Epidemiological and other studies have indicated that where you live can have an impact on your health. People living in rural areas, due to limited or lack of access to health care, have overall poorer health outcomes than people living in urban areas1-5. This is also the case for cancer patients in developed and underdeveloped countries6,7. A 15 year analysis of cancer survival data in England concluded that inequalities in care exist between the rural North and urban South of England, with patients in the South receiving better care. Another British study concluded that the further away one lives from a tertiary healthcare center (more rural), the higher the death rate from cancer, with the exception of breast cancer. The greater the distance patients lived away from the hospital, the higher the probability that their cancer was diagnosed post-mortem8. The authors of the study did suggest that the age of the patients may have played a role in their health outcome, with older, perhaps sicker, people forfeiting treatment due to travel problems.
In many countries there is an intersection between rural living, the elderly and poverty9. In Australia, New Zealand and the Pacific Rim, cancer survival is poorer for rural cancer patients7,10,11. Two European studies indicated that cancer patients living far away from cancer centers are less likely to receive optimal cancer treatment12,13. Rural patients also perceive that they have reduced access to healthcare services14. In Canada, rural women with breast cancer reported having less access to cancer support services than their urban counterparts15. Other Canadian studies have indicated similar disparities in access to cancer care between rural and urban patients11,16. A systematic review concluded that distance away from healthcare centers is a significant variable that determines access to cancer therapy and palliative radiation for cancer patients17.
In addition to the above-mentioned difference between urban and rural cancer care, young adult cancer survivors have unique needs compared to the older cancer population. The unique needs are related to fertility, disclosure, social relations and issues such as work and study, healthcare system issues (supplemental private health insurance)18-21. It is important that for young adult cancer survivors, physical, psychological and social morbidity is minimized because they have potential long and healthy years ahead of them22. In short, the transition from cancer care to survivorship is very important for this age group23,24.
The evidence thus far supports the notion that rural cancer patients have poorer access to healthcare resources. This article examines this notion from the perspective of a unique group of cancer patients: young adults. Qualitative methodology was used to compare the experiences of young adult cancer patients regarding their cancer care in two distinct areas of Canada: New Brunswick, a rural Eastern Canadian province, and Toronto, Canada's largest metropolitan center.
Context
The original research questions were based on the cancer follow-up care experiences of young adult cancer survivors. The authors identified a serious gap in follow-up care for this cancer population. Overall, regardless of the stage of the disease, acute or follow-up care, the participants identified the lack of age-specific care. The participants felt that they have unique needs which are not being met25. It should be noted that, although the focus was on cancer follow-up care, in reality, cancer patients seldom differentiate between diagnosis, acute treatment and cancer follow-up care when discussing their experiences with cancer. For many patients, it is all 'cancer care'.
Analysis of the data revealed issues related to overall cancer care, and it appeared that the patients' experiences were influenced by where they lived and where they had received their cancer treatment. Being aware of the fact that much of the literature makes a strong case for the inferiority of healthcare services in rural areas, it was felt that it would be valuable to compare the experiences of rural and urban participants in the sample.
Setting
New Brunswick is an Eastern rural Canadian province with a population of approximately 750 000. New Brunswick has three major cities. These cities and their surrounding communities have populations ranging from 57 000 to 70 00026. The Greater Toronto area (GTA) has a population of 5.5 million. The geographical area of New Brunswick is roughly 10 times larger than the geographical area of the GTA. New Brunswick is a sparsely populated geographical area with a homogenous population; Toronto is a densely populated multicultural metropolis.
Canada is a loose federation of provinces and territories with a governance system that gives provinces much autonomy. For example, social programs such as universal health insurance and education are provincially run with some federal oversight. Each provincial government is responsible for the delivery of universal healthcare services. By law, every Canadian citizen has access to basic medical coverage. These services include physician and tertiary care services. Medication coverage is not included in the provincial insurable healthcare services. In addition to the basic healthcare insurance, many Canadians purchase private supplemental healthcare insurance to cover the costs associated with medications, orthotics and other non-medicinal aids, and allied health professional treatments such as psychological counseling, and physiotherapy. There is great variability in the medications and services covered by private insurance policies.
