full article:
Namibia is a vast country with a population of only 2.1 million1, making it the second least densely populated country in the world. Long distances between health facilities challenge the provision of healthcare services, especially for rural communities. This is illustrated by an average one-way distance of 64 km for rural communities to access a clinic and 107 km to access a hospital in rural areas2.
With an estimated HIV adult prevalence rate of 13.4%3, AIDS has serious public health consequences, including impacts on morbidity and mortality of the Namibian workforce4. Non-communicable diseases (NCDs) are steadily becoming a larger public health problem in both developed and developing countries5, and Namibia is no exception. A recent household survey in Windhoek, the Namibian capital, revealed prevalence of age-adjusted hypertension of 38.0%, obesity of 17.0% and family history of diabetes, hypertension or heart disease of 5.5%6. Increasing incidence of NCDs will be an additional threat to the productivity of the Namibian workforce7. During World Health Day 2013 (9 April 2013), the Namibian Deputy Minister of Health called for acceleration of actions to address the risk factors contributing to NCDs, especially hypertensive stroke8.
Since July 2009, Namibia has been classified by the World Bank as an upper middle income country9. The experience among health stakeholders is that this has led to a decrease in development assistance for health, which is expected to decrease further. As Namibia's total health expenditure has been heavily dependent on donor funding - an estimated 21.7% of total health expenditure in 2008/2009 was funded by foreign donors10 - innovative approaches are required to ensure the challenges in health care are addressed whilst donor funding is decreasing. The present article describes such an innovative public private partnership model: Bophelo!, a mobile voluntary HIV counseling and testing and general wellness screening (for NCDs) program at workplaces. Bophelo! is compared to a program with fixed site voluntary counseling and testing (VCT) provided by the New Start centers in Namibia.
Bophelo!, a word meaning 'life', is a program run by PharmAccess Foundation, a Dutch non-governmental organization, and has been operational in Namibia since March 2009. It was established as a public-private partnership project between PharmAccess Foundation, the Namibian Business Coalition on AIDS and the Namibian Institute of Pathology, supported by the Namibian Ministry of Health and Social Services (MoHSS). The program aims to provide VCT as well as general wellness screening to the working population of Namibia. Apart from HIV testing, the services provided include determination of blood pressure and rapid testing for blood glucose levels, cholesterol levels, hemoglobin levels, hepatitis B and syphilis. In addition, symptoms of tuberculosis are registered, as well as weight, height, and waist circumference and body mass index.
Bophelo! uses two mobile testing clinics mounted on trucks, containing all equipment necessary to provide patient education, confidential counseling and conduct screening tests. The mobile clinics are licensed by MoHSS to provide VCT, and quality assurance is provided by the Namibian Institute of Pathology.
Bophelo! can be contracted by companies to provide wellness testing at workplaces as an annual or biannual activity. The companies pay for part of the operating costs, with the remainder of the costs paid by the Namibia Global Fund Programme and other donors. Once contracted to visit a work site, all workers who seek HIV screening are offered pre- and post-test counseling. A strict time schedule is kept to minimize disruption, with employees being away from their work for less than 1 hour. Both rural and urban companies have engaged Bophelo! in mobile site wellness testing. Table 1 shows basic information about the organizations that participated in testing.
In general the organizations were invoiced for separate categories of cost, such as the staff time, mobile unit costs (such as insurance and maintenance), staff accommodation, and kilometer charges. Each itemized invoice clarified which items would be covered by subsidy from Namibia Global Fund Programme funds. The organizations paid the remaining items.
Patients remain in the consultation room during the time that the blood tests develop and this time is used for other screening tests, as previously described. This means that all patients receive their results directly after the tests have been performed. Patients who test positive for HIV, syphilis or hepatitis B, or have abnormal values for the anthropometric measurements, are given a referral letter to a private physician (if insured), or to a MoHSS health facility (if not insured), indicating the condition(s) detected. In addition, these patients receive a form without diagnosis, requesting time off from the employer to visit a referral facility. This form is to be signed by the provider and returned to the employer as evidence that the employee did seek further medical consultation. To ensure confidentiality of patients, employers are only provided anonymous summary reports on the health indicators for their workers.
As part of the Bophelo! screening process, patients complete a self-administered survey (a standardized list of questions on their knowledge, attitudes and behaviors regarding the diseases being screened for). This information, along with the test results, is gathered in a fully anonymous database, without individual identifiers.
