An estimated 40 250 new cases of oral and pharyngeal cancer are expected to occur in the USA in 20121. Risk factors for oral and pharyngeal cancer include the use of smoked and smokeless tobacco products, betel quid chewing, alcohol consumption, human papillomavirus (HPV) infection, sunlight exposure and poor nutrition1-4. Five-year relative survival rates for these cancers range from 32% for distant cancers up to 83% for localized cancers5. Only 33% of oral and pharyngeal cancers, however, are diagnosed at the localized stage5. This remains below the Healthy People 2020 Objective of diagnosing 35.8% of these cancers at the localized stage6.
Dentists and primary care physicians can often detect premalignant abnormalities and early stage oral cancers through visual inspection (ie oral cancer screening)1, which is important given the higher survival rates for these early stage cancers5. There are currently no established guidelines for oral cancer screening, but the American Cancer Society recommends that oral cancer screening be included in adults' cancer-related check-ups7. The American Dental Association also recommends that clinicians remain alert for signs of potentially malignant lesions or early-stage cancers in all patients while conducting routine visual and tactile examinations8. Although oral cancer screening may help increase the proportion of oral cancers detected at an early stage, only 12-35% of adults in the USA report ever having had an oral cancer screening examination9-13. Most of these individuals report that dentists or dental hygienists had performed their examinations9,10.
The Appalachian region of the USA extends from New York state to Mississippi, consisting of more than 400 counties in 13 states14. A majority of Appalachian counties are non-metropolitan and just under half of Appalachian residents live in rural areas15. Appalachian residents tend to have a lower socioeconomic status and poorer health compared to the rest of the USA15,16. Similar to other cancer types16,17, Appalachia appears to suffer from an excessive burden of oral and pharyngeal cancer incidence and mortality. Many states in the Appalachian region have higher incidence (six states) or mortality (nine states) rates for oral and pharyngeal cancers compared to the national averages18. The elevated rates may be partly attributable to poorer health behaviors in these Appalachian states (eg smoking and use of smokeless tobacco16,19).
Ohio Appalachia is a 32 county region (29 at the time of this study) in the southern and eastern parts of the state, with demographic characteristics similar to Appalachia as a whole15. In terms of oral and pharyngeal cancer, data suggest the mortality rate for Ohio Appalachia may be higher than the national rate (2.9 vs 2.6 deaths per 100 000)18, and several counties have elevated incidence rates (11.3 to 16.5 cases per 100 000; national rate = 10.8 cases per 100 000)20. Risk factors for these cancers are very prevalent in Ohio Appalachia, with high rates of tobacco use and heavy alcohol use18.
Oral cancer screening may be particularly important to residents of Appalachia given the elevated oral and pharyngeal cancer mortality rates and high prevalence of risk factors for these cancers in this region. To our knowledge, however, only one study has reported such data, with 32% of non-Amish males and 22% of non-Amish females in Ohio Appalachia reporting a previous oral cancer screening examination21. Although this study provided initial insight into oral cancer screening in Appalachia, it included residents from only two Ohio Appalachian counties. The current study expands on these findings by examining oral cancer screening among women throughout the entire Ohio Appalachian region and identifying correlates of having received a previous screening examination. Women's recent dental care use was also examined since oral cancer screening examinations are most commonly performed by dentists.
Study design
The Community Awareness Resources Education (CARE) study was one of eight Centers for Population Health and Health Disparities (P50) funded by the National Institutes of Health (NIH) and has been described in detail in previous publications22,23. The overall goal of the CARE study was to address the high cervical cancer incidence and mortality rates in Ohio Appalachia. The first phase of the CARE study, completed in June 2006, collected cross-sectional survey data on women from participating health clinics to investigate the factors associated with obtaining regular Pap smears. A secondary aim of the surveys was to examine oral cancer screening and dental care use among these women, the focus of this article.
A total of 22 health clinics in Ohio Appalachia were approached to participate in the CARE study, of which 14 (64%) agreed. Within each participating clinic, a monthly random sample of women was selected and their medical records were reviewed to determine eligibility. Samples were selected from lists of all female patients obtained at regular intervals from each health clinic. Eligible women had to be at least 18 years of age, reside in an Ohio Appalachian county, not be pregnant, have no history of hysterectomy or invasive cervical cancer, and have been seen in a participating clinic within the prior two years. If a woman was eligible and agreed to participate, a meeting to complete the baseline survey was scheduled. The baseline survey included a face-to-face interview and a self-administered questionnaire.