The GTA is home to three large cancer hospitals that serve the entire GTA population. The Princess Margaret Hospital is the largest cancer center in Canada and the world, while the Sunnybrook Hospital focuses on three specific cancers, and Women's College is a leader in breast cancer treatment and research. All three hospitals combined treat more patients in one given year than the entire population of New Brunswick. In addition, they have very active cancer survivorship programs and cancer research programs. On average, GTA patients would not be more than 40 to 50 kilometers from a cancer hospital that can be reached by public transportation. New Brunswick has three major hospitals, although only two provide comprehensive cancer care that includes surgery (to all types of cancer), chemotherapy and radiation therapy. None of the New Brunswick hospitals provides cancer services specifically focused on young adults or extensive cancer survivorship programs. Cancer research is also limited in these three tertiary hospitals. Many patients must travel between hospitals for treatment, a distance of more than 120 km, and some patients must travel out of province to larger tertiary care hospitals in Nova Scotia or Quebec to seek adjuvant cancer therapies. Cancer patients who do not live in the three urban centers in New Brunswick will have to travel by private means for hours for cancer treatment. New Brunswick has a very poor public transportation system.
Definitions
This article is based on a qualitative study that examined the experiences of young adult cancer survivors with cancer follow-up care in Canada25. The term 'young adult' is not uniquely defined in the context of cancer27. The authors chose to include people who were diagnosed with cancer between the ages of 18 and 39 years of age as 'young adults' in this study. The rationale for this definition is a combination of factors that are patho-physiological (non-epithelial vs. epithelial cancers) and psychosocial in nature19,28. The young adult cancer patient's life stage is uniquely characterized by the quest for independence, educational credentials and parenthood19.
Participants
Young adults diagnosed with any kind of cancer between the ages of 18 and 39 were recruited 1 to 5 years post-diagnosis. Participants were required to speak either English or French, and capable of providing informed consent. Participants received a $20 gift certificate for participation. The recruitment strategy included posts on social networking sites (ie Facebook), online classified websites (ie Kijiji), newsletters and online messages boards at cancer advocacy groups (ie Young Adult Cancer Canada), posters in various oncology clinics, newspapers and other media articles and interviews. This strategy was used across the country. As with many studies, participants were self-selected29. In general qualitative data is not generalizable to the larger population, but it provides in-depth insight in patients' experiences that cannot be obtained from data-set analysis or survey data30.
Interested young adults who contacted us to indicate their willingness to participate were screened to ensure that they met the inclusion criteria. A consent form was sent by email, and a telephone interview date and time were set. Before the telephone interview commenced, the participant and the interviewer discussed the consent form to ensure that the participant was fully aware of the study procedures, risks and benefits, and oral consent was obtained over the telephone. Further, the participants were asked to consent to the audio recording of the interview. If the participants did not consent to being audio recorded, the interviewer took notes. In the larger study, 67 young adult cancer survivors from across Canada contacted us and 55 individuals participated in a telephone interview. Some people did not fit the inclusion criteria and others chose not to be interviewed after the initial contact. The interviews lasted between 45 and 90 min. Theme saturation (no new information reported by the participants) was reached at approximately interview 35; however, recruitment continued in order to ensure a diverse sample of participants based on gender, geographic location, and cancer type.
Data collection
Qualitative interviews were conducted using an interview schedule with open-ended questions. The first two questions were designed to make the participants feel comfortable and to have them tell their story regarding their cancer journey. Question 3 asked about current cancer follow-up care and questions 4 to 9 asked about broad cancer follow-up care issues that can be characterized as physical, psychological, relationship and social. Questions 10 and 11 related to experiences and satisfaction with current care, and recommendations for improvement. After the interview, sociodemographic information was collected to create a profile of the participants. Two research assistants, a male and female, conducted all of the interviews; the interviewees and the interviewers were gender paired. Data was collected over a 12 month period between 2010 and 2011.