Since 2004, VCT in Namibia has also been provided through 18 fixed site New Start centers funded by USAID. New Start's focus in 2009 was on the provision of VCT either through standalone VCT centers or as a fixed site VCT center within an existing public, or faith-based, health facility. The VCT provision at the New Start centers is of a much larger scale than Bophelo!: New Start tested 70 143 people in 2009 compared to 5967 through Bophelo. This research focuses on the period March 2009 until December 2009, as the Bophelo! project was not in full operation during January and February 2009. Figure 1 shows the testing locations of both Bophelo! and New Start.
In Kenya it has been shown that mobile VCT provision is superior to fixed site VCT in terms of cost per client and reaching previously untested populations11. However, the mobile VCT model evaluated in that study differed markedly from Bophelo! In Kenya, mobile VCT was provided as a standalone service (without testing for NCDs), the testing vehicle would stop and reside within community settings (as opposed to workplaces) and the program was commissioned by a donor agency (as opposed to a public-private partnership).
The key objective of the present study was to make a detailed assessment of the costs of an innovative mobile VCT and wellness service delivery in Namibia (Bophelo!), compare these to non-mobile services and to put these different costs in the context of the effectiveness of mobile versus non-mobile service delivery. Given these differences, the present study compares fixed site VCT to a novel form of mobile VCT, rather than mobile VCT as provided in the Kenya example.
Table 1: Organizations at which Bophelo! mobile voluntary counseling and testing took place in 2009
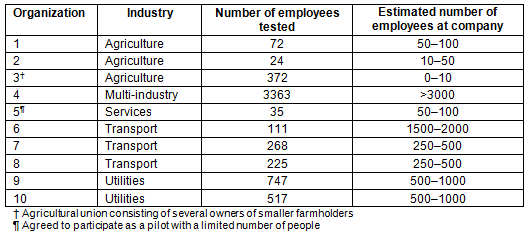
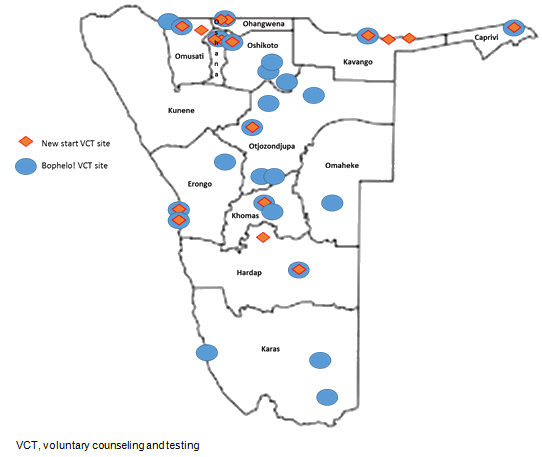
Figure 1: Locations of New Start and Bophelo! voluntary counseling and testing sites in Namibia.
The underlying objective of the cost study was to obtain and allocate the full direct cost of all resources used by the mobile (Bophelo!) and fixed (New Start) site testing programs, including depreciation of capital investment and the market value of any services or goods that were provided for free. Costs of preparation and planning in previous years were not included, neither was any out-of-pocket cost to patients. Employer payments to Bophelo! are included, but any additional employer costs, such as employee time lost to screening, are not. Cost data were collected through reviewing financial information, such as general ledgers, invoices and petty cash registers of the organizations involved in both Bophelo! and New Start, and discussions with financial and operational managers.
As far as possible, costs are based on actual expenses. However, assumptions were made for some cost categories, such as senior management time, depreciation and overheads. One key assumption was that providing a HIV test alone would take around 85% of the time of providing a full wellness screening. The estimated incremental costs of providing wellness screening comprise:
- costs of additional test kits for wellness screening tests
- additional staff time of 17.65% (15/85) compared to providing only HIV testing.
Further background and detail on the assumptions is explained in Boston University's report 'Comparison of key unit costs and outcomes for mobile and fixed site screening/testing programs in Namibia'12.
PharmAccess maintains an anonymous Microsoft ACCESS database with all wellness and HIV test results and the outcomes of the survey as well as socioeconomic and risk factors for each worker tested. These data were used as a basis for analyzing the health status of employees tested by Bophelo! from March 2009 until December 2009.
The counselors at New Start centers capture client data on demographics, risk factors and test results on a client intake form. These data are later transferred into an electronic database, kept by New Start centers using coded patient identifiers. Data from the pre-test, HIV testing and post-test are also captured. Data from both PharmAccess and New Start centers were captured and analyzed using Microsoft Excel.
