Introduction
There has been a significant increase in the burden of renal disease among Aboriginal Australians over the past 15 years in particular. Recently, rates of end-stage renal disease (ESRD) among Aboriginal Australians and New Zealand Maori people were reported as being eight-fold that of non-Indigenous people1. In Australia, rates of ESRD among Aboriginal people are generally correlated with increasing degrees of remoteness, with some of the highest rates recorded in the desert regions of the Northern Territory, Western Australia, South Australia and far western Queensland2. Aboriginal communities from these remote regions often have poor access to health care and general goods and services, especially fresh foods. They may also endure poor environmental living conditions and poor water quality, and suffer social disadvantage which may contribute to their susceptibility to chronic disease3.
Renal disease is often asymptomatic and, as a result, effective health strategies are needed for the early identification of Aboriginal people at risk of renal disease4,5. Once detected, there is a critical requirement for structured renal disease management programs because the natural history of renal disease is amenable to change through the judicious use of antihypertensive agents such as angiotensin-converting enzyme inhibitors (ACEI) and angiotensin receptor blockers6-10.
Urine albumin:creatinine ratio (ACR) is a well established biochemical marker of microalbuminuria11-13 and this test can now be conveniently performed on the DCA 2000 point-of-care (POC) analyser (Bayer Australia; Melbourne, VIC, Australia)14. Point-of-care testing (POCT) enables this pathology test to be performed on-site in the Aboriginal community setting by a trained health professional, with quantitative results available within 7 min. The Community Point-of-Care Services unit at Flinders University conducted the first scientific validation of this new POC test in Australia14 and pioneered its application in the Aboriginal community setting through the Umoona Kidney Project15-16.
The Umoona Kidney Project was a cooperative partnership between the Umoona Tjutagku Health Service at Coober Pedy in South Australia's far north, 850 km north of Adelaide, and the renal units at the Flinders Medical Centre and the Women's and Children's Hospital, Adelaide. The project was conducted between 1998 and 2000 and the aims of the adult component of the project were two-fold: to determine the renal disease risk profile among the community's adults, and to implement a management program to monitor those adults at greatest risk of developing ESRD.
The results of the risk assessment arm of the program have previously been reported15-16. This article describes the results of the renal disease management program, and investigates the following research questions: can urine ACR POCT be applied in a culturally and clinically effective manner within a management framework, and how well was the Umoona Kidney Project accepted by the community?
Methods
Community consultation
Prior to commencing the management program, clinical and scientific staff from the Renal Unit at Flinders Medical Centre conducted a series of meetings with the community and the Board of the Umoona Tjutagku Health Service to discuss the most culturally appropriate way of introducing the management program. A culturally appropriate community brochure on the Umoona Kidney Project was also developed by the community nurse, members of the Aboriginal health worker team and the program manager, and disseminated to all members of the community.
Ethics approval
Ethics approval to conduct the management arm of the Umoona Kidney Project was obtained from the Aboriginal Health Research Ethics Committee of South Australia and the Flinders Medical Centre Committee on Clinical Investigation.
Flinders renal clinical and scientific team
The Flinders Medical Centre renal team consisted of the program manager/scientist, two renal specialists (one male and one female), a scientist responsible for patient data management and a nutritionist. A Flinders' medical student (MB) was supported through a National Health and Medical Research Council (NHMRC) scholarship to conduct a community satisfaction survey.
Selection of patients for inclusion in the management program
Following completion of the risk assessments, adult members of the community considered at highest risk for renal disease underwent a clinical and pathology review by the Flinders renal specialists. The ACEI perindopril was subsequently offered to members of this group clinically assessed as being likely to receive the greatest benefit from this intervention.
All persons were individually counselled about the potential benefits and side-effects of ACEI medication (in particular angioneurotic oedema and anaphylaxis, which required urgent medical attention). Significant adverse side-effects were required to be reported to Flinders Medical Centre's Committee on Clinical Investigation and the Clinical Drug Trials Committee.
The following people were excluded from treatment with ACEI: pregnant or breast-feeding women, women at risk of becoming pregnant, persons with a plasma potassium greater than 5.2 mmol/L, and persons who had a demonstrated history of intolerance to ACEI.