Analysis
A Constructivist Grounded Theory (CGT) approach was used to analyze the data. CGT rejects the notion of an objective reality. It views realities as social constructs that may have multiple meanings/realities and that knowledge is mutually created, or in other words a co-construction of meaning by the 'viewer and the viewed'31. This, in the authors' view, is a more relevant interpretation of qualitative data than the objective reality promoted by grounded theorists32. CGT methods are often used in health research when researchers wish to hear the 'voices' of participants33,34. After the interviews were conducted they were transcribed verbatim and proofread. Four team members read the same three transcripts independently and developed a coding scheme. The remaining transcripts were coded line-by-line by one researcher to ensure consistency, using the constant comparison method and through continued discussions among the team members35-37. The coding process was facilitated by using the qualitative data analysis program NVivo 9 (www.qsrinternational.com). The sociodemographic information was analyzed using the statistical data analysis program Statistical Package for Social Sciences v15 (www.spps.com).
Ethics approval
The study was approved by Dalhousie University (#2009-2098) and Memorial University (#10-46) research ethics boards.
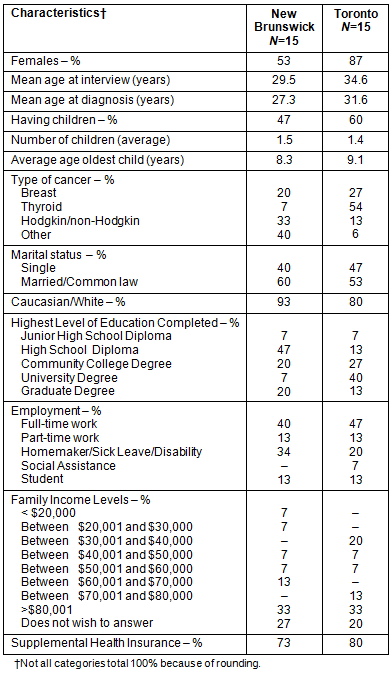
This article focuses exclusively on two subgroups: 15 young adults from New Brunswick and 15 young adults from the GTA. These two subgroups were not specifically recruited for the sub-study; it was serendipity that one group of 15 participants was living in New Brunswick and the other group of 15 was living in the GTA. Hence, a more detailed sub-analysis of these two groups was conducted.
The mean age of the participants was 29 years at time of diagnosis and 32 years at the time of the interview (Table 1). The cohorts of study participants are similar, except for the types of cancer, with a higher number of thyroid cancer patients in Toronto and a higher number of lymphoma patients in New Brunswick. The Toronto cohort also has higher levels of education and higher levels of income.
Table 1: Demographic characteristics of participants
Themes
The three major themes identified in the data were 'delayed diagnosis', 'cost related to cancer treatment' and 'community support'. Further, the 'satisfaction with care' question was analyzed.
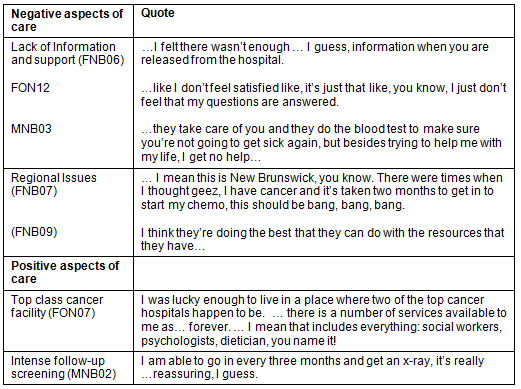
Delayed diagnosis: Three quotes that relate to the theme of 'delayed diagnosis' are presented (Table 2). The first patient describes how her complaints were dismissed for years. In the second case the physician did not take the patient's complaints seriously, and in the third example, physicians made the assumption that the patient was too young to have this type of cancer. Overall, more New Brunswick participants discussed a delayed diagnosis than did the GTA participants. For the New Brunswick participants, more than half reported having experienced a delay in their diagnosis, compared with 1 in 3 for the urban participants.
Table 2: Theme: Delayed diagnoses

Out of pocket cost: Several issues are listed (Table 3) that relate to healthcare costs and the unique position of young adults who are often not permanently employed, are still in school or dependent on their parents. The first illustrates difficulties experienced by some students when they are diagnosed with cancer and must withdraw from university or other schooling. This usually results in the loss of supplemental health coverage. As the patient in the second quote illustrates, similar difficulties are experienced by part-time workers who do not have paid sick days and limited supplemental health insurance.