A total of 571 women (cooperation rate = 71%) completed baseline surveys22. Data on oral cancer screening and dental care use were collected during the self-administered questionnaire, which 517 women completed. Data on 477 women who answered the item regarding oral cancer screening history are included in this article, excluding 40 women who did not provide data for this item. Women included in this article were similar to those not included in terms of demographic characteristics examined (all p>0.05).
Measures
Women were asked whether they had ever had an examination for oral cancer, with the examination described as one 'in which the doctor or dentist pulls on your tongue, sometimes with gauze wrapped around it, and feels under the tongue and inside the cheeks'. Women were classified as having had a previous oral cancer examination or not. Women who had had a previous examination were asked what type of healthcare provider performed their last oral cancer examination and the main reason for the examination.
Participants indicated the behaviors they thought were risk factors for oral cancer. Response options included: spending too much time in the sun (correct); drinking too much alcohol (correct); smoking cigarettes, cigars or a pipe (correct); use of chewing tobacco or snuff (correct); and drinking too much coffee (incorrect). Participants could select more than one response. Women were also asked to name one early sign of oral cancer, with response options of: white patches in mouth which are not painful; red patches in mouth which are not painful; sore or lesion in mouth which does not heal; sore or lesion in mouth; and bleeding in mouth. Women could indicate that they did not know an early sign of oral cancer.
Women's dental care use was examined by asking when their last dental visit occurred. Women whose last visit occurred within the last year were considered as having had recent dental care. Those women who had not had recent dental care were asked the main reason why they had not seen a dentist during the previous year. Participants also indicated the number of teeth they had had removed due to tooth decay, infection or gum disease. Items concerning oral cancer, oral cancer screening and dental care use were based on those from national surveys24,25.
Surveys assessed a wide range of demographic factors, health behaviors and psychosocial factors given the CARE study used the Social Determinants of Health Model as its theoretical framework (Tables 1,2)26. For this article, constructs from this model and other variables thought to be potentially important to oral cancer screening behaviors were examined. The socioeconomic status (SES) measure was loosely based on the Hollingshead scale and combined information on occupation, education and income27. The SES scores ranged from 0 to 6, with a score of 4 or higher indicating high SES22. Appalachian self-identity was measured with the item, 'Do you consider yourself to be Appalachian?'28. Given HPV infection is a risk factor for oral and pharyngeal cancers, women were asked to indicate if they had ever been told by a doctor that they had an HPV infection (described to participants as venereal warts, condylomas or papillomavirus infections)2.
Existing instruments were used to measure depression (Center for Epidemiologic Studies Depression [CES-D] scale29), anxiety (Beck Anxiety Inventory [BAI]30), perceived stress (Perceived Stress Scale [PSS]31), discrimination (Detroit Area Study Discrimination Questionnaire [DAS-DQ]32), trust in physician (Trust in Physician Scale [TPS]33), major life events (Life Stressor Checklist-Revised [LSC-R] scale34) and locus of health control (God Locus of Health Control [GLHC] measure35).