Similar to the risk assessment arm, participation of Umoona community members in the management phase of the project was voluntary, and patients could withdraw from taking medication at any time and for any reason. Every patient who voluntarily participated in the study signed a written consent form in the presence of the clinic nurse and an Aboriginal health worker.
Monitoring of patients in the management arm
Adults participating in the management arm were initially given a low dose of ACEI to assess their tolerance and identify any side-effects. Blood pressure, plasma potassium and creatinine were measured within 7 to 10 days after initiation of treatment (via a blood sample sent to the pathology laboratory at Flinders Medical Centre). Subsequently, dosage of medication was titrated to a maximum of 8 mg for each person, with the aim of achieving a target blood pressure of less than or equal to 130 mmHg (systolic) and 80 mmHg (diastolic). If the patient's blood pressure was still high after reaching the maximal effective dose of ACEI, then a calcium channel blocker was added to achieve optimal blood pressure control.
Adults in the management arm were monitored at every field visit through clinical consultation with the Flinders renal specialists. Selected renal markers, including urine ACR by POCT, plasma electrolytes, urea, creatinine, calculated glomerular filtration rate and blood pressure were measured at six-monthly intervals or when deemed appropriate by the specialists. The urine ACR was measured on a fresh first morning urine sample by POCT, with responsibility for urine ACR testing taken by the Umoona Aboriginal health worker team. Dipstick urinalysis was performed prior to urine ACR POCT and a microscopic urine assessment was conducted on-site if the urine specimen exhibited gross proteinuria or haematuria. A 24 hour urine protein estimation was performed on those patients who were or had become macroalbuminuric (urine ACR greater than or equal to 30 mg/mmol). HbA1c was measured at three-monthly intervals for patients with diabetes and 12 monthly for non-diabetics. Possible side-effects (including angioneurotic oedema, persistent cough, hypotension and dizziness) were monitored at every visit.
In addition to on-going specialist renal advice, patients were counselled on the management of other risk factors such as alcohol, smoking and obesity. The Flinders nutritionist also provided dietary advice.
Compliance
Patients in the management arm of the program were required to visit the Umoona clinic on a monthly basis to collect their tablets in individual dosette boxes prepared by the Umoona clinic nurse. Compliance was assessed through monthly tablet counts performed by the clinic nurse and recorded on a manual record system, which detailed the date when a new supply of tablets was given to each patient and the date when the patient returned to collect their next month's supply.
Patients were considered compliant if they took at least 80% of required number of tablets each month. Patients were considered non-compliant if they took less than 50% of their monthly tablets. For patients who were non-compliant or who were unable to attend the clinic, Umoona's Aboriginal health worker team prepared individual dosette boxes which were hand-delivered to patients as part of a 'tablet run' at 8 am each morning.
Electronic documentation of management plans
Patient information was entered electronically into a data management program (designed specifically for the program using Microsoft Access software). To maintain confidentiality each patient was assigned an individual code number which was used as their unique identifier in their electronic record. The master copy of the electronic data base was held by the scientist responsible for data management, while access to the data base was password protected and restricted to the data manager, project manager, renal specialists and clinic nurse. All biochemical and haematological results and current treatment plans could be reviewed, in chronological sequence, at each clinical consultation by the renal specialist team.
Point of care testing for urine ACR
The program manager delivered a series of continuing education and training sessions about urine ACR POCT for Umoona's Aboriginal health worker team, during which the health worker team was progressively taught how to conduct urine ACR POCT on patients, and how to perform quality control testing to monitor the analytical performance of the DCA 2000. A series of laminated posters were provided to visually demonstrate the step-by-step processes of urine ACR patient and quality testing on the DCA 2000. By the commencement of the management arm of program, Umoona's Aboriginal health worker team had taken over responsibility for urine ACR POCT on the DCA 2000.
The POCT analyser provided a quantitative measurement of urine albumin by immunoturbidimetry, urine creatinine by colorimetry and then calculated the ratio of these analytes17. The urine ACR test, performed using DCA 2000 Microalbumin/Creatinine reagent kits (Bayer Australia; Melbourne, VIC, Australia), was completed in 7 min on the DCA 2000.