The study has focused on the costs of delivering VCT through mobile and fixed site facilities. As such the study did not include the full cost and benefit to employers, which is beyond the scope of this study and for which there is insufficient data.
Ethics approval
Ethical clearance to conduct the study was obtained both from the Namibian Ministry of Health and Social Services (17/3/3AP), and Boston University's Institutional Review Board (H-29197(BU)).
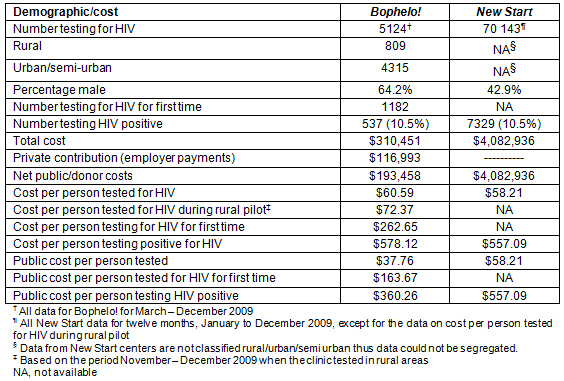
Table 2 summarizes the key findings from the study; a more complete description of the findings follows.
Table 2: Results summary of all data for Bophelo!, March - December 2009, and all New Start data, January - December 2009
Demographics and bio-data
From March 2009 to December 2009, 5734 patients were screened in the Bophelo! mobile clinics. This represents 1.7% (5734/331 44413) of the formally employed population in Namibia in 2008. Some patients (10.6%, 610/5734) declined the HIV test, but were tested for other conditions. Of all people tested for HIV, 23.1% (1182/5124) were tested for HIV for the first time. All patients tested for HIV received their results, as the additional wellness testing meant that the patient remained within the consulting room during the development of the HIV test. Although data is not available for the number of patients leaving before test results are made available at New Start, this is a recognized problem for VCT in general and something that seems to have been avoided by the additional wellness testing.
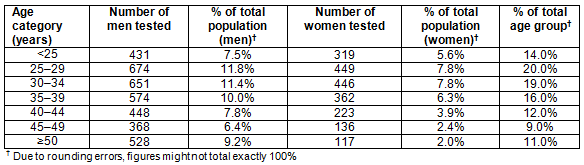
The majority of patients screened classified themselves as laborers (75%, 4194/5579) (These data were only available for 5579 clients, representing 97.3% of the population tested for wellness screening from March to December 2009), with the remainder classifying themselves as management (6.4%, 358/5579), supervisor (7.6%, 423/5579) and administration (10.8%, 604/5579). Demographic data were only available for 5726 clients (99.9% of the population for wellness screening from March to December 2009); 55.1% of the patients screened were between the ages of 25 and 39 years and 64.2% were male (3674/5726) (Table 3). These data were not available from New Start testing sites.
Table 3: Age/sex distribution of people receiving testing at Bophelo!
A total of 10.5% (537/5124) of the workers tested positive for HIV. Some of those who declined the HIV tests might already have known that they were HIV positive, hence the actual prevalence rate in the tested population could be higher than the recorded 10.5%. The data show that 50.6% (2900/5734) of all screened patients tested positive for one or more conditions, including HIV. A similar proportion of those tested at New Start centers also tested positive for HIV (10.5%, 7329/70 143).
Costs of service delivery
In 2009, the average cost per person tested for HIV at the Bophelo! mobile clinic was calculated at US$60.59 (US$310,451/5124 people). Per newly identified HIV case, the cost is US$578.12 (US$310,451/537 people). The incremental cost associated with testing for conditions other than HIV infection was US$11.35, an increase of 18.7%, consisting of the costs of additional test kits (US$8.62, including a 10.0% provision for wastage) and additional staff time per person tested (US$2.73). This is based on the period March to December 2009. During November and December 2009 only rural areas were visited. During this time, the cost per person was US$65.86 (US$58,546/889 people). The higher costs are caused by higher accommodation, per diems and fuel costs.
The costs of testing one person for HIV at the New Start centers in 2009 were US$58.21 (US$4,082,936/70 143 people) versus US$60.59 (US$310,451/5124 people) at the mobile Bophelo! clinics, a difference of 3.9%. Although the scale of VCT provision at the New Start centers is much larger than the mobile Bophelo! operation, costs were comparable.
The cost per newly identified HIV case at New Start was US$557.09 (US$4,082,936/7329 people). Although the cost per newly identified case is slightly higher at the Bophelo! mobile site, caution should be exercised in interpreting this finding, given the different demographic of the testing population.