Table 1: Categorical correlates of ever having had an oral cancer screening examination (n=477)
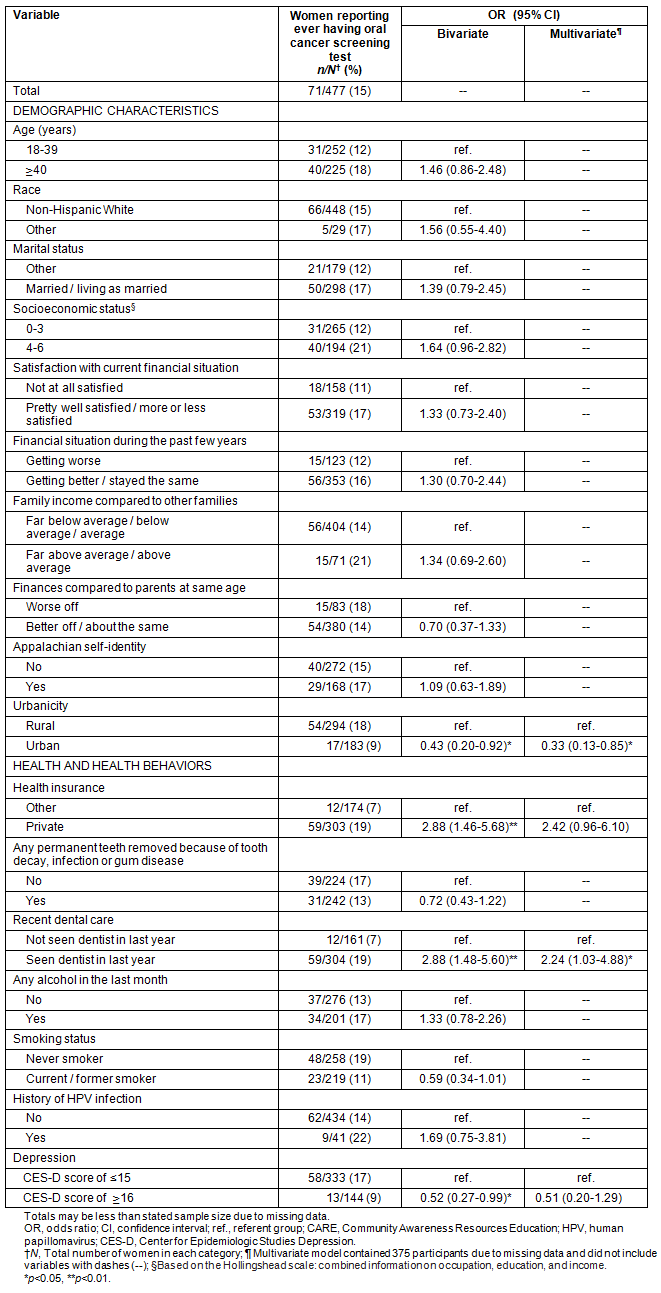
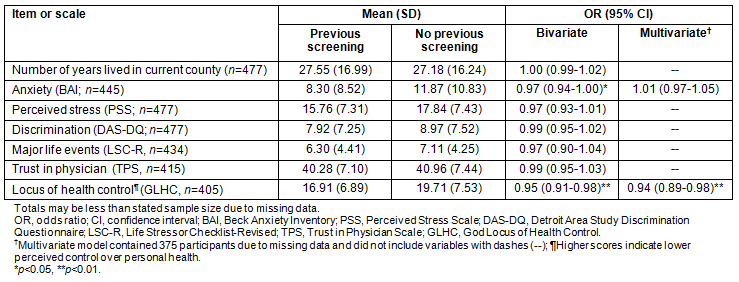
Table 2: Continuous correlates of ever having had an oral cancer screening examination (n=477)
Data analysis
The primary outcome was participants' report of having ever received an oral cancer screening examination. Because most people report receiving oral cancer examinations from dentists or dental hygienists, recent dental care was examined as a secondary outcome10. For each outcome, logistic regression was used to identify bivariate correlates. Statistically significant bivariate correlates (p<0.05) were then entered into a multivariate logistic regression model, from which adjusted odds ratios (OR) and 95% confidence intervals (CI) were obtained. All logistic regression models included health clinic as a random effect. Data were analyzed using SAS v9.2 (SAS; Cary, NC, USA) and all statistical tests were two-tailed, using a critical value of α=0.05.
Ethics approval
The institutional review boards at The Ohio State University, the University of Michigan and the Centers for Disease Control and Prevention (CDC) approved this study (#2004C0104).
Participant characteristics
Most women were non-Hispanic white (94%), married or living as married (62%), low SES (58%), from a rural area (62%), and had private health insurance (64%). About half of the women were aged 40 or older (47%), current or former smokers (46%), and had consumed alcohol within the last month (42%). About one-third of women identified themselves as Appalachian (38%), and few (9%) reported ever having been told they had an HPV infection.