The DCA 2000 Microalbumin/Creatinine Low and High Control kit (Bayer Australia; Melbourne, VIC, Australia) was used to monitor the analytical imprecision of urine ACR POCT results. A quality control test (alternating between the low and high control) was performed each time a new reagent kit (containing 10 cartridges) was opened. Imprecision was calculated as a coefficient of variation (CV%) from the results of repeated analysis of each quality control sample.
Survey of community satisfaction
The Flinders NHMRC medical student developed a culturally appropriate questionnaire to assess community attitudes towards, and satisfaction with, the Umoona Kidney Project18. The satisfaction tool for this project was initially developed in collaboration with behavioural psychologists from the School of Medicine at Flinders Medical Centre. The tool was then validated for its cultural appropriateness through consultation with the manager of the Aboriginal Health Unit at Flinders Medical Centre and the director, health workers and the clinic nurse from Umoona. The Board of the Umoona Tjutagku Health Service approved the final questionnaire. The questionnaire was implemented at the community level by Umoona's Aboriginal health workers, the nurse-in-charge, two community leaders and the medical student.
The questionnaire consisted of a series of general questions, to which multiple responses could be ticked, and a set of more specific questions, based on the 5 point Likert scale19, with respondents rating their attitude to the questions posed (from 'very much yes' to 'very much no').
Statistical analysis
Analysis of the results from patient management was performed using both the Analyse-It (Analyse-It Software Ltd; Leeds, UK) and SPSS Version 12.0.1 for Windows (SPSS Headquarters; Chicago, IL, USA) statistical software packages.
Results
On-site consultations with the renal specialists
The Flinders renal specialists conducted 231 patient consultations in the two years of the management program, with over 70% of patients having four or more (up to a maximum of eight) consultations.
Patients entering the management program
Fifty-seven community members were initially assessed as being at risk for renal disease. All were overtly hypertensive (systolic blood pressure greater than 140 mmHg and/or diastolic blood pressure greater than 90 mmHg over three independent blood pressure readings), hypertensive (systolic blood pressure greater than 130 mmHg and/or diastolic blood pressure greater than 80 mmHg over three independent readings) with other risk factors, or had diabetes. Ten patients were excluded from the study because they were already taking ACEI medication, they were women of child-bearing age, their blood pressure had improved through lifestyle changes, or their blood pressure was too unstable to commence treatment. Six males did not wish to participate for personal reasons, while four people no longer lived in the community. Two commenced a low dose of ACEI, but were withdrawn due to a perceived increase in aggression (n = 1) or to the effects of headache, itchiness and minor numbness on the right side of the face (n = 1). The remaining 35 patients voluntarily entered the management arm of the program. The baseline characteristics of these patients in the management group are shown (Table 1).
Table 1: Baseline characteristics of 35 patients taking ACE inhibitor medication (number or percent for each selected parameter).

Patient management results
The renal and blood pressure status of these patients were followed for a mean of 63 ± 4.5 weeks. There were no significant adverse events reported or observed clinically in this group. At the completion of the study period, 39% of patients were taking 2 mg, 29% were taking 4 mg, 3% taking 6 mg and 29% taking 8 mg of ACEI, respectively. A total of 72% of clients were clinically assessed as compliant, 22% were considered compliant 65% of the time and 5% were non-compliant.
Over the 2 years of the management phase of the project, 111 urine ACR tests were performed by POCT for patient management (mean 3.2 tests per patient). There was no statistical difference in urine ACR levels in the study period (median [and inter-quartile range] 5.7 mg/mmol [1.2-15.2] pre-ACEI and 4.3 mg/mmol [1.3-16.7] post-ACEI treatment p = 0.50, Wilcoxon signed ranks test). Categorisation of patients into those exhibiting normoalbuminuria, microalbuminuria and macroalbuminuria (Table 2) revealed a trend towards improving albuminuria although these trends did not reach statistical significance due to small patient numbers (χ2 test; (χ2statistic = 0.496, degrees of freedom = 2, p = 0.780). One patient with initial microalbuminuria recorded a normal ACR post ACEI. The urine ACR levels in three diabetes patients with initial macroalbuminuria decreased by 27%, 64% and 78% respectively, with each patient falling within the microalbuminuria category post-ACEI treatment. The urine ACR in one patient with microalbuminuria deteriorated from 13.5 to 36.6 mg/mmol post ACEI. This patient was non-compliant in taking medication and was repeatedly hospitalised for other illnesses during the study period. Previous studies have shown that urine ACR levels deteriorate by approximately 15% per annum if antihypertensive treatment is not undertaken20. The calculated glomerular filtration rate of the group altered from 110 to 118 mL/min (p = 0.019, paired t-test). There was no change in the group's plasma potassium, urea and creatinine (Table 2). Collectively these results indicate a stabilisation in renal function among the management group.
