full article:
Cardiovascular disease (CVD) mortality is the leading cause of death in the USA1. CVD risk factors including high blood pressure (BP), poor lipid profile, and impaired glucose tolerance are now prevalent in youth as well as in adults2,3. Data from National Health and Nutrition Examination Survey (NHANES) shows that, in the USA, 14% of adolescents had elevated BP, 22% had borderline-to-high or high low-density lipoprotein cholesterol (LDL-C), 6% had low high-density lipoprotein cholesterol (HDL-C) (NHANES: 1999-2008)4 and 32% children aged 2-19 years were either overweight or obese (NHANES: 2009-2010)5. Pediatric obesity also increases the likelihood of development of other CVD risk factors during childhood and adolescence2. Moreover, research shows that childhood CVD risk factors such as obesity, high BP, and abnormal lipids also track over time into adulthood6.
Several studies have examined CVD risk factor prevalence and its contributing factors by stratified analysis of various sociodemographic characteristics (such as age, gender, racial/ethnic background and income)4,5,7-9; a select few have examined these differences by urban and rural geographic distribution in the USA. These studies have revealed mixed findings by age. For example, data from a nationally representative study showed that older rural children (12-19 years) had 30% higher odds of being overweight or obese compared to urban children, although no significant differences were observed in younger children (2-11 years)10. A recent meta-analysis using data of US children aged 2-19 years found that rural children compared to urban children are 26% more likely to be obese (odds ratio (OR)=1.26; 95% confidence interval (CI)=1.21-1.32)11.
Different states have different rural/urban prevalence distributions by CVD risk factors as well. For example, a study in North Carolina found no differences in total cholesterol (TC) and BP of rural and urban children, but found obesity rates to be significantly greater for rural children within the state12. Another study demonstrated a significantly higher prevalence of obesity in rural children when compared to children living in metropolitan centers of Pennsylvania13.
The mixed findings on the differences of urban/rural prevalence of various childhood CVD risk factors suggest the importance of investigating this issue further. Some researchers suggest that merely living in a certain geographic location is not, in itself, a risk factor, but factors that differ between urban/rural residence contribute to the observed differences in CVD risk factors14. Others argue that there remains a strong link between rurality and obesity that cannot be explained by demographic factors alone15.
Some researchers have reasoned that the mixed findings may be due to the broad classification of urban/rural (81% vs 19%) or metropolitan/non-metropolitan areas (85% vs 15%)11. A nationally representative study using census block-group level found no significant associations between urban/rural status and childhood obesity after controlling for individual-level and zip code-level covariates16. In comparison, researchers using nationally representative data of grade 7-12 students classified neighborhood patterns in six categories and found that adolescents in select neighborhoods, including those living in rural working class, ex-urban, and mixed-race urban areas, were approximately 30% more likely to be overweight compared to those living in the newer suburban areas, independent of age, race, and socioeconomic status17. Thus, carefully defining urban and rural locations may be an important piece of the puzzle. In order to overcome the limitations of the broad classification system, the US Department of Agriculture has introduced a multitier classification scheme derived from the Office of Management and Budget and US Census definitions, which includes the Urban Influence Codes, Rural-Urban Continuum Codes (RUCC), and the Rural-Urban Commuting Area18. For the purpose of this study the authors used the nine-tier RUCC classification system, which is based on county-level data that can be matched to zip codes, and broken down into finer residential groups, beyond urban/rural, which is particularly useful for the analysis of trends in non-metro areas that are related to population density and metro influence.
Beyond the broad classifications of urban or rural location definitions, there remain conflicting results regarding the influence of rural or urban living on cardiovascular risk factors in children. A recent systematic review notes a lack of several key factors needed to make accurate conclusions from the data, including representative data, appropriate use of clustering, controlling for individual socioeconomic factors, longitudinal designs and intermediate mechanisms between environmental characteristics and cardiometabolic risk factors19. Thus, in addition to carefully defining rural urban divisions, this study includes several of these predefined factors, including appropriate use of statistical clustering analysis, representative data, and inclusion of some individual socioeconomic factors.
This exploration of the topic begins in a section of the USA with a large rural population. The Appalachian region consists of 420 counties in 13 states of which 42% of the population lives in rural areas20. Appalachian populations generally have higher rates of CVD risk factors and CVD mortality compared to the rest of the nation21. According to the Behavioral Risk Factor Surveillance System 2013 survey, West Virginia, a state entirely located in the Appalachian region, now ranks number one in adult obesity in the nation22. The 2011 Youth Risk Behavior Survey showed that 15.7% of adolescents were overweight and 14.6% were obese in West Virginia compared to the national averages of 15.2% and 13% respectively23.
