rural study. However, third trimester women were selected for the urban study in order to assess baseline compliance. As a result, the rural and urban women differed in their stage of pregnancy from an average of 9 to 16 weeks and this could have influenced the observed differences in the prevalence periodontal pockets between the two groups. Moreover, the women were not followed up to assess their pregnancy outcomes as this was beyond the scope of the present studies. Future studies are therefore recommended. A minor methodological difference in assessing the perceptions of the safety of receiving dental care during pregnancy between two groups, as described in the methodology, should also be considered.
The performance indicators of the national program of oral health care for antenatal women are reported to be low. For example, as revealed by the Oral Health Unit, Family Health Bureau, in 2010 the Sri Lanka screening coverage was 28% and the treatment completion was 10%. Other than the relatively short duration of its existence, this low performance could be attributed to other factors, such as negative perceptions on safety of receiving oral health care during pregnancy among antenatal women. Service provision issues, which were not addressed by the present study, could be another contributory factor. Previous studies have reported multidimensional barriers to the receipt dental care by pregnant women, including a lack of knowledge, misinformation about safety and the importance of dental care for the mother and the foetus, as well as negative perceptions and practices by dentists themselves32. An in-depth performance appraisal of the national program could identify multidimensional barriers and so contribute to better future management of the program.
Pregnancy is an important milestone in a women's life and it indicates an increased need for dental care, among other health needs. Both rural and urban pregnant women in Sri Lanka carry a high burden of dental caries and periodontal disease. In this study, rural pregnant women were found to have a significantly higher burden of dental caries but urban pregnant women had a significantly higher burden of periodontal pockets. Urban women were more likely to use dental services during pregnancy and had lower negative perceptions of the safety of receiving dental care during pregnancy. Therefore, oral health care services should be made more accessible and acceptable to all rural and urban pregnant women in Sri Lanka through the national program. Negative perceptions of receiving dental care should be addressed by delivering appropriate messages via oral health education programs conducted at antenatal clinics.
Acknowledgements
The two studies reported here were self-funded and the authors alone are responsible for the content and writing of this article. The guidance given by Professor SL Herath, Faculty of Dental Sciences, the University of Peradeniya and Dr RDFC Kanthi, Deputy Director, the Health Education Bureau, Colombo, Sri Lanka is gratefully appreciated. The authors are also grateful to the participants of the two studies and the health staff in the two regions.
References
1. Department of Census and Statistics Sri Lanka 2011. Estimated mid year population by sex and districts 2010. (Online) 2011. Available: http://www.statistics.gov.lk/PopHouSat/Mid%20Year%20Population/midyearsex&district.PDF (Accessed 25 September 2011).
2. Department of Census and Statistics Sri Lanka. Demographic and Health Survey 2006-2007. Colombo, Sri Lanka: Department of Census and Statistics Sri Lanka, 2008.
3. Family Health Bureau Sri Lanka. Home page. (Online) no date. Available: http://www.familyhealth.gov.lk/ (Accessed 8 May 2012).
4. Family Health Bureau, Ministry of Health. Oral health care during pregnancy. Practice Guidelines. Colombo, Sri Lanka: Family Health Bureau, Ministry of Health, 2009.
5. Fernando TPB. Study of the prevalence of pregnancy gingivitis and the attitudes of pregnant women regarding oral health in Nugegoda MOH area (MSc Dissertation). Colombo, Sri Lanka: Post Graduate institute of Medicine, 1991.
6. Rajapakse PS, Nagarathne M, Chandrasekera KB, Dasanayake AP. Periodontal disease and prematurity among Sri Lankan women. Journal of Dental Research 2005; 84: 274-277.
7. Acharya A, Parvati V, Bhat MD. Oral-Health-Related quality of life during. Journal of Public Health Dentistry 2009; 69(2): 75-77.
8. Evans RD, Briggs PF. Tooth surface loss due to pregnancy induced vomiting. Prime Dental Care 1994; 1(1): 24-26.
9. Khamrco T, Saleh KM, A comparative study in dental caries prevalence and treatment needs of pregnant women and single females in Mosul city, Iraq. Al-Raf1dian Dental Journal 2003: 3(1): 57-64.
10. Gursory M, Pajukanta R, Sorsa T, Konen E. Clinical changes in periodontium during pregnancy and post-partum. Journal of Clinical Periodontology 2008; 35(7): 576-583.
11. Straka M. Pregnancy and Periodontal tissues. Neuroendocrinology Letters 2011; 32(1): 34-38.
12. Xiong X, Elkind-Hirsch KE, Vastardis S, Delarosa RL, Pridjian G, Buekens P. Periodontal disease is associated with gestational diabetes mellitus: a case control study. Journal of Periodontology 2009; 80(11): 1742-1749.
13. Xiong X, Buekens P, Vastardis S, Pridjian G. Periodontal disease and gestational diabetes mellitus. American Journal of Obstetrics and Gunecology. 2006; 105(4): 1080-1089.