Table 2: Albuminuria status and selected biochemical measures in patient group (n = 35) pre- and post-ACE inhibitor treatment
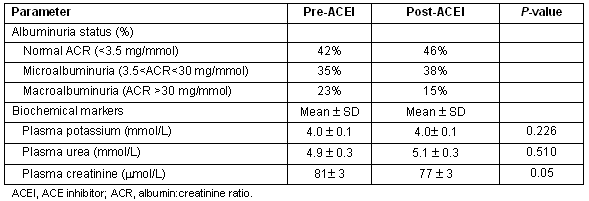
In the study period, there was a sustained and statistically significant reduction in mean blood pressure (both lying and standing) after ACEI intervention (Fig 1). Systolic blood pressure fell from 151±3 mmHg to 137±3 mmHg (lying) and from 147±3 mmHg to 131±3 mmHg (standing) (p <0.001, paired t-test). Diastolic blood pressure similarly fell from 92±2 mmHg to 84±2 mmHg (lying) and from 94±2 mmHg to 84±2 mmHg (standing) (p <0.001, paired t-test).
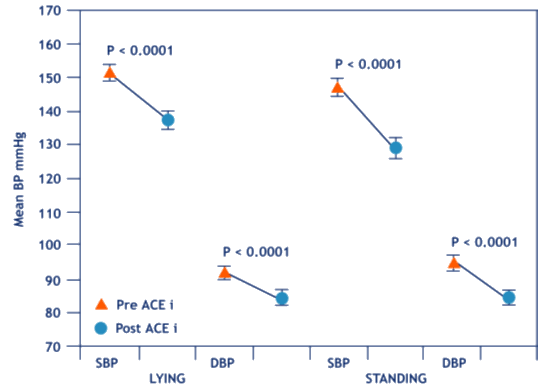
Figure 1: Improvement in mean blood pressure (BP; both lying and standing) in patient group (n = 35) following ACE inhibitor (ACE i) treatment. DBP, diastolic BP; SBP, systolic BP.
Quality testing
For quality control testing conducted during the management phase of the Umoona Kidney Project (n = 20), the DCA 2000 recorded an imprecision (coefficient of variation, CV%) of 8.3% and 3.9% for urine albumin (for quality control samples with concentrations of approximately 36 and 210 mg/L respectively), 5.5% and 3.9% for urine creatinine (9 and 35 mmol/L) and 7.4% and 3.0% for urine ACR (ratios of 4.0 and 6.7 mg/mmol). These levels of imprecision are consistent with that observed for quality control testing in the entire life of the Umoona Kidney Project15 and are well within national and international precision goals of 10%, 6% and 12% for urine albumin, creatinine and urine ACR respectively that have been derived from biological variation and other international consensus data on performance criteria21-23.
Community acceptance
Fifty community members completed the questionnaire on the Umoona Kidney Project, including 76% of the patients who participated in the program. The results obtained for selected questions relating to this article are shown (Table 3). Over two-thirds of the community were concerned about developing renal disease. Three-quarters felt there were no cultural barriers in providing a urine sample for ACR POCT.
Table 3: Summary of questionnaire responses (n = 50) to ascertain community attitudes towards the Umoona Kidney Project
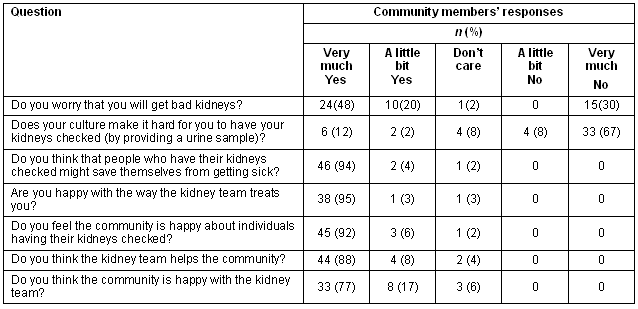
Of respondents, 98% understood the project's educational messages about how regular 'checks' for kidney disease could lessen their risk of developing advanced renal disease and they believed the community was comfortable with the concept of risk assessment. Over 95% were satisfied with the way the Flinders' renal team worked with them as individuals, while 94% agreed the renal team had helped the community overall in addressing renal disease.