Oral cancer knowledge and screening behaviors
Most women indicated that use of chewing tobacco or snuff (90%) or smoking cigarettes, cigars or pipes (80%) increases a person's chances of getting oral cancer. Fewer indicated too much time in the sun (18%) or excessive alcohol intake (9%) as risk factors. About 3% of women incorrectly identified excessive coffee intake as an oral cancer risk factor. Almost half of women (43%) did not know an early sign of oral cancer, with fewer indicating a sore or lesion in the mouth that does not heal (29%), white patches in the mouth that are not painful (11%), a sore or lesion in the mouth (8%), red patches in the mouth that are not painful (3%) or bleeding in the mouth (3%).
Only 15% (71/477) of women reported ever having had an oral cancer screening examination (Table 1). Among those who had had a previous examination, most reported receiving their most recent examination from a dentist (80%), with fewer reporting a dental hygienist (11%) or physician (7%). Almost all women who had had a previous examination indicated their most recent examination was part of a routine dental examination (83%) or routine physical examination (13%). In bivariate analyses, women with private health insurance or who had received recent dental care were more likely to have reported having ever had an oral cancer screening examination (both p<0.01) (Table 1). Oral cancer screening was less common among women from urban areas, with depressive symptoms, greater anxiety or perceived a lower locus of control over their health (all p<0.05) (Tables 1,2).
In multivariate analyses, women were more likely to have reported having ever had an oral cancer screening examination if they had had recent dental care (OR = 2.24, 95% CI: 1.03-4.88) (Table 1). Women were less likely to have reported a previous oral cancer screening examination if they were from urban areas (OR = 0.33, 95% CI: 0.13-0.85) or perceived lower locus of control over their health (OR = 0.94, 95% CI: 0.89-0.98) (Tables 1,2).
Dental care use
About two-thirds (65%) of women indicated they had had recent dental care (Table 3). Among women who had not had recent dental care, 40% indicated their last examination was 1-2 years ago, 37% indicated 2-5 years ago, 22% indicated more than 5 years ago and 1% indicated they had never been to a dentist. The most common reasons for not seeing a dentist in the last year were cost (50%); not having a reason to go to the dentist (17%); fear, worry or dislike of going to the dentist (11%); and having other priorities (5%). All other responses were given by less than 5% of women.
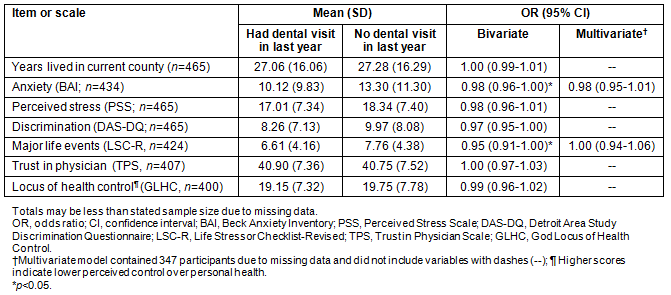
In bivariate analyses, women were more likely to report recent dental care if they were married or living as married; had a high SES; were pretty well or more or less satisfied with their current finances; had finances that had been getting better or staying about the same during the past few years; were financially better off or about the same compared to their parents at the same age; had private health insurance; or had consumed any alcohol in the last month (all p<0.05) (Table 3). Recent dental care was less common among women who identified themselves as Appalachian, had greater anxiety or reported more major life events (all p<0.05) (Tables 3,4).
In multivariate analyses, women were more likely to have reported recent dental care if they were of a high SES (OR = 2.83, 95% CI: 1.58-5.06), had private health insurance (OR = 2.20, 95% CI: 1.21-3.97) or had consumed any alcohol in the last month (OR = 2.03, 95% CI: 1.20-3.42) (Table 3).
