This clinical case report from rural Brazil documents a severe case of oral myiasis in a 52 year old male. This rare infection resulted from a combination of factors that made the patient susceptible. In Brazil, there are great differences in income, education, and access to health care between Brazil's rural and urban populations, where the per capita income of those in rural areas is half that of urban dwellers. In Brazil 8% (rural) and 24% (urban) of adults between the ages of 40 and 60 years have at least 11 years of formal education, and 14% (rural) and 36% (urban) of the population has private health insurance1. The term 'structural violence' has been used by medical anthropologists2-4 to show how social organization, political economy, and biology can interact to cause and perpetuate disparities in access, development and economic status which foster the spread of disease. Structural violence affects populations differently so it is essential to recognize where macro-structural forces manifest in the suffering of specific populations. This case study demonstrates the way biological, ecological, social, and economic factors related to rural residence collectively resulted in this uncommon case of oral myiasis.
The rural resident in this case report was eventually treated through the public healthcare system in Montes Claros, Minas Gerais, Brazil. Montes Claros is the center of a regional network of the national public healthcare system. Montes Claros and the Northern Minas Gerais region are located on a frontier between the more developed south-east and the less developed north-east of the country. Politically, Northern Minas Gerais is part of the State of Minas Gerais in Brazil's south-east region. However, because Northern Minas Gerais is included in an ecological region called 'the polygon of drought', it has been included in developmental stimulus programs administered by the Superintendent for the Development of the North East (SUDENE).
Myiasis
Myiasis is a parasitic infection that affects the organs and tissue of humans and livestock when fly larvae invade live or necrotic tissue5,6. The incidence of myiasis is higher in rural areas (where the fly is more common), and most commonly occurs in domestic animals and house pets. It has been linked to low socioeconomic status and overall vulnerability to disease in humans, with those affected likely to be very young or very old, sick or debilitated, and have very poor personal hygiene. Alcoholism, homelessness, low socioeconomic status, and predisposing pathologies such as epilepsy and epidermal carcinoma have all been associated with the disease, which occurs primarily in the tropical regions of underdeveloped countries5-8.
A diagnosis of myiasis is based on a clinical evaluation and confirmed by laboratory tests that identify the larvae. Treatment consists of total removal of the larvae and the administration of oral antiparasitic agents such as ivermectin9-11.
The larvae that produce myiasis can be obligatory parasites (eg Dermatobia hominis and Cochliomyia hominivorax spp.) whose development occurs only in a healthy host tissue. They can also be facultative parasites (eg Musca domestica, Cochliomyia macellaria) where normal larvae development occurs in decomposed organic matter12-15.
A myiasis infection can be a very serious condition, for instance C. hominivorax can deposit 400 eggs on an open wound5. Necrobiophagous flies (which feed on necrotic tissue) produce secondary myiasis. This is the most common type found in humans and usually affects patients with lesions in necrotic cavities. Myiasis can also be classified according to the site of infestation: cutaneous or tegumentary, cavitated or deep, and visceral; the first two of these are most likely to be implicated in maxillofacial infections5,9,13,15-18.
Oral myiasis cases have been reported most frequently in epileptic patients with lacerated lips after a convulsion, in children with lip alterations or a thumb sucking habit, in patients with advanced periodontal disease, at dental extractions sites, in patients with neoplasia of the oral mucosa, and in patients with a predisposition to maintaining an open mouth. The larvae can lodge in the palate or the gingival tissue where it produces ulcerative lesions that may go unnoticed or produce inflammation10,12,16,19,20.
Myiasis is usually treated with debridement or scission, which consists of manual removal, sometimes with a topical asphyxiating medication that forces the larvae to emerge. The most efficient and safest systemic pharmacologic therapy in humans is ivermectin, which acts by disabing the parasite's nervous system9,11,13-18. Recommended standard treatments also include the use of profuse and deep irrigation with home disinfectants, other antibiotics or larvicides and antiparasitics to ensure complete elimination of the infectious agents. The risk of poisoning or cancer associated with the use of creolina (readily available in Brazil) as an irrigating agent prevents its use as the treatment of choice14,17,21-26.