In response to the general question Can you tell me why the kidney team comes here?, 63% of the 49 respondents ticked the following two responses Help people at risk of getting bad kidney disease to stay healthy and Find out who has bad kidney disease. One-third ticked the response Help the community to understand the benefits of treatment.
Discussion
The Umoona Kidney Project had many unique features which set it apart from other chronic disease programs for Aboriginal Australians. It adopted a strong family-orientated holistic approach (involving both adults and their children) and empowered and encouraged community ownership. Community members received on-site clinical advice (for both renal disease prevention and management) from a team of renal specialists who conducted 24 visits to the community over 3 years. While acknowledging this approach was intensive and difficult to sustain, it occurred in a time when the community did not have its own salaried medical officer. Importantly, POC pathology testing for urine ACR was used for the first time in an Aboriginal community setting in the Umoona Kidney Project. Umoona's Aboriginal health worker team received a continuous, structured education and training program on POCT delivered by the program manager. The health worker team were taught not only how to conduct urine ACR POCT on patients, but they also undertook their own quality testing procedures to assess the performance of their DCA 2000 analyser for urine ACR measurement. These processes provided a significant sense of empowerment for the health workers24. For Umoona's community members, the immediacy of the urine ACR POCT result meant they could see the renal specialist and have their treatment modified without the need to attend a follow-up visit to obtain their result. A further unique design aspect of this study was the specific requirement for patients to provide a first morning urine sample to test for ACR in preference to the more easily obtained random sample. The first morning urine is the recommended specimen of choice for the urine ACR test13. It provides the most accurate measure of urine ACR, as it is not subject to the wide diurnal biological variation exhibited by urine albumin and therefore has a much lower rate of false positive results than the random sample.
The value of POCT for community risk assessment has previously been described by our group15-16 but this article reports for the first time the practical and culturally appropriate use of urine ACR POCT on the DCA 2000 for the management of Aboriginal renal disease. The renal function of the patient group stabilised over the 2 year study period, as measured by POC urine ACR testing and other renal markers. The use of POC urine ACR testing was analytically sound and comparable to the performance expected of a pathology laboratory, while the use of POCT was culturally appropriate and widely accepted within the Aboriginal community setting. Community-based POCT not only raised the cultural awareness of renal disease, but also facilitated the development of a number of other community health promotion programs, particularly centred on nutrition, as previously reported by our group25.
The efficacy of the ACEI medication in improving renal and cardiovascular function in the Aboriginal health setting has also been confirmed in this study, and our results are consistent with other workers in this field26-27.
The high level of acceptance of the Umoona Kidney Project by the community was confirmed from the results of the community survey. As further testament to the success of the program, the inaugural director of the Umoona Tjutagku Health Service wrote the following comments in a letter to the program manager at the conclusion of her tenure as Director:
In my twenty years of working with Aboriginal people I have never seen such dedication, coupled with sensitivity in a group working with Aboriginal people. The [renal] team has built up a feeling of trust amongst community members and has made many friends. The team's willingness to listen and involve the community has provided a good model for future projects.
It should be acknowledged that this study was conducted in a single community with a relatively small samples size and was not a randomised controlled study. Nonetheless, in December 2000, the Umoona Kidney Project was handed over to the Umoona Community as a self-sustaining activity fully integrated into the health service infrastructure. Both the South Australian Government's Department of Human Services Renal and Urology Services Implementation Plan 2000-201128 and the state-wide Iga Warta Aboriginal Renal Disease Summit 1999 endorsed the Umoona model and recommended its expansion to other Aboriginal communities in rural and remote South Australia.