Table 3: Categorical correlates of recent dental care (n=465)
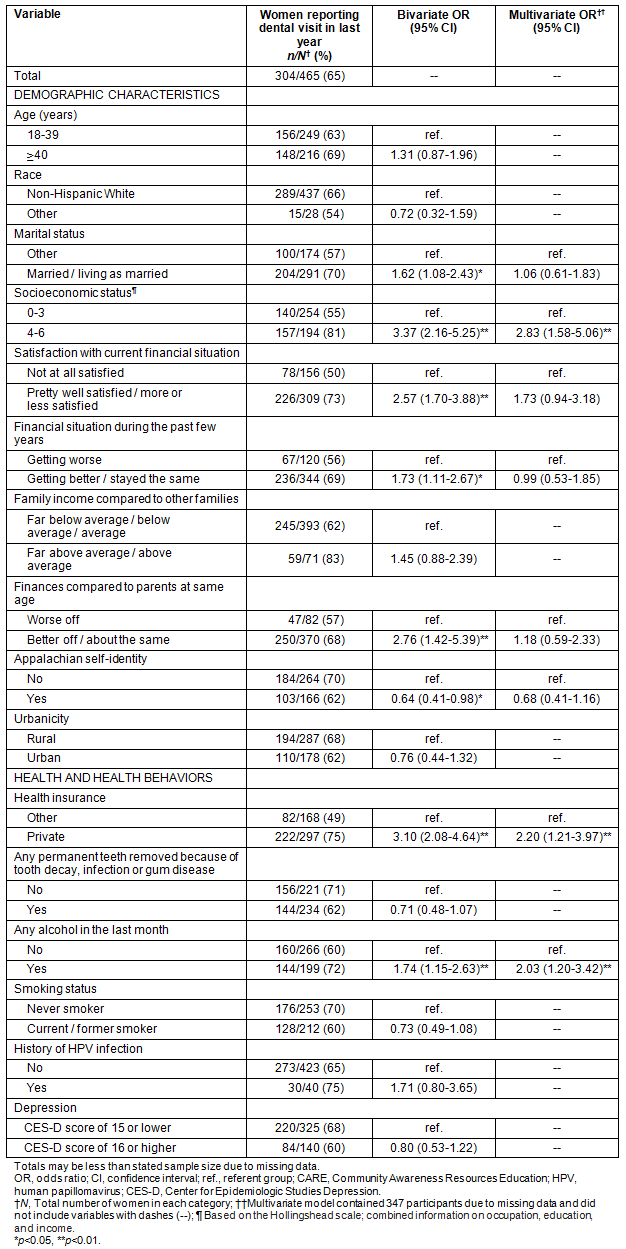
Table 4: Continuous correlates of recent dental care (n=465)
Discussion
Oral cancer screening can often help detect premalignant abnormalities and early stage oral cancers1, which have noticeably higher survival rates compared to late stage cancers5. Oral cancer screening may be particularly important in Appalachia, a region with higher oral and pharyngeal cancer mortality rates and increased prevalence of risk factors for these cancers16,18. Limited research, however, has examined oral cancer screening in Appalachia21. The current study assessed the oral cancer screening behaviors of women throughout the entire Ohio Appalachian region, as well as their recent dental care use since oral cancer screening examinations are most commonly performed by dentists9,10.
Only 15% of women from Ohio Appalachia reported ever having had an oral cancer screening examination. This is similar to previous estimates among adults from the USA (12-15%), suggesting that oral cancer screening is not commonly performed throughout the USA, including Appalachia9,11,12. Dentists appear to play a key role in the limited amount of oral cancer screening that is occurring in Ohio Appalachia. Dentists were not only the most common source of women's most recent examinations, but women were more likely to report having ever had an oral cancer screening examination if they indicated recent dental care. These findings support those from past research, in which most individuals (approximately 70%) with a prior oral cancer screening examination received the examination from a dental care provider9,10 . It is worth noting, however, that only 19% of women in our study who had had recent dental care reported ever having an oral cancer screening examination. This suggests that many missed opportunities for oral cancer screening examinations are occurring during dental visits. Some of these missed opportunities may be due to dental providers' lack of knowledge about oral cancer and oral cancer screening36,37. Education programs about the importance of opportunistic oral cancer screening examinations during routine visits may therefore be beneficial for dental care providers and help to improve screening in Appalachia.
Although our results indicate that dental care providers are performing most of the oral cancer screening examinations in Ohio Appalachia, dental care providers may not be able to reach all residents of this region. Many parts of Ohio Appalachia lack dental care providers38, and approximately 35% of women in this study reported not having had a dental visit in the last year (similar to past estimates for non-Hispanic whites39). Women of low SES and those without private health insurance were less likely to report recent dental care. Thus, oral cancer screening by dental care providers may not be a feasible option for adults who lack the access or resources to get dental care. For these individuals, physicians may be an option for screening, as adults are comfortable in discussing oral cancer with their regular physicians40. Physicians and other healthcare providers in this region therefore also need to be knowledgeable and comfortable in providing oral cancer screening examinations.