Case report
A 52 year old, dark-skinned male weighing 45 kg was admitted to a hospital in Montes Claros, Minas Gerais, Brazil via the emergency room. Montes Claros is a mid-sized city in the interior of Brazil that serves as the center for medical treatment to a large surrounding rural population. The patient lived in a rural area and had been admitted to the same hospital with a diagnosis of Alzheimer's disease 5 years earlier. He had not received specialized medical treatment for Alzheimer's since that time. Patients who suffer from Alzheimer's disease are generally dependent on assistance from family members to maintain their overall health, including oral hygiene27.
The emergency room diagnosis was a severe case of myiasis infestation. Clinical examination revealed edema of the upper lip with a small opening (required for the larvae to breathe). The patient reported the sensation of things moving inside the swollen area. After local anesthesia at the site of infection, an incision was made in the upper lip (Fig1) and the larvae were removed surgically (Fig2). Two 6 mg ivermectin tablets were prescribed. Following the procedure the patient was hospitalized for medical care to address his underlying poor health (primarily severe under-nutrition). The larvae were subsequently submitted for parasitology testing which reported them to be D. hominis.
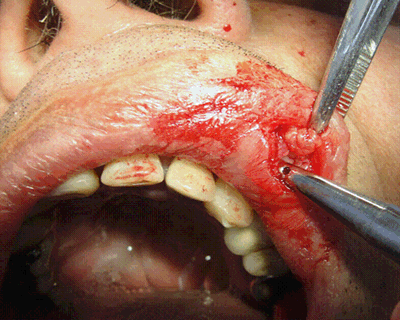
Figure 1: Surgical removal of larvae from upper lip.

Figure 2: Larvae were removed.
It is important to detect this condition in its early stages7,8; however, this was not possible for our patient. In Brazil, treatment to arrest the progression of Alzheimer's disease is not readily accessible in rural areas, and it is difficult to recruit public healthcare physicians to rural posts28. Even with higher remuneration than for urban positions it is difficult to keep rural health posts staffed, and in order to see medical specialists such as neurologists, rural residents must be transported to urban centers. However, such transport is often limited due to rural socioeconomic constraints. Access to education and health knowledge are limited in rural areas, and the self-care limitations of Alzheimer's disease also significantly contributed to the vulnerability of this patient. In this case report, the sum of a number of patient factors led to a unique case of myiasis in the upper lip9-11.
Conclusions
The nutritional, physical and mental health conditions in rural regions of Latin American countries such as Brazil predispose the population to pathologies not normally observed elsewhere14,23. In such a situation the risk factors for disease can increase exponentially1-4. Myiasis is an example of this mechanism, for it is associated with extreme poverty or cases of incapacitating mental conditions where self-care is limited, making individuals vulnerable to a disease very uncommon in human populations18,21,25,26. This disease is more prevalent in rural populations, demonstrating that structural violence contributes to the financial and social exclusion of such populations. When risk factors combine to produce unfavorable results5,6, one of these factors in Brazil is living in a rural or remote location.
References
1. Kassouf AL. Acesso aos serviços de saúde nas áreas urbana e rural do Brasil. Revista Brasileira de Economia e Sociologia Rural 2005; 43(1): 29-44.
2. Farmer P. An Anthropology of Structural Violence. Current Anthropology 2004; 45(3): 305-324.
3. Lockhart C. The Life and Death of a Street Boy in East Africa. Medical Anthropology Quarterly 2008; 22(1): 94-115.
4. Roberts JE, Jones KM, Espírito Santos LR, Roque MJG, Silveira MF et al. Expanding Access to HIV Testing in Northern Minas Gerais, Brazil. Southern Anthropologist 2009; 34(1&2): 54-70.
5. Sharma J, Mamatha GP, Acharya R. Primary oral myiasis: a case report. Medicina Oral, Patología Oral y Cirugía Bucal 2008; 13(11): E714-E716.
6. Hopkins R. Re: Oral myiasis. British Journal of Oral and Maxillofacial Surgery 2003; 41(6): 422.
7. Droma EB, Wilamowski A, Schnur H, Yarom N, Scheuer E, Schwartz E. Oral myiasis: a case report and literature review. Oral surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontics 2007; 103(1): 92-96.
8. Abdo EN, Sette-Dias AC, Comunian CR, Dutra CE, Aguiar EG. Oral myiasis: a case report. Medicina Oral, Patología Oral y Cirugía Bucal 2006; 11(2): E130-E131.