From this pioneering work, a national Australian Government funded POCT program called QAAMS (Quality Assurance for Aboriginal Medical Services) has been developed in which POCT for both HbA1c and urine ACR is used to assist the management of Aboriginal patients with diabetes and associated renal disease29-31. Sixty-five Aboriginal medical services encompassing every state and territory in Australia now participate in this unique POCT program. Both the HbA1c and urine ACR POC tests have their own Medicare rebate item number, which enables the costs of performing these tests to be fully refunded to the participating health services and ensures the program is cost-neutral in terms of on-going reagents needed to perform the POC tests and consumables.
Acknowledgements
The Umoona Kidney Project was funded by grants from the South Australian Health Commission (1998), the Office for Aboriginal and Torres Strait Islander Health (OATSIH) in the Commonwealth Department of Health and Aged Care (1999-2000), Servier Laboratories (Australia) Pty Ltd, the Australian Kidney Foundation and Janssen-Cilag Pty Ltd. Dean Whiting and Alison Halfnights from Bayer Australia are also thanked for their support of the Umoona Kidney Project. Our now retired clinical colleague, Dr Lindsay Barratt devoted significant time, commitment and energy to the Umoona Kidney Project. Our close friends and colleagues at the Women's and Children's Hospital (Dr Ken Juriedini, Dr Margie van Renen and Sandra Harris) are gratefully acknowledged for their work on the concurrent children's arm of the project. All members of the Umoona community are sincerely thanked for their participation and long-term support of the Umoona Kidney Project. Maryanne Hudson, Cissie Riessen, Michael Brown and Gaye McLeod worked as Aboriginal health workers during the program, along with Sisters Vicki McCormack and Chris Durden (clinical nurses at Umoona). Ian Crombie (former Chairman of the Board of the Umoona Tjutagku Health Service) and Waluwe Simpson-Lyttle and Janice Braun (former Directors of the Umoona Tjutagku Health Service) all had major input to the success of the program at the community level.
References
1. McDonald SP, Russ GR. Current incidence, treatment patterns and outcome of end-stage renal disease among indigenous groups in Australia and New Zealand. Nephrology 2003; 8: 42-48.
2. Cass A, Cunningham J, Wang Z, Hoy W. Regional variation in the incidence of end-stage renal disease in Indigenous Australians. Medical Journal of Australia 2002; 175: 24-27.
3. Cass A, Cunningham J, Snelling P, Wang Z, Hoy W. End-stage renal disease in Indigenous Australians: a disease of disadvantage. Ethnicity and Disease 2002; 12: 373-378.
4. Thomas MAB. Kidney disease in Australian Aboriginals: time for decisive action. Medical Journal of Australia 1998; 168: 532-533.
5. Hoy W. Renal disease in Australian Aboriginals. Medical Journal of Australia 1996; 165: 126-127.
6. Ravid M, Savin H, Jutrrin I, Bental T, Katz B, Lishner M. Long term stabilising effect of angiotensin converting enzyme inhibition on plasma creatinine and on proteinuria in normotensive type 2 diabetic patients. Annals of Internal Medicine 1993; 118: 577-581.
7. Giatras I, Lau J, Levey AS. Effect of angiotensin-converting enzyme inhibitors on the progression of nondiabetic renal disease: A meta-analysis of randomized trials. Angiotensin-Converting-Enzyme Inhibition and Progressive Renal Disease Study Group. Annals of Internal Medicine 1997; 127: 337- 345.
8. Maschio M, Marcantoni C. Angiotensin converting enzyme inhibitors in non-diabetic renal disease. Current Opinion in Nephrology and Hypertension 1998; 7: 253-257.
9. Ruggenenti P, Fassi A, Ilieva AP, Bruno S, Iliev IP, Brusegan V et al. Preventing microalbuminuria in type 2 diabetes. New England Journal of Medicine 2004; 351: 1941-1951.
10. Keane WF, Brenner BM, De Zeeuw D, Grunfeld JP. The risk of developing end-stage renal disease in patients with type 2 diabetes and nephropathy: The RENAAL Study. Kidney International 2003; 63: 1499-1507.
11. American Diabetes Association. Diabetic nephropathy. Diabetes Care 2002; 25(Suppl): S85-S89.
12. Jerums G. Microalbuminuria in diabetes. In: L Coulston (Ed.). Diabetes, glycation and complications. The clinical biochemist monograph. Perth, WA: Australasian Association of Clinical Biochemists, 1990; 55-59.