In order to understand the geographic disparities in youth CVD risk factors, the authors aim to examine rurality and CVD risk factors in a large, representative sample from the Appalachian state of West Virginia, using appropriate clustering analytic techniques and controlling for several individual-level socioeconomic factors. It is hypothesized that more rural regions will be associated with poor CVD risk factor outcomes. Examining CVD risk factors during childhood by geographic distribution in the Appalachian region is the first step towards understanding the health disparities in this area.
The Coronary Artery Risk Detection In Appalachian Communities (CARDIAC) project started as a small school-based CVD surveillance project piloted in three rural West Virginia counties in 1998, and now includes services to all 55 West Virginia counties and more than 480 schools. For nearly two decades, CARDIAC has provided information to participating families, communities, the state and nation24 about chronic illnesses including hyperlipidemia25, abnormal blood lipids and obesity26,27, asthma28, decreasing cholesterol risk29, pre-diabetic conditions30, health behaviors31, and intervention factors32,33. Average findings for the program period demonstrate that from 1998 to 2014, 47.1% of fifth-grade students in West Virginia were either overweight or obese (body-mass index (BMI) percentile≥85th). Only fifth-grade participants receive lipid profiles and thus are the only participants included in this study.
With an active consent process for parents of fifth-grade participants, response rates by year range from 31% to 49% since 1998. These response rates for a health surveillance program are typical of the active consent process in elementary and middle school settings34,35. Previous work has shown that the differences between participants and non-participants are minimal. Non-participants are less likely to have a primary care provider and to have health insurance, but there is no difference in BMI or any other demographic variables analyzed in the present study36.
Measures
The comprehensive risk screening for fifth-grade participants included calculation of BMI from height and weight, resting diastolic BP (DBP) and systolic BP (SBP), and either a fasting or non-fasting lipid profile (FLP).
Children's heights (cm) and weights (kg) were measured using the SECA Road Rod stadiometer and the SECA 840 Personal Digital Scale. Students were asked to remove shoes and outerwear prior to height and weight measurements. These measurements were used to determine each child's BMI and BMI percentile, calculated using CDC Epi Info v3.5.4 (Centers for Disease Control and Prevention; http://www.cdc.gov/epiinfo). Percentage above ideal BMI (BMI% above ideal) was calculated using 100*log base e (BMI/median BMI)37 to control for age, gender and height and avoid the ceiling effect seen when using BMI percentile.
BP was taken after the child had been resting for 5 minutes. The first Korotkoff sound was used to record SBP and the fifth Korotkoff sound was used to record DBP.
All cholesterol levels were obtained in either a private area of the school or children were given a voucher to have an FLP conducted in a nationally available laboratory network or hospital. The FLP data analyzed in this manuscript start in 2003, and do not include any finger-stick obtained cholesterol levels (1998-2002) in order to avoid potential bias due to different methods of cholesterol measurements. Consistent blood specimens were taken since 2003, and all labs (hospital and the laboratory) used consistent methods to process the specimens.
RUCC were retrieved by zip code from the Missouri Census Data Center using the Beale 2003 RUCC code; multiple codes within zip codes were resolved by taking the largest proportion within the zip code. RUCC, a nine-point classification system based on county-level data, was further reduced to six categories for this analysis: large metro (counties in metro areas of ≥1 million people), metro (250 000-1 million), small metro (<250 000), non-metro urban (≥20 000), urban (2500-20 000), and rural (<2500).
Other covariates included parent-reported child birth date and calculated age at screening date, gender, race (six categories: white, black, Asian, Hispanic, bi-racial, and other), and mother's education (six categories: eighth grade or less, some high school, high school or GED [General Educational Development test], some college or technical training, college graduate, completed graduate school).
Statistical analysis
Data for CARDIAC were stored in the Statistical Package for the Social Sciences v21 (IBM; http://spss.com). All analyses conducted in this manuscript used Statistical Analysis Software v9.4 (SAS Institute; http://www.sas.com). ArcGIS Desktop v10 (Environmental Systems Research Institute; http://www.esri.com/arcgis) was used to develop RUCC maps. The data for this project include 73 014 fifth-grade children who participated in CARDIAC between 2003 and 2014. Missing data was assumed to be either missing completely at random or missing at random, and dealt with using pairwise deletion.