With respect to external support, the mix of funding for the two programs was very different. The fixed site New Start centers were 100% funded by public sector resources, in this case through donor support from USAID. At Bophelo!, 37.7% (US$116,993/US$310,451) of costs were borne by the private sector (employers), with the remainder paid for by donors (US$189,982.14, 61.20%) and the Namibian public sector (US$3476.12, 1.1%). On average, employers paid US$22.83 per person tested for HIV (US$116,993/5124 people) in the Bopehlo! clinics, with the public donors contributing US$37.76 (US$193,458.26/5124 people). Full costs to the employer would additionally include the costs of lost staff time. However, there is insufficient data available to carry out this analysis.
As the fixed site New Start VCT operations in this case were fully publicly funded, the total cost to government and donors was US$58.21 per person tested, while the total cost of the Bophelo! operations to the public sector per person tested was 65% of that. Private formal sector payments reduced the public cost for Bophelo!'s wellness screening program, while the lower overhead and lower staff costs offset the added costs of the mobile clinic (petrol, vehicle maintenance, staff travelling allowances).
Mobile services providing increased outreach to difficult target groups
Of all patients, 64.2% (3674/5726) tested through Bophelo! were male. In comparison, at the New Start centers this figure was 42.9% (3674/5726). In addition, the mobile service has been able to provide access to rural populations, which might otherwise struggle to access VCT services. This is illustrated by the Bophelo! data over the last 2 months of 2009, when the mobile clinic visited rural areas in Namibia. Of the patients tested for HIV in this period, 43.6% (353/809) had never tested for HIV before, compared to 19.2% (829/4315) first time testers in urban and semi-urban areas.
Bophelo! requires those being tested to leave their workplace for 1 hour, costing the employer in terms of lost staff time. New Start clinics were only open during normal working hours. In order to be tested at these fixed sites, staff would need to be absent from work for substantially longer than this 1 hour as they would need to travel to the clinic and potentially incur waiting time. The costs to the employer in terms of lost time for mobile testing are therefore likely to be substantially lower than they would be for granting leave for fixed site testing. However, the full cost and benefit to the employer was outside the scope of this study.
Screening for additional diseases
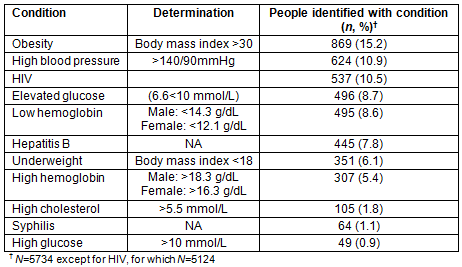
The present study demonstrates that one of every two people screened by Bophelo! (50.6% (2900/5734)) tested positive for one or more chronic conditions, including HIV. A total of 15.2% (869/5734) of the population was classified as obese and 10.9% (624/5734) was diagnosed with high blood pressure. Table 4 provides more details on the prevalence of all conditions screened during 2009.
Table 4: Prevalence of various conditions for Bophelo! screening program, March - December 2009
Discussion
This study has shown that mobile VCT (Bophelo!) can be comparable in costs to fixed site VCT (New Start) in Namibia. This finding is not unique to this study and demonstrates that well run mobile services can operate effectively in rural and remote areas to provide services to otherwise hard-to-reach populations who do not have easy access to fixed sites. A study in South Africa indicated that a mobile male circumcision program was able to compete in price with fixed site settings, partly due to the lower operating overheads14.
Moreover, the Bophelo! program has shown that private funding can be mobilized for a mobile wellness screening program - including VCT and screening for risk factors of NCD. Although concern for their employees motivated some employers to financially contribute to the cost of screening, most were interested in acquiring an understanding of the health risks within the workforce, to plan workplace programs and access to care interventions, based on the anonymous statistical reports provided to the organization after the screening. It could be assumed that employer willingness to invest in screening of employees and the utilization of the information obtained from the anonymous statistical reports to plan workplace program interventions would have resulted in employers providing more support to HIV positive workers and/or access to general health care for their employees. Further research would be required to test this assumption.
Employer payments reduced the costs to the public sector to 65% of the unit cost of fixed site testing. This is a particularly important finding for middle-income countries seeking to provide access to services for rural and remote populations in the face of donor funding that is declining and expected to decline further.
