Dear Editor
Until the recent widespread availability of modern communication technology, traditional paper mood monitoring data was plagued with limited compliance (approximately 11%) and inaccuracy1. This restricted clinicians' capacity to meaningfully assess patient progress or evaluate their treatments by the preferred practice-based method: the single-case experimental design.
A fundamental element of single-case methodology is the collection of repeated measures of behaviour or mood (typically daily or on several occasions per week) from which patterns and stability can be analysed using visual inspection2. This data must also be collected in at least three phases: (i) baseline or pre-treatment; (ii) treatment; and (iii) post-treatment or follow up - with a minimum of 5 data points per phase3. Effective treatment is demonstrated when there is stability of symptoms at baseline (ie minimal slope), followed by a reduction in symptom severity during treatment (ie level), and the maintenance of gains at follow up2(Fig1).
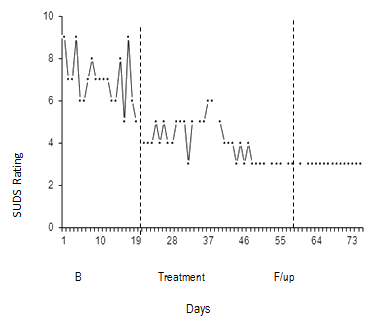
Figure 1: Example of graphed daily subjective units of distress ratings across
baseline, treatment and follow up with dotted lines indicating change in phase.
Our pilot study explored the feasibility of adding the use of technology - text messaging to collect daily ratings and customised software for analysis - to provide accurate data and permit the significance testing of changes in level and slope within a single-case design. Conducted at a rural Australian university psychology clinic, the study involved six patients (3 men and 3 women, mean age 34.33 years [SD 16.11, range 20-63 years]) who were referred by their GPs for treatment for anxiety or depression. Each met the inclusion criterion of willingness to provide daily mood ratings, either via text messaging or in pencil-and-paper format. All participants, except the eldest person who did not own a mobile phone, chose to use text messaging.
To aid participants' understanding, 'mood' was conceptualised as subjective units of distress (SUDS) rated on a 0-10 scale with individualised anchor points that were identified at a pre-treatment interview4. After this, the recording of baseline daily SUDS ratings commenced. At a pre-determined time, participants were sent a text message requesting this data. Ratings were collected for a minimum of 70 days and covered all phases.
All participants were compliant with the daily ratings activity and each missed only 2-5 days during the treatment phase, which ran for 4-6 weeks. The SUDS ratings were analysed using the publically available and simply operated software package SMA v8.3.3 (http://www.clinicalresearcher.org/SMA_Windows_8_4_11.zip). Significant findings were shown, some of which were not readily identifiable on visual inspection.
Our conclusions were that it easy to collect daily mood or distress data from rural patients using mobile phone text messaging (and without the need for commercial packages or iPhone Apps); and that with accurate data and free analysis software, mental health clinicians can evaluate the efficacy of their services using a single-case experimental design. Together these strategies facilitate the assessment of rural mental health outcomes and address a cornerstone of ethical and accountable practice.
Other applications and developments of this technology could include inviting clients to comment on their daily rating, and adding checks for personal early warning signs of problems. Combined, these low-time-demand activities potentially provide significantly increased support and care for clients in rural settings with limited access to mental health services.
Debra A Dunstan, PhD & Susan M Tooth, MPsych
School of Behavioural, Cognitive and Social Sciences
University of New England, Armidale
New South Wales, Australia
References
1. Stone AA, Shiffman S, Schwartz JE, Broderick JE, Hufford MR. Patient compliance with paper and electronic diaries. Control Clinical Trials 2003; 24(2): 182-199.
2. Kazdin AE. Research Design in Clinical Psychology, 4th edn. Sydney, NSW: Allyn and Bacon, 2003.
3. Borckardt JJ, Nash M, Murphy M, Moore M, Shaw D, O'Neil P. Clinical practice as natural laboratory for psychotherapy research. American Psychologist 2008; 63(2): 77-95.
4. Wolpe J, Lazarus AA. Behavior Therapy Techniques. New York: Pergamon, 1966.