Women were less likely to report a previous screening examination if they perceived lower locus of control over their health or were from urban areas. The correlation with locus of health control may be partly attributable to fatalism, a value traditionally associated with the Appalachian region. Fatalism is the belief that events are determined by fate and out of human control, and there is some evidence suggesting fatalism influences the health behaviors of Appalachian residents41,42. Thus, it may be important for healthcare providers in Appalachia to stress to their patients that oral cancer screening examinations can in fact often help detect early stage oral cancers1,5. It is not entirely clear why oral cancer screening examinations were less common among women living in urban areas. Within Appalachia, many people are migrating from rural to urban areas, and it is possible that individuals new to urban areas may have been unsure of where to seek medical care43. It is also possible that insurance coverage played a role, as fewer women living in urban areas in our study had private health insurance compared to those from rural areas (53% vs 70%). Future research is needed to further explore these potential differences between urban and rural residents within Appalachia.
Interestingly, use of alcohol or tobacco and history of HPV infection were not correlated with oral cancer screening, even though all are risk factors for oral and pharyngeal cancer1-4. Findings regarding alcohol and tobacco use are consistent with past research where neither was associated with oral cancer screening in multivariate analyses10. Our results further suggest that healthcare providers are often not screening individuals who are at increased risk for oral and pharyngeal cancer. The suggested education programs for dental and other healthcare providers should include information about high-risk populations (eg smokers) and how opportunistic oral cancer screening can benefit their patients.
Our study had several strengths including participants being from a region with high mortality rates for oral and pharyngeal cancer and the examination of a wide range of potential correlates for both oral cancer screening and recent dental care. Limitations include unknown generalizability of findings since all participants were women recruited from health clinics in Ohio Appalachia and most were non-Hispanic white. A total of eight health clinics chose not to participate in the CARE study, though over 60% did agree to participate. Self-reported data were used for history of oral cancer screening examinations and recent dental care use, which may have been subject to recall and social desirability error. Individuals more commonly over-report health behaviors (ie cancer screening behaviors), suggesting actual oral cancer screening and dental care use in Ohio Appalachia may be lower than our estimates44. A description of oral cancer screening examinations was, however, provided to all women to help improve recall. There may also be some variables important to these health behaviors that were not measured in our study.
Oral cancer screening was not common among adult females from Ohio Appalachia. Although most women reported receiving their most recent examination from a dentist, there appears to have been many missed opportunities occurring at dental visits. Education programs about the importance of opportunistic oral cancer screening examinations during routine visits may be beneficial for dentists, as well as other healthcare providers who may have contact with women who do not receive regular dental care. Future research is needed to examine the acceptability of such education programs to healthcare providers in the Appalachian region.
Acknowledgments
The authors acknowledge support from the following grants: P50 CA105632 (EDP); P30 CA016058 (Behavioral Measurement Shared Resource at The Ohio State University Comprehensive Cancer Center); and K23 DE016890 (AGW).
References
1. American Cancer Society. Cancer facts & figures 2012. Atlanta, GA: American Cancer Society, 2012.
2. Termine N, Panzarella V, Falaschini S, Russo A, Matranga D, Lo Muzio L et al. HPV in oral squamous cell carcinoma vs head and neck squamous cell carcinoma biopsies: a meta-analysis (1988-2007). Annals of Oncology 2008; 19(10): 1681-1690.
3. Petti S. Lifestyle risk factors for oral cancer. Oral Oncology 2009; 45(4-5): 340-350.
4. Warnakulasuriya S. Global epidemiology of oral and oropharyngeal cancer. Oral Oncology 2009; 45(4-5): 309-316.
5. Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA: A Cancer Journal for Clinicians 2011; 61(4): 212-236.
6. U.S. Department of Health and Human Services. Healthy People 2020. (Online) 2011. Available: http://www.healthypeople.gov/2020/default.aspx (Accessed 17 November 2012).