While the goal of many young adults is to become independent, cancer can have a significant negative impact on this quest as illustrated in the third quote. This young man became entirely dependent on his parents due to the financial hardships associated with his cancer treatments.
Table 3: Theme: Cancer care cost

Community support: Specific questions about community support were not asked, but several participants brought this up. Few rural participants discussed community support. One rural participant was grateful that her community had organized a benefit breakfast to provide her with some financial and emotional support. None of the urban participants described any such event, but many more urban participants described receiving support either from faith communities or from cancer support organizations such as WellSpring. Cumulatively, urban participants reported receiving almost three times the level of community support received by rural participants. (Table 4).
Table 4: Theme: Community support

Satisfaction: The final theme is related to the satisfaction levels with cancer care, and it was found that the urban participants reported higher levels of dissatisfaction (negative experiences) with their care compared to the rural participants A substantial number of negative comments were related to the fact that participants felt that they had not received enough information about what to expect after their acute treatment was completed. Interestingly, many rural participants said they were satisfied with their care but this was qualified by the acknowledgement of the limited resources in their community. Many rural participants had low expectations of their care, so when they received more than expected, they were inclined to be satisfied (Table 5).
Table 5: Satisfaction levels (positive and negative)
Discussion
The profiles of the 30 young adult cancer survivors described in this study are similar except for three characteristics. There was a difference in the proportions of cancer types between the rural and urban groups, not an unusual finding given the number of participants in groups. Rural participants have lower educational achievements, less full-time employment and therefore lower average incomes. This is in keeping with the overall patterns for the two provinces: New Brunswick and Ontario (GTA)38,39. A greater variety in participants' ethnic backgrounds was expected, given that the GTA is multicultural; however, ethnicity was not selected as an inclusion criteria. Considerably more rural participants discussed having experienced a delay in their cancer diagnosis than did the urban participants. The issue of delayed diagnosis is complex. Sometimes patients delay seeking medical assistance, and sometimes the delay rests with the physician or the system40,41. A previous study, conducted exclusively in New Brunswick with young adult cancer survivors, also identified a problem of delayed diagnosis for young adult cancer patients42. It is not always clear if a delayed diagnosis leads to higher mortality, but it is clear that a perceived delayed diagnosis adds considerably to the stress experienced by the patient18,42. Nevertheless, delayed diagnosis may lead to more physical and psychological morbidity, which, in the long term, can have very negative consequences for young adult cancer survivors.
The sources of out-of-pocket expenses for cancer patients during the cancer journey are many. While medications administered for in-patients are provided free of charge, the provincial health insurance programs do not cover the cost of medications used outside the hospital setting. Private, supplemental health insurance pays all or some of the medication costs. In addition to medication costs, cancer patients may be burdened with a number of other expenses related to treatment or cosmetic issues following those treatments, such as travel and lodging, prostheses, and wigs. Some cancer patients have generous sick leave benefits from their employers, while others have either no health benefits or limited benefits. Students lose their supplemental health insurance when they stop attending on a full-time basis. Approximately 1 in 3 Canadians do not have private supplemental health insurance43. These patients may face considerable out-of-pocket expenses for cancer treatments. Although there was no data to support this fact, the authors believe it is likely that few young people have supplemental health insurance. Thus young adults with cancer may face higher out-of-pocket expenses than similarly stricken older persons. Travel expenses can add considerably to the financial burden of cancer patients44,45. From the literature, it is known that rural patients often are more adversely affected by out-of-pocket cancer treatment expenses44,45. It is not clear why the present study rural participants discussed out-of-pockets expenses less often than did their urban counterparts. It may be that travel costs are considered 'normal' for the rural participants and that they do not consider them 'out-of-pocket' cancer expenses.