Besides reducing the cost to the public sector, mobile clinics provide opportunities for wellness testing of groups that are normally difficult to reach, such as rural and remote communities. Due to low population density, distance from urban clinics, geographic barriers, lack of transportation and other reasons, many rural and remote residents are isolated from fixed VCT services. The high percentage of rural individuals who elected to take a HIV test in the Bophelo! mobile clinics is an encouraging lesson that such populations will have testing when facilities are easily accessible.
Men, and especially young men, are a difficult target group to reach with VCT, as illustrated in a 2004 study in South Africa where only 15% of men aged 15-24 years had tested for HIV before, compared to 25% of women in the same age group15. More recent findings from South Africa provide evidence for the use of mobile VCT as the method of choice in reaching men in both rural and urban settings16. The large proportion of men tested in the mobile VCT program (Bophelo!) is consistent with these findings and suggests that hard-to-reach men can be effectively reached through mobile VCT services. This finding should be replicable in other countries in Africa and globally where fixed site services are distant or difficult to access.
There are three main explanations for increased access among men. First, as men make up the majority of the formal work force in Namibia13, mobile wellness screening at workplaces can be an effective way to reach men who are reluctant or unable to visit fixed site testing centers, or who believe they are not at risk of HIV infection. Second, mobile services are able to access workforces and populations that normally do not have easy access to testing facilities. Bophelo! illustrated this by recording a first-time HIV tester rate of 43.6% in the rural areas, versus 19.2% in urban and semi-urban areas.
Finally, and unique to Bophelo! in mobile VCT testing, clients are offered tests for multiple chronic conditions. As a result, participants do not self-identify as being 'at risk' of HIV infection when they visit Bophelo! A recent qualitative study from Zambia found that people ascribed different negative meanings to visiting standalone VCT centers. Such visits suggested morally reproachable sexual behavior and the likelihood of HIV positive status17. These stigmatizing attitudes were facilitated by the location of VCT centers that put clients in full view of onlookers who might come from the same community as the client. It is not possible to prove through this study the impact of multiple condition testing on VCT stigma. However, the high percentage of men reached for VCT through mobile services indicates that stigma might have been reduced because Bophelo! offered holistic wellness screening, not just VCT. Further research is required in this area, but packaging VCT in a broader screening program may reduce the stigma associated with HIV testing for rural, remote and even urban populations.
Bophelo! demonstrated that wellness testing, including tests for indicators of NCDs, could be added to HIV testing at low (18.7%) incremental costs. Complementary NCD testing provided employees with more information on their health status and the opportunity to take preventive action. Screening for chronic NCDs addresses a growing need in both developing and developed countries. The number of people affected by NCDs will increase substantially in the next decades as populations age. Half of the deaths from NCDs will occur in the most productive age groups18 and the latest Global Burden of Disease study indicates NCDs account for a significantly greater fraction of mortality in low and middle-income countries in 2010 compared to 199019. Mobile wellness testing services could therefore be an important tool in tackling the NCD challenges, especially in rural and remote communities.
Limitations of the study
Due to the operational nature of the study, which focused on the comparison of the direct costs of providing the services, this study was not designed to collect data or perform an analysis of the costs to the employer. The study could not compare cost of testing in rural and urban/semi-urban sites, nor the cost of first-time HIV testers, as these data were not available from New Start Centers. Costings used 5-year depreciation for the vehicles and an imputed rent for the fixed site facilities. This provides a rough comparison of the allocated capital costs, but is subject to uncertainty both in the useful life and imputed rent.
At the time of the study, neither fixed nor mobile testing facilities were able to collect data tracing patients into the public healthcare system for follow-up support. Patients who presented at fixed sites may be more likely to follow through with referral and care because of the proximity of health facilities, but no data were available to test this hypothesis. Maximizing the probability of follow-up on a positive diagnosis is a factor to consider in the roll-out of mobile testing services in other settings.
The experience in Namibia and employer recognition of the difficulty of follow-up led to the implementation of a subsequent mobile primary healthcare service - the Mister Sister Mobile Primary Healthcare Clinics. This service commenced in 2010 as a public-private partnership between PharmAccess Foundation and the Ministry of Health and Social Services in Namibia, providing monthly primary healthcare services to rural, remote and underserved populations in an attempt to narrow the treatment gap and follow up on referral services.
In Namibia, mobile clinics provide cost-effective HIV and wellness testing services at the workplace. As a consequence, in 2011 both the Ministry of Health and Social Services (funded by the Centers for Disease Control and Prevention) and IntraHealth (which runs the New Start program funded by USAID) commissioned mobile clinics identical to the Bophelo! clinics to expand the provision of mobile VCT and special program services in Namibia. These new clinics continue to work together with PharmAccess to improve mobile healthcare delivery.