The statistical approach used a clustered design nesting individual children's FLP, BMI and BP results within their home zip code coded using RUCC and controlling for individual and socioeconomic status covariates. CARDIAC participants were matched to the RUCC code data file clustered in 725 zip codes. Triglycerides (TRIG) were log-transformed for the analyses. No interaction terms between socioeconomic status indicators and outcome variables were significant; results presented here include a two-level random-effects linear mixed model (students nested within zip codes) with a variance components covariance structure (chosen via Akaike information criterion fit) and restricted maximum likelihood estimation. Reference categories within the mixed model for categorical variables were set to white (race), male (gender), some high school (mother's education), and rural (RUCC). Least square means are presented with all pairwise comparisons between RUCC categories conducted with type I error adjusted for using Tukey-Kramer method, alpha set to 0.05.
Ethics approval
West Virginia University Institutional Review Board approved the study protocol (IRB 1606162244).
RUCC classification using the six-category system can be seen in Figure 1. Table 1 shows number of CARDIAC participants within each region.
Significant nested omnibus effects were seen for all outcomes after controlling for covariates, including BMI, HDL, SBP, DBP, log-transformed TRIG, LDL, and TC (p<0.0001; Table 2). However, posthoc comparisons disputed the hypothesis that rural areas would have significantly higher risk factors than urban or metro areas (Fig2). Specific outcomes are presented in more detail below. Omnibus type 3 tests of fixed effects are presented in text along with least square means and adjusted p-values for significant pairwise comparisons; all fixed effects are presented in more detail in Table 2.
Table 1: Demographics of Coronary Artery Risk Detection In Appalachian Communities fifth-grade participants (N=73 014)
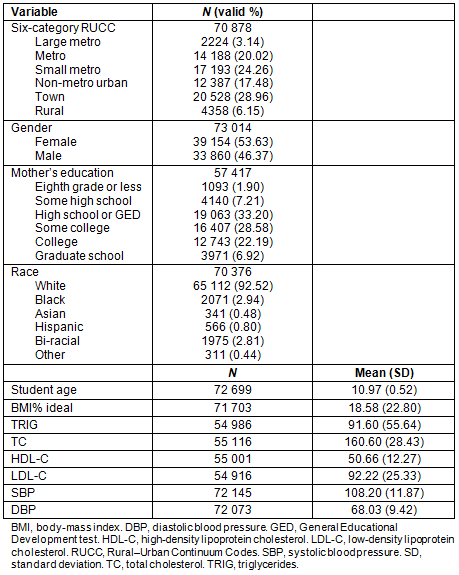
Table 2: Solution for fixed effects of outcomes
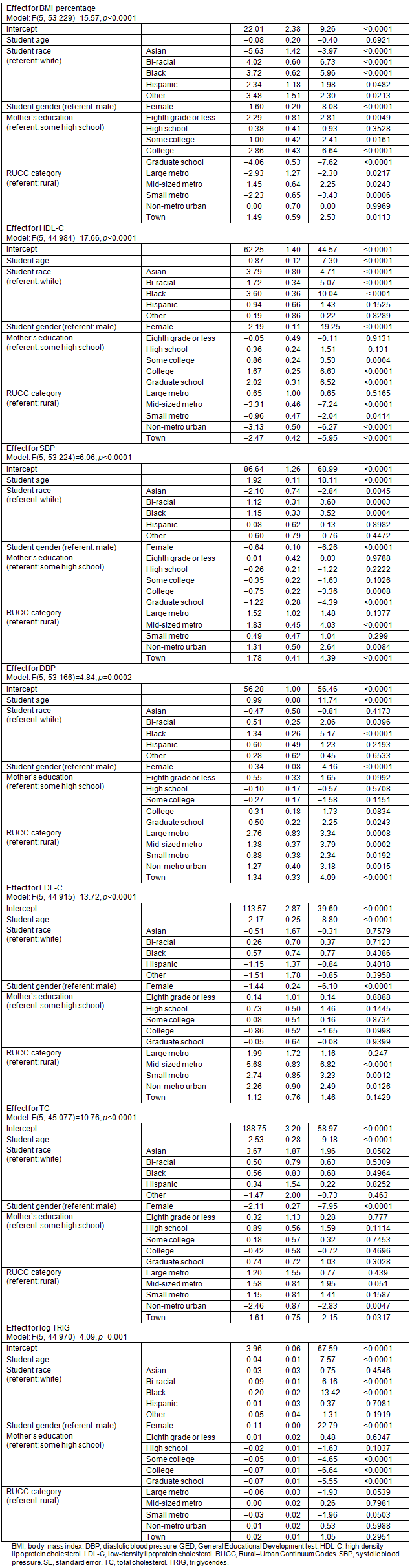
Figure 1: ArcGIS map of Coronary Artery Risk Detection In Appalachian Communities fifth-grade participants
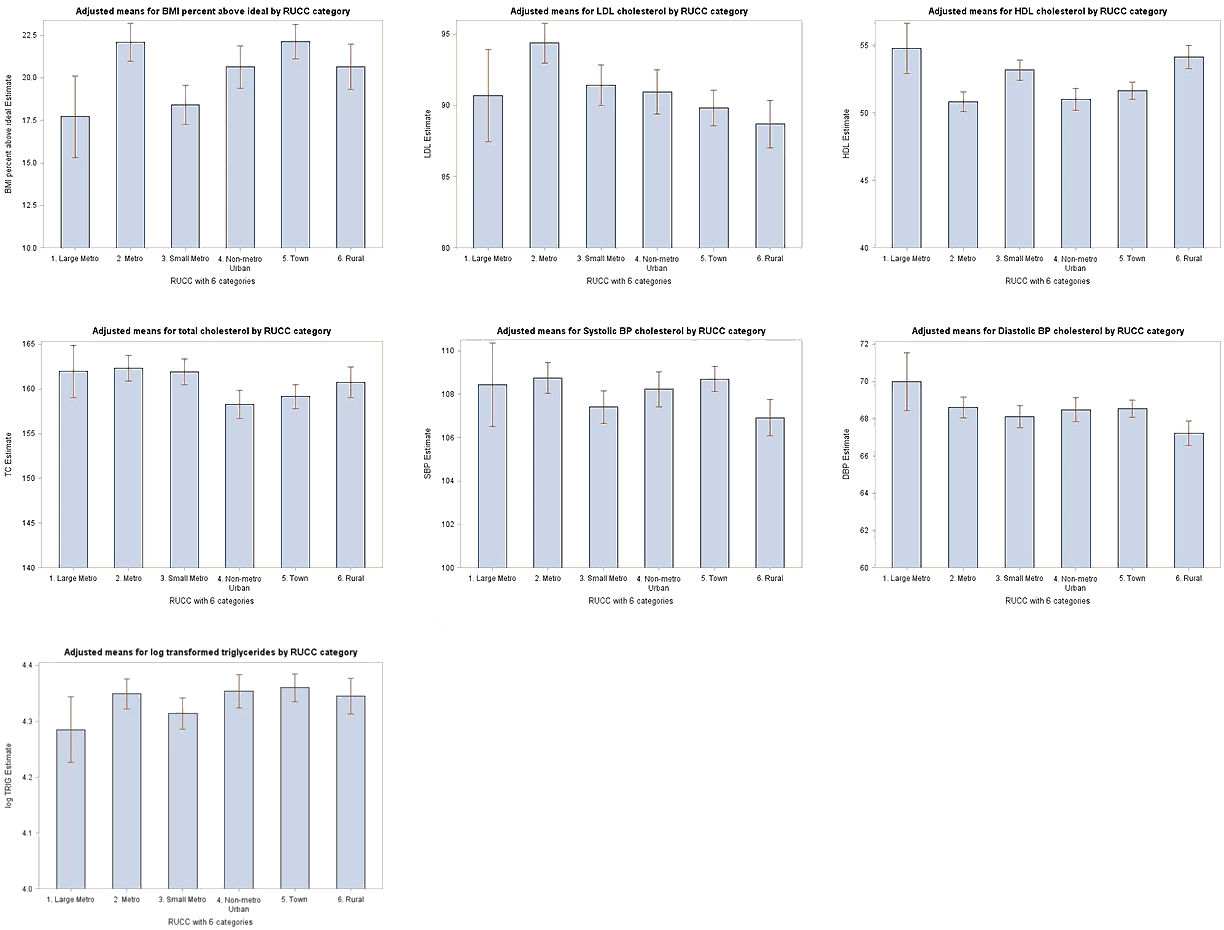
Figure 2: Least square adjusted means for study outcomes by Rural-Urban Continuum Codes category
Outcomes
BMI% above ideal: Significant type 3 test of fixed effects for RUCC, F(5, 53 229)=15.57, p<0.0001; race, F(5)=20.32, p<0.0001; gender, F(1)=65.22, p<0.0001; and maternal education, F(5)=33.8, p<0.0001; but not student age (p=0.69). Mid-sized metro (mean=22.09) and urban (mean=22.13) had significantly higher means than large metro (mean=17.72) and small metro (mean=18.41, all adjusted p<0.05). Rural (mean=20.64) and non-metro urban (mean=20.64) were significantly higher means than small metro (all adjusted p<0.05).