14. Saddki N, Bachok N, Hussain NHN, Zainudin SLA, Sosroseno W. The association between maternal periodontitis and low birth weight infants among Malay women. Community Dentistry Oral Epidemiology 2008; 36(4): 296-304.
15. Cruz SS, Costa MCN, Gomes-Filho IS, Rezende EJC, Barreto ML, dos Santos CAST et al. Contribution of periodontal disease in pregnant women as a risk factor for low birth weight. Community Dentistry Oral Epidemiology 2009; 37(6): 527-533.
16. Dasanayake AP, Chhun N, Tanner ACR, Criag RG, Lee MJ, Moore AF et al. Periodontal pathogens and gestational diabetes mellitus. Journal of Dental Research 2008; 87: 328-333.
17. Li Y, Cuafield PW, Dasanayake AP, Wiener HW, Vermund SH. Mode of delivery and other maternal factors influence the acquisition of Streptococcus mutans in infants. Journal of Dental Research 2005; 84(9): 806-811.
18. Gunay H, Dmoch-Bockhon K, Gunay Y, Gurtsen X. Effect on caries experience of a long term preventive program for mothers and children starting during pregnancy. Clinical Oral Investgations 1998; 2(3): 882.
19. Ministry of Health. National Oral Health Survey Report 2002-2003. Colombo, Sri Lanka: Ministry of Health, 2009.
20. Kumarihamy SLM, Subasinghe L, Jayasekera P, Kularatna SM, Palipana PD. The prevalence of early childhood caries in 1-2-year-old in a semi-urban area of Sri Lanka. BMC Research Notes 2011; 4: 336. Available: http://www.biomedcentral.com/1756-0500/4/336
21. Gunasekera HR. Some challenges in conducting population and housing census in Sri Lanka. In: Proceedings, 23rd Population Census Conference; 16-18 April 2007; Christchurch, New Zealand, 2007. Available: http://www.ancsdaap.org/cencon2007/Papers/Sri%20Lanka/SriLanka_Gunasekera.pdf
22. Gunasekera HR. Some improvements to 2011 population and housing census in Sri Lanka. In: Proceedings, 25th Population Census Conference; 25-27 May 2011; Seoul Korea, 2011. Available: http://ancsdaap.org/cencon2011/Papers/SriLanka/SriLanka_paper_2011.pdf
23. Lwanga SK, Lemeshow S. Sample size determination in health studies. A practical Manual. Geneva: World Health Organization, 1991.
24. Segovia-Villanueva A. Dental caries associated with dental services utilization in pregnant women. Poster presentation at: ADEA/AADR/CADR Meeting & Exhibition; 8-1 March 2006. Available: http://iadr.confex.com/iadr/2006Orld/techprogram/abstract_73458.htm (Accessed 26 September 2011).
25. Moser C, Karlton G. Survey methods in social investigations, 2nd edn. Aldershot: Ashgate, 2001.
26. World Health Organization. Oral health surveys basic methods, 4th edn. Geneva: WHO, 1997.
27. Malmo University Oral Health Database. Extracts of the third edition of Oral Health Survey Basic Methods. (Online) 1987. Available: http://www.mah.se/CAPP/Methods-and-Indices/for-Measurement-of-dental-diseases/Extracts-from-WHO-Oral-Health-Surveys-3rd-ed-1987/Tooth-status/ (Accessed 8 May 2012).
28. Le M, Riedy C, Weinsten P, Milgrom P. Barriers to utilization of dental services during pregnancy: a qualitative analysis. Journal of Dentistry for Children (Chic) 2009; 76(1): 46-52.
29. Saddiki N, Yusoff A, Hwang YL, Factors associated with dental visit and barriers to utilization of oral health care services in a sample of antenatal mothers in Hospital University Sains Malaysia. BMC Public Health 2010; 10: 75. Available: http://www.biomedcentral.com/1471-2458/10/75
30. Berkowitz RJ. Acquisition and transmission of mutans streptococci. Journal of Californian Dental Association 2003; 31(2): 135-138.
31. Douglass JM, LI Y, Tinanoff N. Systematic review of the association between mutans streptococci in primary caregivers and mutans streptococci and dental caries in their children. Pediatric Dentistry 2008, 30(5): 375-387.
32. Baelum V, Papapanou PN. CPITN and the epidemiology of periodontal disease. Community Dentistry Oral Epidemiology 1996; 24: 367-368.
33. Bassani DG, da Silva CM, Opermann RV. Validity of the Community Periodontal Index of Treatment Needs' (CPITN) for population periodontal screening. Cad. Sa?de P?plica Rio de Janeiro 2006; 22(2): 277-283.
34. Shuk-Yin Lee R, Milgrom P, Huebner CE, Conred DA. Dentists' perceptions of barriers to providing dental care to pregnant women. Women's Health Issues 2010; 20(5): 359-365.