13. Mogensen C. Microalbuminuria. A marker for organ damage. London: Science Press, 1996.
14. Shephard MDS, Barratt L, Simpson-Lyttle W. Is the Bayer DCA 2000 acceptable as a screening instrument for the early detection of renal disease? Annals of Clinical Biochemistry 1999; 36: 393-394.
15. Shephard MDS, Allen G. Screening for renal disease in a remote Aboriginal community using the Bayer DCA 2000. Australian Journal of Medical Science 2001; 22: 164-170.
16. Shephard MDS, Allen GG, Barratt LJ, Paizis K, Brown M, Barbara JAJ et al. Albuminuria in a remote South Australian Aboriginal community: results of a community-based screening program for renal disease. Rural and Remote Health 3: 156. (Online) 2003: Available: http://rrh.org.au. (Accessed 7 April 2006).
17. Parsons MP, Newman DJ, Newall RG, Price CP. Validation of a point-of-care assay for the urinary albumin:creatinine ratio. Clinical Chemistry 1999; 45: 414-417.
18. Batterham M, Shephard MDS, Barratt LJ, Braun J. Local Aboriginal people's attitudes towards the Umoona Kidney Project at Coober Pedy. Proceedings: Australian and New Zealand Society of Nephrology: Annual Scientific Meeting; Melbourne, Australia; 15-17 March 2000: 18.
19. Estermann A. The Likert scale. Australian Epidemiologist 2003; 10: 46-48.
20. Hoy W, Kelly A, Jacups S, McKendry K, Baker P, Mcdonald S et al. Stemming the tide: reducing cardiovascular disease and renal failure in Australian Aborigines. Australian and New Zealand Journal of Medicine 1999; 29: 480-483.
21. Shephard MDS. Analytical performance criteria for point-of-care testing instruments. In: Standards for point of care testing in general practice. Incorporating POCT trial guidelines. Canberra: Australian Government Department of Health and Ageing, 2004; 101-140.
22. Sacks DB, Bruns DE, Goldstein DE, MacLaren NK, McDonald JM, Parrott M. Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Clinical Chemistry 2002; 48: 436-472.
23. Ricos C, Alvarez V, Cava F, Garcia-Lario JV, Hernandez A, Jiminez CV et al. Current databases on biological variation: pros, cons and progress. Scandinavian Journal of Clinical Laboratory Investigation 1999; 59: 491-500.
24. Shephard MDS, Brown M, Hudson M, Riessen C, Braun J. The Umoona Kidney Project. Aboriginal Health Worker Journal 2000; 24: 12-15.
25. Zeunert S, Cerro N, Boesch L, Duff M, Shephard MD, Jureidini KF et al. Nutrition Project in a remote Australian Aboriginal community. Journal of Renal Nutrition 2002; 12: 102-106.
26. Hoy W, Baker P, Kelly A and Wang Z. Reducing premature death and renal failure in Australian Aborigines: results of a community-based treatment program. Medical Journal of Australia 2000; 172: 473-478.
27. Hoy WE, Wang Z, Baker PRA and Kelly AM. Reduction in natural death and renal failure from a systematic screening and treatment program in an Australian Aboriginal community. Kidney International 2003; 63(Suppl 83): S66-S73.
28. Department of Human Services. Renal and urology services. Implementation plan 2000-2011. Adelaide: Government of South Australia, 2000.
29. Shephard MDS, Gill JP. An innovative Australian point-of-care model for urine albumin:creatinine ratio testing that supports diabetes management in indigenous medical services and has international application. Annals of Clinical Biochemistry 2005; 42: 208-215.
30. Shephard MDS, Gill J. Results of an innovative education, training and quality assurance program for point-of-care HbA1c testing using the Bayer DCA 2000 in Australian Aboriginal community controlled health services. Clinical Biochemist Reviews 2003; 24: 123-131.
31. Shephard M. Clinical and cultural effectiveness of the 'QAAMS' point-of-care testing model for diabetes management in Australian Aboriginal medical services. Clinical Biochemist Reviews 2006; 27: 161-170.