HDL-C: Significant type 3 test of fixed effects for RUCC, F(5, 44 984)=17.66, p<0.0001; race, F(5)=28.29, p<0.0001; gender, F(1)=370.55, p<0.0001; maternal education, F(5)=22.55, p<0.0001; and student age, F(1)=53.31, p<0.0001. Large metro (mean=54.78), rural (mean=54.13) and small metro (mean=53.17) areas had the best (highest) HDL as compared to mid-sized metro (mean=50.82), non-metro urban (mean=51.0), and urban areas (mean=51.65, all adjusted p<0.05).
SBP: Significant type 3 test of fixed effects for RUCC, F(5, 53 224)=6.06, p<0.0001; race, F(5)=6.69, p<0.0001; gender, F(1)=39.15, p<0.0001; maternal education, F(5)=6.77, p<0.0001; and student age, F(1)=327.95, p<0.0001. Rural areas (mean=106.92) had significantly lower SBP than mid-sized metro (mean=108.75), and urban (mean=108.70) areas (all adjusted p<0.05). Small metro (mean=107.40) was also significantly less than mid-sized metro and urban areas (all adjusted p<0.05). Non-metro urban (mean=108.23) and large metro (mean=108.44) areas were not significantly different than other areas.
DBP: Significant type 3 test of fixed effects for RUCC, F(5, 53 166)=4.84, p=0.0002; race, F(5)=6.38, p<0.0001; gender, F(1)=17.28, p<0.0001; maternal education, F(5)=3.08, p=0.009; and student age, F(1)=137.73, p<0.0001. Rural (mean=67.22) had significantly lower means than urban (mean=68.55), large metro (69.98), non-metro urban (mean=68.49), and mid-sized metro (mean=68.60) areas (all adjusted p<0.05). Small metro (mean=68.10) was not significantly different than other areas.
Log TRIG: Significant type 3 test of fixed effects for RUCC, F(5, 44 970)=4.09, p=0.001; race, F(5)=42.49, p<0.0001; gender, F(1)=519.45, p<0.0001; and maternal education, F(5)=20.77, p<0.0001. Town (mean=4.36) was significantly larger than small metro (mean=4.32) area (adjusted p=0.0014). No differences were found among large metro (mean=4.28), mid-sized metro (mean=4.35), non-metro urban (mean=4.35), or rural (mean=4.35) areas.
LDL-C: Significant type 3 test of fixed effects for RUCC, F(5, 44 915)=13.72, p<0.0001; student age, F(1)=77.5, p<0.0001; gender, F(1)=37.27, p<0.0001; and maternal education, F(5)=4.77, p=0.0002; but not race (p=0.81). Mid-sized metro (mean=94.37) presented with higher LDL than rural (mean=88.70), urban (89.81), small metro (mean=91.43), and non-metro urban (mean=90.95) areas, and small metro was also significantly larger than rural (all adjusted p<0.05). Large metro (mean=90.69) was not significantly different than other areas.
TC: Significant type 3 test of fixed effects for RUCC, F(5, 45 077)=10.76, p<0.0001; student age, F(1)=84.23, p<0.0001; gender, F(1)=63.17, p<0.0001; and maternal education, F(5)=2.91, p=0.013; but not race (p=0.81). Mid-sized metro area (mean=162.35) and small metro (mean=161.91) presented with higher TC over non-metro urban (mean=158.3) and urban (mean=159.15) areas (all adjusted p<0.0001). No differences were found with large metro (mean=161.97), small metro (mean=161.91) or rural (mean=160.77) areas.