7. American Cancer Society. American Cancer Society guidelines for the early detection of cancer. (Online) 2011. Available: http://www.cancer.org/docroot/PED/content/PED_2_3X_ACS_Cancer_Detection_Guidelines_36.asp?sitearea=PED (Accessed 17 November 2012).
8. Rethman MP, Carpenter W, Cohen EE, Epstein J, Evans CA, Flaitz CM et al. Evidence-based clinical recommendations regarding screening for oral squamous cell carcinomas. Journal of the American Dental Association 2010; 141(5): 509-520.
9. Centers for Disease Control and Prevention. Current trends examinations for oral cancer - United States, 1992. MMWR Morbidity and Mortality Weekly Report 1994; 43(11): 198-200.
10. Oh J, Kumar J, Cruz G. Racial and ethnic disparity in oral cancer awareness and examination: 2003 New York state BRFSS. Journal of Public Health Dentistry 2008; 68(1): 30-38.
11. Cruz GD, Le Geros RZ, Ostroff JS, Hay JL, Kenigsberg H, Franklin DM. Oral cancer knowledge, risk factors and characteristics of subjects in a large oral cancer screening program. Journal of the American Dental Association 2002; 133(8): 1064-1071; quiz 1094.
12. Horowitz AM, Nourjah PA. Factors associated with having oral cancer examinations among US adults 40 years of age or older. Journal of Public Health Dentistry 1996; 56(6): 331-335.
13. Patton LL, Agans R, Elter JR, Southerland JH, Strauss RP, Kalsbeek WD. Oral cancer knowledge and examination experiences among North Carolina adults. Journal of Public Health Dentistry 2004; 64(3): 173-180.
14. Appalachian Regional Commission. The Appalachian region. (Online) no date. Available: http://www.arc.gov/appalachian_region/TheAppalachianRegion.asp (Accessed 17 November 2012).
15. Pollard KM. Appalachia at the millennium: an overview of results from Census 2000. (Online) 2003. Available: http://www.arc.gov/research/researchreportdetails.asp?REPORT_ID=42 (Accessed 17 November 2012).
16. Halverson JA. An analysis of disparities in health status and access to health care in the Appalachian region. (Online) 2004. Available: http://www.arc.gov/research/researchreportdetails.asp?REPORT_ID=82 (Accessed 17 November 2012).
17. Wingo PA, Tucker TC, Jamison PM, Martin H, McLaughlin C, Bayakly R et al. Cancer in Appalachia, 2001-2003. Cancer 2008; 112(1): 181-192.
18. Casto BC, Sharma S, Fisher JL, Knobloch TJ, Agrawal A, Weghorst CM. Oral cancer in Appalachia. Journal of Health Care for the Poor and Underserved 2009; 20(1): 274-285.
19. Centers for Disease Control and Prevention. State-specific prevalence of cigarette smoking and smokeless tobacco use among adults - United States, 2009. MMWR Morbidity and Mortality Weekly Report 2010; 59(43): 1400-1406.
20. National Cancer Institute. State cancer profiles. (Online) no date. Available: http://statecancerprofiles.cancer.gov/cgi-bin/quickprofiles/profile.pl?39&003 (Accessed 17 November 2012).
21. Katz ML, Ferketich AK, Paskett ED, Harley A, Reiter PL, Lemeshow S et al. Cancer screening practices among Amish and non-Amish adults living in Ohio Appalachia. Journal of Rural Health 2011; 27(3): 302-309.
22. Paskett ED, McLaughlin JM, Reiter PL, Lehman AM, Rhoda DA, Katz ML et al. Psychosocial predictors of adherence to risk-appropriate cervical cancer screening guidelines: a cross sectional study of women in Ohio Appalachia participating in the Community Awareness Resources and Education (CARE) project. Preventive Medicine 2010; 50(1-2): 74-80.
23. Paskett ED, McLaughlin JM, Lehman AM, Katz ML, Tatum CM, Oliveri JM. Evaluating the efficacy of lay health advisors for increasing risk-appropriate Pap test screening: a randomized controlled trial among Ohio Appalachian women. Cancer Epidemiology, Biomarkers & Prevention 2011; 20(5): 835-843.