Social support is important for cancer patients46. The authors were struck by the difference between the rural and urban study participants in their discussions of the level of community support. Although it appears that urban participants received more community support than the rural participants, this may also be the result of how the term 'community' was individually defined and interpreted by the participants, as this was not specifically defined for them. Community can be broadly defined as a specific group with which one is associated (eg church group, support group). An important finding from the present study was that despite the fact that it is often assumed that urban dwellers lack a sense of community due to large population size and urban sprawl, many urban participants relied on their personal 'communities' for support in difficult times, such as church groups and parent groups. Some studies have concluded that community support for cancer patients is not very different between urban and rural patients47. The present study cannot conclusively make a statement about more or less community support based on geography; however, good evidence was found that cancer support organizations, mostly associated with urban centers, have a positive impact on urban cancer survivors. Institutions such as WellSpring48 or dragon boat racing49 were reportedly much appreciated by the urban participants. Unfortunately, many rural cancer survivors do not have access to such services.
Rural participants reported having less trouble with out-of-pocket expenses, even though it can be assumed that they have at least as many, if not more, out-of-pocket expenses as their urban counterparts46. Rural participants also reported less community support and higher levels of delayed diagnosis. Despite these 'negative' experiences they reported higher levels of satisfaction. This is not an unusual finding; a study in the USA also reported that rural participants reported higher levels of satisfaction with health care than urban participants50. In the present study, rural participants seemed to have had low cancer care expectations, and most considered themselves to be lucky to have received the best care possible given their location and available services.
The authors acknowledge that provinces with small populations and large geography are not able to provide optimal cancer care for young adult cancer survivors, but they hope that with new technology some of these care impediments can be bridged, particularly in the area of psychological and social care issues.
Limitations
The study results are based on a theoretical sample and therefore cannot be generalized to all young adult cancer survivors. A larger proportion of female than male participants in this study is acknowledged, but this is very common phenomenon; females often participate more easily in research than males42,51. Nonetheless, the authors are confident that the participants represented a wide variety of young adult cancer survivors, and included diversity in the types of cancers and participants from a variety of geographical areas. As in most research, particularly in self-referral studies such as this one, participants who have experienced significant challenges may have been more inclined to participate than young adults who did not.
Administrative data supports the notion that, in general, rural cancer patients have less access to cancer care than urban cancer patients. This study examined the experiences of cancer care for a group of urban and rural young adult cancer patients. The rural participants were less likely to speak about the financial burden of their cancer experiences, less likely to speak about significant community support, spoke more often about a delay in their cancer diagnosis, and yet expressed a higher level of satisfaction with their cancer care. They were aware of the limitations of the treatments that were available to them, and yet felt positively toward their care providers who they felt did all that was possible under the circumstances. There is no evidence to support the notion that this level of satisfaction translated to better overall psychosocial health for this age group but would think that this association would exist, and it is believed that this needs to be studied further through intervention studies. Further, the authors feel it is important to test new technologies to bridge the gap between rural and urban cancer care for young adults.
Acknowledgements
The authors thank Dr Sue Tatemichi for reading and providing feedback on the draft manuscript. Canadian Institutes for Health Research MOP97737.
References
1. Valet RS, Perry TT, Hartert TV. Rural health disparities in asthma care and outcomes. The Journal of Allergy and Clinical Immunology 2009; 123(6): 1220-1225.
2. Yao N, Matthews SA, Hillemeier MM. White infant mortality in Appalachian states, 1976-1980 and 1996-2000: changing patterns and persistent disparities. Journal of Rural Health 2012; 28(2): 174-182.
3. Redwood D, Provost E, Perdue D, Haverkamp D, Espey D. The last frontier: innovative efforts to reduce colorectal cancer disparities among the remote Alaska Native population. Gastrointestinal Endoscopy 2012; 75(3): 474-480.
4. Karve S, Balkrishnan R, Seiber E, Nahata M, Levine DA. Population trends and disparities in outpatient utilization of neurologists for ischemic stroke. Journal of Stroke and Cerebrovascular Diseases 2011; epub (in press).
5. Goodridge D, Lawson J, Rennie D, Marciniuk D. Rural/urban differences in health care utilization and place of death for persons with respiratory illness in the last year of life. Rural and Remote Health 2010; 10(2): 1349.
6. Dong X, Simon MA. Health and aging in a Chinese population: urban and rural disparities. Geriatrics & Gerontology International 2010; 10(1): 85-93.