Discussion
The study's findings directly counter previous literature connecting more rural locations to CVD risk factors, particularly obesity10,12-14. The authors instead note particularly poor outcomes in mid-sized metro areas in the Appalachian state of West Virginia, using clustered analysis techniques, representative sample, and adjusting for individual-level covariates. The mid-sized metro areas include counties in metro areas having a population density of 250 000 to 1 million, which is an urban/metropolitan area according to the binary urban/rural or metropolitan/non-metropolitan classification system. Specifically, mid-sized metro and urban areas presented with the highest average BMI% above ideal for age and gender among fifth-grade participants in the state, the lowest HDL-C, and highest mean DBP, LDL-C, TC, and log-transformed TRIG levels. In contrast, the rural areas (population density <2500) tended to have among the best outcomes for certain CVD risk factors, including LDL-C, HDL-C, and BP.
Although the authors note that rural areas have better outcomes compared to other regions in West Virginia, these regions continue to have high CVD risk compared to other regions in the nation23. It is also noted that large metro areas generally presented with some of the best outcomes in the state, perhaps most consistent with previous research. However, large metro also presented the highest average DBP. This may be an artifact of only one region in West Virginia being considered large metro: the eastern panhandle of West Virginia, a distal suburb of Washington DC. Again, an examination of intervening mechanisms is needed.
Although not a focal point of this article, examination of the covariates also yields some interesting findings. Female students generally had improved outcomes over males, except for HDL-C and log TRIG. This may be due to lower cholesterols occurring with puberty at younger ages in females. As maternal education increased, outcomes consistently improved on average. Although the majority of the state of West Virginia is white, this study notes worse outcomes in terms of BMI, SBP and DBP for other racial groups, but surprisingly improved outcomes such as HDL-C and log TRIG among all other racial groups.
Limitations include use of cross-sectional data, limiting any type of causal inference. Beale RUCC codes were from 2003, so more recent zip codes added since 2003 could not be included in this analysis. Additionally, Beale RUCC codes are based on county-level data, which limits within-county conclusions. Also, the authors could not explore mechanisms for the associations between geographic locations and CVD risk outcomes in this particular study. Despite these limitations, these results add to the literature in terms of presenting data representative of the generally rural Appalachian region with appropriate statistical modeling techniques and individual-level covariate inclusion. Furthermore, this study used the RUCC classification system and aims to overcome some of the potential limitations of a broad binary classification system that fails to account for some of the variances present in areas that have a population greater than 2500 but are either adjacent to a metro area (small metro) or not adjacent to a metro area (non-metro urban). Results themselves were counter to the authors' prior hypotheses, suggesting the relationship between rurality and CVD risk factors to be more complex than previously supposed. The study adds to the understanding of the differences in geographic CVD risk factors distribution in the Appalachian region of West Virginia. Future research is needed to identify the factors associated with the differences observed. This can lead to potential interventions geared specially in geographic areas where the risk factors are significantly higher.
In general, mid-sized metro areas presented with the worst CVD risk factors outcomes in children and adolescents in the Appalachian state of West Virginia. This data contradicts previous literature suggesting a straightforward link between rurality and cardiovascular risk factors. Future research should include a longitudinal design and explore some of the mechanisms between cardiovascular risk factors and rurality.
Acknowledgements
The authors thank Evan Fedorko of the West Virginia Geographic Information System (GIS) Technical Center for his work with the GIS maps, Paula Nicholson and Emily Polak for CARDIAC data management, and the CARDIAC area coordinators (Kathryn Greenlief, Janetta Massie, Dalena Riggs, Robin VanFleet, Tammy Vicars, Tina Whitt) for data collection. Special thanks also to Eloise Elliott, Valerie Minor, and Susie Ritchie for their work with the CARDIAC project.