24. Centers for Disease Control and Prevention. National Health Interview Survey. (Online) 2011. Available: http://www.cdc.gov/nchs/nhis.htm (Accessed 17 November 2012).
25. Centers for Disease Control and Prevention. Behavioral Risk Factor Surveillance System. (Online) 2011. Available: http://www.cdc.gov/brfss (Accessed 17 November 2012).
26. Marmot M, Wilkinson R. Social determinants of health. Oxford: Oxford Press, 1999.
27. Hollingshead A. A four-factor index of social status. New Haven, CT: Department of Sociology, Yale University, 1975.
28. Reiter PL, Katz ML, Ferketich AF, Ruffin MT, Paskett ED. Appalachian self-identity among women in Ohio Appalachia. Journal of Rural Community Psychology E12(1). (Online) 2009. Available: http://www.marshall.edu/jrcp/VE12%20N1/Reiter%20JRCP.pdf (Accessed 17 November 2012).
29. Radloff LS. The CES-D scale: a self-report depression scale for research in the general population. Applied Psychological Measurement 1977; 1: 385-401.
30. Beck AT, Steer RA. Beck Anxiety Inventory manual. San Antonio, TX: Psychological Corporation, 1990.
31. Cohen S, Kamarck T, Mermelstein R. A global measure of perceived stress. Journal of Health and Social Behavior 1983; 24(4): 385-396.
32. Taylor TR, Kamarck TW, Shiffman S. Validation of the Detroit Area Study Discrimination Scale in a community sample of older African American adults: the Pittsburgh Healthy Heart Project. International Journal of Behavioral Medicine 2004; 11(2): 88-94.
33. Anderson LA, Dedrick RF. Development of the Trust in Physician scale: a measure to assess interpersonal trust in patient-physician relationships. Psychological Reports 1990; 67(3 Part 2): 1091-1100.
34. Wolfe J, Kimerling R. Gender issues in the assessment of posttraumatic stress disorder. In: Assessing psychological trauma and PTSD. New York: Guilford Press, 1997.
35. Wallston KA, Malcarne VL, Flores L, Hansdottir I, Smith CA, Stein MJ et al. Does God determine your health? The God Locus of Health Control scale. Cognitive Therapy and Research 1999; 23(2): 131-142.
36. Yellowitz JA, Horowitz AM, Drury TF, Goodman HS. Survey of U.S. dentists' knowledge and opinions about oral pharyngeal cancer. Journal of the American Dental Association 2000; 131(5): 653-661.
37. Patton LL, Ashe TE, Elter JR, Southerland JH, Strauss RP. Adequacy of training in oral cancer prevention and screening as self-assessed by physicians, nurse practitioners, and dental health professionals. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, Endodontology 2006; 102(6): 758-764.
38. Horner MW, Mascarenhas AK. Analyzing location-based accessibility to dental services: an Ohio case study. Journal of Public Health Dentistry 2007; 67(2): 113-118.
39. Doty HE, Weech-Maldonado R. Racial/ethnic disparities in adult preventive dental care use. Journal of Health Care for the Poor and Underserved 2003; 14(4): 516-534.
40. Horowitz AM, Canto MT, Child WL. Maryland adults' perspectives on oral cancer prevention and early detection. Journal of the American Dental Association 2002; 133(8): 1058-1063.
41. Walker R, Lucas W, Crespo R. The West Virginia Rural Cancer Prevention project. Cancer Practice 1994; 2(6): 421-426.
42. Deskins S, Harris CV, Bradlyn AS, Cottrell L, Coffman JW, Olexa J et al. Preventive care in Appalachia: use of the Theory of Planned Behavior to identify barriers to participation in cholesterol screenings among West Virginians. Journal of Rural Health 2006; 22(4): 367-374.
43. Lichter DT, Garratt J, Marshall ML, Cardella M. Emerging patterns of population redistribution and migration in Appalachia. (Online) 2005. Available: http://www.arc.gov/research/researchreportdetails.asp?REPORT_ID=26 (Accessed 17 November 2012).
44. Rauscher GH, Johnson TP, Cho YI, Walk JA. Accuracy of self-reported cancer-screening histories: a meta-analysis. Cancer Epidemiology, Biomarkers and Prevention 2008; 17(4): 748-757.