The lessons learned from this innovative mobile screening service could be used to develop similar wellness screening programs in Africa and low-middle countries with rural/remote health access barriers.
References
1. Namibia Statistics Agency. Namibia 2011 population and housing census preliminary results. (Online) 2011. Available: http://nsa.org.na/microdata1/index.php/catalog/19/related_materials (Accessed 12 November 2015).
2. PharmAccess Foundation. Agriculture employer association of Namibia survey. Namibia: PharmAccess Foundation Namibia, 2007.
3. UNAIDS. Namibia UNAIDS factsheet. (Online). Available: http://www.unaids.org/en/regionscountries/countries/namibia/ (Accessed 30 April 2013)
4. International Labour Organization. ILO programme on HIV/AIDS and the World of Work Branch (ILOAIDS). (Online). Available: http://www.ilo.org/aids/lang--en/index.htm (Available 2 May 2013).
5. World Health Organization. Global status report on noncommunicable diseases 2010. (Online). Available: http://www.who.int/nmh/publications/ncd_report2010/en (Accessed 2 May 2013).
6. Hendricks M, Wit F, Roos M, Brewster L, Akande T, de Beer I, et al. Hypertension in sub-Saharan Africa: cross-sectional surveys in four rural and urban communities. PLOS One 2012; 7(3): e32638.
7. Guariguata L, de Beer I, Hough R, Bindels E, Weimers-Maasdorp D, Feeley F 3rd, et al. Diabetes, HIV and other health determinants associated with absenteeism among formal sector workers in Namibia. BMC Public Health 2012; 12: 44.
8. Kamwi R. Keynote address on the commemoration of World Health Day, 9 April 2013.
9. World Bank. World Bank development indicators: Namibia. (Online). Available: http://data.worldbank.org/country/namibia (2 May 2013).
10. Namibia Health Resource Tracking. HIV/AIDS resource tracking: 2007/08 and 2008/09. Namibia: Ministry of Health and Social Services. 2010.
11. Grabbe K, Menzies N, Taegtmeyer M, Emukule G, Angala P, Musango G, et al. Increasing access to HIV counseling and testing through mobile services in Kenya: strategies, utilization, and cost-effectiveness. Journal of Acquired Immune Deficiency Syndromes 2010; 54(3): 317-323.
12. Feeley F 3rd, Bindels E, Rinke de Wit T, de Beer I. Comparison of key unit costs and outcomes for mobile and fixed site screening/testing programs in Namibia. (Online). 2010. Available: http://www.bu.edu/cghd/files/2010/12/Bophelo-Report-FINAL-for-CGHD.pdf (Accessed December 2010).
13. Ministry of Labour and Social Welfare (Namibia). Namibia labour force survey 2008. Namibia: Ministry of Labour and Social Welfare, 2010.
14. Soboil N, Cockburn D, Rech D, Taljaard D. A comparative analysis of two high-volume male medical circumcision (MMC) operational models with similar service delivery outcomes in different settings within Gauteng and KwaZulu-Natal provinces in South Africa: Urban Centre for HIV/AIDS Prevention Study. Poster presentation at XIX International Aids conference, Washington DC, 2012.
15. Pettifor A, Rees H, Steffenson A, Hlongwa-Madikizela L, MacPhail C, Vermaak K, et al. HIV and sexual behavior among young South Africans: a national survey of 15-24 year olds. Johannesburg: Reproductive Health Research Unit, University of the Witwatersrand, 2004.
16. Van Rooyen H, McGarth N, Chirowodza A, Jospeh P, Fiamma A, Gray G, et al. Mobile VCT: reaching men and young people in urban and rural South African pilot studies (NIMH project accept, HPTN 043). AIDS and Behavior. 2012; 11.
17. Jurgensen M, Tuba M, Fylkesnes K, Blystad A. The burden of knowing: balancing benefits and barriers in HIV testing decisions. a qualitative study from Zambia. BMC Health Services Research; 2012; 12: 2.
18. Bloom B, Cafiero E, Jané-Llopis E, Abrahams-Gessel S, Bloom L, Fathima S, et al. The global burden of disease: generating evidence, guiding policy. Institute for Health Metrics and Evaluation, 2011.
19. Institute for Health Metrics and Evaluation. GBD 2010: change in leading causes and risks between 1990 and 2010. (Online) 2012. Available: http://vizhub.healthdata.org/gbd-compare/ (Accessed 12 November 2015).