References
1. Murphy SL, Xu J, Kochanek KD. Deaths: final data for 2010. National Vital Statistics Reports 2013; 61(4): 1-117.
2. Friedemann C, Heneghan C, Mahtani K, Thompson M, Perera R, Ward AM. Cardiovascular disease risk in healthy children and its association with body mass index: systematic review and meta-analysis. British Medical Journal 2012; 345: e4759. https://doi.org/10.1136/bmj.e4759
3. Falkner B. Hypertension in children and adolescents: epidemiology and natural history. Pediatric Nephrology 2010; 25(7): 1219-1224. https://doi.org/10.1007/s00467-009-1200-3
4. May AL, Kuklina EV, Yoon PW. Prevalence of cardiovascular disease risk factors among US adolescents, 1999-2008. Pediatrics 2012; 129(6): 1035-1041. https://doi.org/10.1542/peds.2011-1082
5. Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity and trends in body mass index among US children and adolescents, 1999-2010. JAMA 2012; 307(5): 483-490. https://doi.org/10.1001/jama.2012.40
6. Juhola J, Magnussen CG, Viikari JS, Kahonen M, Hutri-Kahonen N, Jula A, et al. Tracking of serum lipid levels, blood pressure, and body mass index from childhood to adulthood: the Cardiovascular Risk in Young Finns Study. Journal of Pediatrics 2011; 159(4): 584-590. https://doi.org/10.1016/j.jpeds.2011.03.021
7. Cassedy A, Drotar D, Ittenbach R, Hottinger S, Wray J, Wernovsky G, et al. The impact of socio-economic status on health related quality of life for children and adolescents with heart disease. Health and Quality of Life Outcomes 2013; 11: 99. https://doi.org/10.1186/1477-7525-11-99
8. Ogden CL, Lamb MM, Carroll MD, Flegal KM. Obesity and socioeconomic status in children and adolescents: United States, 2005-2008. NCHS Data Brief 2010; 51: 1-8.
9. Caprio S, Daniels SR, Drewnowski A, Kaufman FR, Palinkas LA, Rosenbloom AL, et al. Influence of race, ethnicity, and culture on childhood obesity: implications for prevention and treatment. Obesity (Silver Spring) 2008; 16(12): 2566-2577. https://doi.org/10.1038/oby.2008.398
10. Liu JH, Jones SJ, Sun H, Probst JC, Merchant AT, Cavicchia P. Diet, physical activity, and sedentary behaviors as risk factors for childhood obesity: an urban and rural comparison. Childhood Obesity 2012; 8(5): 440-448. https://doi.org/10.1089/chi.2011.0090
11. Johnson JA, 3rd, Johnson AM. Urban-rural differences in childhood and adolescent obesity in the United States: a systematic review and meta-analysis. Childhood Obesity 2015; 11(3): 233-241. https://doi.org/10.1089/chi.2014.0085
12. McMurray RG, Harrell JS, Bangdiwala SI, Deng S. Cardiovascular disease risk factors and obesity of rural and urban elementary school children. Journal of Rural Health 1999; 15(4): 365-374. https://doi.org/10.1111/j.1748-0361.1999.tb00760.x
13. Bailey-Davis L, Horst M, Hillemeier MM, Lauter A. Obesity disparities among elementary-aged children: data from school-based BMI surveillance. Pediatrics 2012; 130(6): 1102-1109. https://doi.org/10.1542/peds.2012-0192
14. Davis AM, Bennett KJ, Befort C, Nollen N. Obesity and related health behaviors among urban and rural children in the United States: data from the National Health And Nutrition Examination Survey 2003-2004 and 2005-2006. Journal of Pediatric Psychology 2011; 36(6): 669-676. https://doi.org/10.1093/jpepsy/jsq117
15. Lutfiyya MN, Lipsky MS, Wisdom-Behounek J, Inpanbutr-Martinkus M. Is rural residency a risk factor for overweight and obesity for U.S. children? Obesity (Silver Spring) 2007; 15(9): 2348-2356. https://doi.org/10.1038/oby.2007.278
16. Zhang X, Onufrak S, Holt JB, Croft JB. A multilevel approach to estimating small area childhood obesity prevalence at the census block-group level. Preventing Chronic Disease 2013; 10: E68. https://doi.org/10.5888/pcd10.120252
17. Nelson MC, Gordon-Larsen P, Song Y, Popkin BM. Built and social environments associations with adolescent overweight and activity. American Journal of Preventative Medicine 2006; 31(2): 109-117. https://doi.org/10.1016/j.amepre.2006.03.026
18. Hart LG, Larson EH, Lishner DM. Rural definitions for health policy and research. American Journal of Public Health 2005; 95(7): 1149-1155. https://doi.org/10.2105/AJPH.2004.042432
19. Leal C, Chaix B. The influence of geographic life environments on cardiometabolic risk factors: a systematic review, a methodological assessment and a research agenda. Obesity Reviews 2011; 12(3): 217-230. https://doi.org/10.1111/j.1467-789X.2010.00726.x
20. Appalachian Regional Commission. ARC Designated Distressed Counties - fiscal year 2009. (Internet). Available: http: //www.arc.gov/appalachian_region/TheAppalachianRegion.asp 2011 (Accessed 9 September 2016).
21. Halverson JA, Ma L, Harner EJ, Hanham R, Braham V. Adult obesity in Appalachia: an atlas of geographic disparities. Morgantown, WV: West Virginia University, Prevention Research Center, Center for Healthy Communities, 2004.