7. Walters S, Quaresma M, Coleman MP, Gordon E, Forman D, Rachet B. Geographical variation in cancer survival in England, 1991-2006: an analysis by Cancer Network. Journal of Epidemiology and Community Health 2011; 65(11): 1044-1052.
8. Jones AP, Haynes R, Sauerzapf V, Crawford SM, Forman D. Geographical access to healthcare in Northern England and post-mortem diagnosis of cancer. Journal of Public Health (Oxford, England) 2010; 32(4): 532-537.
9. Mata F, Bollman RD. An analysis of socio-economic strains and population gains: urban and rural communities of Canada 1981-2001. Canadian Studies in Population 2007; 32(2): 191-215.
10. Olver I, Marine F, Grogan P. Disparities in cancer care in Australia and the Pacific. Oncologist 2011; 16(7): 930-934.
11. McDonald JT, Sherman A. Determinants of mammography use in rural and urban regions of Canada. Canadian Journal of Rural Medicine 2010; 15(2): 52-60.
12. Dejardin O, Bouvier AM, Herbert C, Velten M, Buemi A, Delafosse P, et al. Social and geographic disparities in access to reference care site for patients with colorectal cancer in France. British Journal of Cancer 2005; 92(10): 1842-1845.
13. Baird G, Flynn R, Baxter G, Donnelly M, Lawrence J. Travel time and cancer care: an example of the inverse care law? Rural and Remote Health 2008; 8(4): 1003.
14. Hausdorf K, Rogers C, Whiteman D, Newman B, Coxeter P, Youl P, et al. Rating access to health care: Are there differences according to geographical region? Australian and New Zealand Journal of Public Health 2008; 32(3): 246-249.
15. Gray RE, James P, Manthorne J, Gould J, Fitch MI. A consultation with Canadian rural women with breast cancer. Health Expectations 2004; 7(1): 40-50.
16. Pong RW, Desmeules M, Lagace C. Rural-urban disparities in health: how does Canada fare and how does Canada compare with Australia? Australian Journal of Rural Health 2009; 17(1): 58-64.
17. Maddison AR, Asada Y, Urquhart R. Inequity in access to cancer care: a review of the Canadian literature. Cancer Causes & Control 2011; 22(3): 359-366.
18. Miedema B, Hamilton R, Easley J. From "invincibility" to "normalcy": coping strategies of young adults during the cancer journey. Palliative & Support Care 2007; 5(1): 41-49.
19. Cancer Care Ontario. Cancer in Young Adults in Canada. Toronto: Canadian Cancer Society, 2006.
20. Miedema B, Easley J. Barriers to rehabilitative care for young breast cancer survivors: a qualitative understanding. Supportive Care in Cancer 2012; 20(6): 1193-1201.
21. Carpentier MY, Fortenberry JD. Romantic and sexual relationships, body image, and fertility in adolescent and young adult testicular cancer survivors: a review of the literature. Journal of Adolescent Health 2010; 47(2): 115-125.
22. Nathan PC, Hayes-Lattin B, Sisler JJ, Hudson MM. Critical issues in transition and survivorship for adolescents and young adults with cancers. Cancer 2011; 117(10 Suppl): 2335-2341.
23. Albritton KH, Phillips M, Siegel SE (Eds). Cancer in Adolescents and Young Adults. Berlin: Springer-Verlag, 2007.
24. Hewitt M, Greenfield S, Stovall E. From cancer patient to cancer survivor: lost in transition. Washington, DC: The National Academies Press, 2006.
25. Miedema B, Easley J, Robinson L. Do current cancer follow-up care practices meet the needs of young adult cancer survivors in Canada? A qualitative inquiry. Current Oncology 2013; 20(1): 14-22.
26. Government of New Brunswick. New Brunswick Regional Profiles. Department of Post Secondary Education, Traning and Labour. Fredericton, NB: Government of New Brunswick, 2012.
27. Editors. What should the age range be for AYA oncology? Journal of Adolescent and Young Adult Oncology 2011; 1(1): 3-10.
28. Wu X, Groves FD, McLaughlin CC, Jemal A, Martin J, Chen VW. Cancer incidence patterns among adolescents and young adults in the United States. Cancer Causes & Control 2005; 16(3): 309-320.