9. Marquez AT, Mattos Mda S, Nascimento SB. [Myiasis associated with some socioeconomic factors in five urban areas of the State of Rio de Janeiro]. Revista da Sociedade Brasileira de Medicina Tropical 2007; 40(2): 175-180.
10. Faber TE, Hendrikx WM. Oral myiasis in a child by the reindeer warble fly larvae Hypoderma tarandi. Medical and Veterinary Entomology 2006; 20(3): 345-346.
11. Carvalho RW, Santos TS, Antunes AA, Laureano Filho JR, Anjos ED, Catunda RB. Oral and maxillofacial myiasis associated with epidermoid carcinoma: a case report. Journal of Oral Science 2008; 50(1): 103-105.
12. Aguiar AM, Enwonwu CO, Pires FR. Noma (Cancrum oris) associated with oral myiasis in an adult. Oral Disease 2003; 9(3): 158-159.
13. De Araujo RJ, Correa AM, Santos WR, Moreira MT, Jr. Advanced stage of oral myiasis in children: a clinical case report. Quintessence International 2008; 39(1): 39-43.
14. Clyti E, Pradinaud R. [Labial myiasis]. Annales de dermatologie et de venereologie 2008; 135(5): 435-436.
15. Rossi-Schneider T, Cherubini K, Yurgel LS, Salum F, Figueiredo MA. Oral myiasis: a case report. Journal of Oral Science 2007; 49(1): 85-88.
16. Kamboj M, Mahajan S, Boaz K. Oral myiasis misinterpreted as salivary gland adenoma. Journal of Clinical Pathology 2007; 60(7): 848.
17. Gealh WC, Ferreira GM, Farah GJ, Teodoro U, Camarini ET. Treatment of oral myiasis caused by Cochliomyia hominivorax: two cases treated with ivermectin. British Journal of Oral and Maxillofacial Surgery 2009; 47(1): 23-26.
18. Costa DC, Pierre-Filho Pde T, Medina FM, Mota RG, Carrera CR. Use of oral ivermectin in a patient with destructive rhino-orbital myiasis. Eye (London) 2005; 19(9): 1018-1020.
19. Shinohara EH, Martini MZ, de Oliveira Neto HG, Takahashi A. Oral myiasis treated with ivermectin: case report. Brazilian Dental Journal 2004; 15(1): 79-81.
20. Shinohara EH. Re: treatment of oral myiasis with ivermectin. British Journal of Oral and Maxillofacial Surgery 2003; 41(6): 421.
21. Fernandes LF, Pimenta FC, Fernandes FF. First report of human myiasis in GoiaS state, Brazil: frequency of different types of myiasis, their various etiological agents, and associated factors. Journal of Parasitology 2009; 95(1): 32-38.
22. De Souza Barbosa T, Salvitti Sa Rocha RA, Guirado CG, Rocha FJ, Duarte Gaviao MB. Oral infection by Diptera larvaee in children: a case report. International Journal of Dermatology 2008; 47(7): 696-699.
23. Sharma A, Hedge A. Primary oral myiasis due to Chrysomya bezziana treated with Ivermectin. A case report. Journal of Clinical Pediatric Dentistry 2010; 34(3): 259-261.
24. Visser MS, Yoon S, Marley SE. Efficacy of a combination ivermectin/praziquantel paste against nematodes, cestodes and bots in naturally infected ponies. Veterinary Record 2007; 161(21): 722-724.
25. Philip J, Matthews R, Scipio JE. Larvaee in the mouth. British Dental Journal 2007; 203(4): 174-175.
26. Sloop GD, Lopez FA. Clinical case of the month. Furuncular myiasis. Journal of the Louisiana State Medical Society 2006; 158(1): 14-15.
27. Freitas DA, Pereira MM, Moreira G, Mercado LF, Caballero. Cáncer de lengua en un paciente con Alzheimer. Revista Cubana de Estomatologia 2011; 48(2): 156-161.
28. Alcântara LR. Idosos rurais: fatores que influenciam trajetórias e acesso a serviços de saúde no municipio de Santana de Boa Vista/RS [dissertação]. Escola de Enfermagem. Universidade Federal do Rio Grande do Sul, 2009.