22. Behavioral Risk Factor Surveillance System. Obesity prevalence maps. Obesity prevalence in 2013 varies across states and territories. (Internet). Available: http: //www.cdc.gov/obesity/data/prevalence-maps.html 2013 (Accessed 9 September 2016).
23. Eaton DK, Kann L, Kinchen S, Shanklin S, Flint KH, Hawkins J, et al. Youth risk behavior surveillance - United States, 2011. MMWR Surveillance Summaries 2012; 61(4): 1-162.
24. Cottrell L, John C, Murphy E, Lilly CL, Ritchie SK, Elliott E, et al. Individual-, family-, community-, and policy-level impact of a school-based cardiovascular risk detection screening program for children in underserved, rural areas: The CARDIAC Project. Journal of Obesity 2013; 2013: 7. https://doi.org/10.1155/2013/732579
25. John C, Neal W. Screening children for hyperlipidemia by primary care physicians in West Virginia. West Virginia Medical Journal 2012; 108(3): 30-35.
26. Ice CL, Cottrell L, Neal WA. Body mass index as a surrogate measure of cardiovascular risk factor clustering in fifth-grade children: results from the coronary artery risk detection in the Appalachian Communities Project. International Journal of Pediatric Obesity 2009; 4(4): 316-324. https://doi.org/10.3109/17477160802596197
27. Ice CL, Murphy E, Cottrell L, Neal WA. Morbidly obese diagnosis as an indicator of cardiovascular disease risk in children: results from the CARDIAC Project. International Journal of Pediatric Obesity 2011; 6(2): 113-119. https://doi.org/10.3109/17477161003792556
28. Cottrell L, Neal WA, Ice C, Perez MK, Piedimonte G. Metabolic abnormalities in children with asthma. American Journal of Respiratory and Critical Care Medicine 2011; 183(4): 441-448. https://doi.org/10.1164/rccm.201004-0603OC
29. Lilly CL, Gebremariam YD, Cottrell L, John C, Neal W. Trends in serum lipids among 5th grade CARDIAC participants, 2002-2012. Journal of Epidemiology and Community Health 2014; 68(3): 218-223. https://doi.org/10.1136/jech-2013-203253
30. Ice CL, Murphy E, Minor VE, Neal WA. Metabolic syndrome in fifth grade children with acanthosis nigricans: results from the CARDIAC project. World Journal of Pediatrics 2009; 5(1): 23-30. https://doi.org/10.1007/s12519-009-0004-7
31. Cottrell LA, Minor V, Murphy E, Ward A, Elliott E, Tillis G, et al. Comparisons of parent cardiovascular knowledge, attitudes, and behaviors based on screening and perceived child risks. Journal of Community Health Nursing 2007; 24(2): 87-99. https://doi.org/10.1080/07370010701316213
32. Cottrell L, Spangler-Murphy E, Minor V, Downes A, Nicholson P, Neal WA. A kindergarten cardiovascular risk surveillance study: CARDIAC-Kinder. American Journal of Health Behavior 2005; 29(6): 595-606. https://doi.org/10.5993/AJHB.29.6.14
33. Elliott E, Jones E, Bulger S. ActiveWV: a systematic approach to developing a physical activity plan for West Virginia. Journal of Physical Activity & Health 2014; 11(3): 478-486. https://doi.org/10.1123/jpah.2013-0083
34. Doumas DM, Esp S, Hausheer R. Parental consent procedures: impact on response rates and nonresponse bias. Journal of Substance Abuse and Alcohol 2015; 3(2): 1031.
35. Rosenberg KD, Hembroff L, Drisko J, Viner-Brown S, Decker K, Lichter E. New options for child health surveillance by state health departments. Maternal and Child Health Journal 2011; 15: 302. https://doi.org/10.1007/s10995-010-0589-4
36. Harris CV, Neal WA. Assessing BMI in West Virginia schools: parent perspectives and the influence of context. Pediatrics 2009; 124(Suppl 1): S63-72. https://doi.org/10.1542/peds.2008-3586I
37. Cole TJ, Faith MS, Pietrobelli A, Heo M. What is the best measure of adiposity change in growing children: BMI, BMI %, BMI z-score or BMI centile? European Journal of Clinical Nutrition 2005; 59(3): 419-425. https://doi.org/10.1038/sj.ejcn.1602090