29. Creswell JW. Research design: qualitative, quantitative, and mixed methods approaches, 2nd edn. Thousand Oaks, CA: Sage Publications Inc., 2003.
30. Creswell JW. Qualitative inquiry and research design: choosing among five traditions. Thousand Oaks, CA: Sage Publications Inc., 1998.
31. Mills J, Bonner A, Francis K. The development of constructivist grounded theory. International Journal of Qualitative Methods 2006; 5(1): 1-9.
32. Denzin N, Lincoln YS (Eds). Handbook of Qualitative Research, 2nd edn. Thousand Oaks, CA: Sage Publications Inc., 2000.
33. Busby S, Witucki-Brown J. Theory development for situational awareness in multi-casualty incidents. Journal of Emergency Nursing 2011; 37(5): 444-452.
34. Hubbard G, Kidd L, Kearney N. Disrupted lives and threats to identity: the experiences of people with colorectal cancer within the first year following diagnosis. Health (London) 2010; 14(2): 131-146.
35. Charmaz K. Loss of self: a fundamental form of suffering in the chronically ill. Sociology of Health and Illness 1983; 5(2): 168-195.
36. Charmaz K. 'Discovering' chronic illness: using grounded theory. Social Science & Medicine 1990; 30(11): 1161-1172.
37. Charmaz K. Grounded theory: objectivist and constructivist methods. In: N Denzin, YS Lincoln (Eds). Handbook of Qualitative Research, 2nd edn. Thousand Oaks, CA: Sage Publications Inc., 2000.
38. Statisics Canada. Highest level of education attained by province. Ottawa, ON: Government of Canada, 2009.
39. Statisics Canada. Median total income, by family type, by province and territory. Ottawa, ON: Government of Canada, 2012.
40. Tjemsland L, Soreide JA. Operable breast cancer patients with diagnostic delay - oncological and emotional characteristics. European Journal of Surgical Oncology 2004; 30(7): 721-727.
41. Yu CK, Chiu C, McCormack M, Olaitan A. Delayed diagnosis of cervical cancer in young women. Journal of Obstetrics and Gynaecology 2005; 25(4): 367-370.
42. Miedema B, Easley J, Hamilton R. Young adults' experiences with cancer: comments from patients and survivors. Canadian Family Physician 2006; 52(11): 1446-1447.
43. OECD. Health at a Glance 2009: OECD Indicators. Paris: Development Organization of Economic Cooperation, 2009.
44. Mathews M, West R, Buehler S. How important are out-of-pocket costs to rural patients' cancer care decisions? Canadian Journal of Rural Medicine 2009; 14(2): 54-60.
45. Miedema B, Easley J, Fortin P, Hamilton R, Mathews M. The economic impact on families when a child is diagnosed with cancer. Current Oncology 2008; 15(4): 173-178.
46. Reid-Arndt SA, Cox CR. Does rurality affect quality of life following treatment for breast cancer? Journal of Rural Health 2010; 26(4): 402-405.
47. Girgis A, Boyes A, Sanson-Fisher RW, Burrows S. Perceived needs of women diagnosed with breast cancer: rural versus urban location. Australian and New Zealand Journal of Public Health 2000; 24(2): 166-173.
48. Green E, Gorman E, Bartlett K, Brooks L, Struthers C. Canada: an external quality review process - the Wellspring model. Journal for Healthcare Quality 2005; 27(3): 15-21.
49. Sabiston CM, McDonough MH, Crocker PR. Psychosocial experiences of breast cancer survivors involved in a dragon boat program: exploring links to positive psychological growth. Journal of Sport & Exercise Psychology 2007; 29(4): 419-438.
50. Stearns SC, Slifkin RT, Edin HM. Access to care for rural Medicare beneficiaries. Journal of Rural Health 2000; 16(1): 31-42.
51. Miedema B, Hamilton R, Lambert-Lanning A, Tatemichi SR, Lemire F, Manca D, et al. Prevalence of abusive encounters in the workplace of family physicians: a minor, major, or severe problem? Canadian Family Physician 2010; 56(3): e101-108.

