Introduction
The increasing prevalence of obesity, and detrimental health behaviours of high dietary energy intakes and low physical activity levels have been causing concern in Western countries, including New Zealand. In 2003, 21% of New Zealand adults over the age of 15 years were obese, an increase from the 17% recorded in 19971. For New Zealand children, there is also a trend of increasing obesity2,3, with reports of 21% of children being overweight and 9.8% being obese in the latest national survey4. The impact of rising obesity levels in the population is a cause for great concern.
The relationship between obesity, energy intakes and inactivity in disease prevalence is well documented5. Considerable effort has been devoted to understanding the underlying determinants of obesity and its associated health behaviours. For example, physical inactivity is associated with age, sex, socioeconomic status and ethnicity6,7. One determinant that has recently been gaining attention is location of residence, that is, rural or urban dwelling.
There are a number of reports that rural dwelling adults have higher levels of obesity and lower levels of physical activity than their urban counterparts8-10, although the findings for physical activity may not be generalisable10. There is mixed and contradictory evidence of rural-urban differences in children. A cross-sectional study11 of American adolescents found that rural children had higher obesity levels (16.5%) compared with urban children (14.3%). This study also found that urban children had higher levels of physical inactivity compared with rural children. This finding is supported by a study of 8-12 year old children from a Midwestern state of America where more rural children were overweight (25.1%) than urban children (19.4%), and urban children were less active. Furthermore a cross-sectional study of Canadian 11-15 year olds found that levels of overweight and obesity increased with increasing 'rurality'12. No rural-urban difference was reported in this Canadian study for physical activity participation, although rural children had lower levels of 'screen time' (television and video watching and computer use). Rural-urban differences in physical activity participation have been reported for Australian adolescent girls in the summer but not the winter; no differences were found for boys13.
There are no reports referenced on Medline comparing obesity, and associated health behaviours such as energy intake and physical activity levels between rural and urban New Zealand children. There is, however, a report indicating rural New Zealand children are leaner than urban children14. The objective of the current study was to further document and investigate rural-urban differences in body composition, energy intake, physical activity and screen time in New Zealand children.
Methods
This study reports on data collected in a large national cross-sectional population survey of 5-15 year-old New Zealanders (the 2002 National Children's Nutrition Survey, CNS02). Full details of survey methods have been provided elsewhere4.
The survey used a two-stage sampling procedure whereby schools were randomly selected and invited to participate; children were then randomly selected from participating schools. For reasons of cost, some schools were excluded (schools with <50 students, correspondence schools and schools on a remote island). Overall 172 schools agreed to take part in the study, representing a 91% response rate. The total number of children recruited was 4728, of which 3275 finally participated (response rate 69%: 2792 urban children [85.3%], 483 rural children [14.7%]). In the CNS02 study, 'urban' was classified as any school located in either a main urban or secondary urban area, while 'rural' was classified as any school located in either a minor urban or rural area according to Statistics New Zealand geo-coding criteria4.
Data for this study were collected from Food Habits and Physical Activity interviews, the 24 hour diet recall questionnaires, and standard anthropometric measurements4. Most of the interviews were undertaken at the children's homes in the presence of their parent or guardian. The data from the 24 hour diet recall questionnaires were used to determine energy intake4. Physical activity recall data from the previous 7 days were collected using the Physical Activity Questionnaire for Children (PAQ-C)4. The PAC-Q asked children to complete a range of questions relating to the type, frequency and intensity of physical activity undertaken at various times during the past week. The survey contained 8 physical activity questions relating to sport activities/games (such as whether they participated in netball, soccer or dancing), physical activity undertaken at morning recess, lunch time, after school and in the evenings as well as weekend physical activity. The data relating to weekly physical activity performed are the sum of all the sports or games reported as being undertaken during the week and then divided by the number of activities to determine a mean weekly frequency score. Data were assigned an arbitrary unit based on the following Likert scale (1, 0 times; 2, 1-2 times; 3, 3-4 times; 4, 5-6 times; 5, ≥7 times per week).
In addition, a series of questions also asked children to report their screen viewing behaviour (television or video viewing or playing video or computer games). Children reported on viewing frequency (number of days) and duration (number of hours). The time reported for each of the behaviours was summed to get an accumulated screen time and then assigned an arbitrary unit based on the following scale (1, <1 hour; 2, 1-2 hours; 3, 3-4 hours; 4 >4 hours per week).
For anthropometric measurements children wore light clothing and no shoes. Measurements were taken at school by research assistants. Height was measured with a portable stadiometer. Two measurements were made to the nearest 0.1 cm. If these measurements differed by more than 0.5 cm, a third measurement was taken and the mean of the closest two measurements was recorded. Weight was measured on Seca scales (Seca; Hamburg, Germany) to the nearest 0.1 kg. The scales were calibrated with a standard weight prior to use. Two measurements were taken and if these differed by more than 0.5 kg a third measurement was taken and the mean of the closest two measurements was recorded. Triceps (mid-point in the length of the horizontal line between the acromion process and the tip of the olecranon) and subscapular (inferior angle of scapula) skinfold thicknesses were measured with Holtain callipers (Crymych, UK) to the nearest 0.1 mm. Two measurements were made at each site and if they differed by more than 0.5 mm a third was taken and the mean of the closest two measurements was recorded. Similarly, two waist (highest point of the iliac crest during minimal respiration) and arm (mid-point in the length of the horizontal line between the acromion process and the tip of the olecranon) circumferences were measured to the nearest 0.1 cm at each site. If these differed by more than 0.5 cm a third measurement was taken and the mean of the closest two measurements was recorded. All anthropometric measurements were made on the right hand side of the body whenever possible, and the final value was calculated as the mean of the two closest measurements.
Demographic data including ages and ethnicity were collected during the Food Habits and Physical Activity interviews, as was the residential address used to determine socioeconomic status using the New Zealand Deprivation Index (NZDep)4.
The large National Children's Nutrition Survey dataset obtained from the Ministry of Health as a Microsoft Excel spreadsheet was initially transferred to the Statistical Analysis System v9.1.3 (SAS Inst; Cary, NC, USA) for further analysis. Changes in the means of variables between the urban and rural groups were estimated using a mixed modelling procedure (Proc Mixed) within the SAS software. To control for the confounders of ethnicity and socioeconomic status, these two variables and their interactions were added to the model statement as covariates. In addition, nominal variables representing the proportion of children (and subgroups) meeting overweight and obesity cut-offs were compared by categorical modelling using the proc freq procedure in SAS. A type I error of 5% was chosen for declaration of statistical significance; precision of estimates were represented by the 95% confidence limits (CL, the likely range of true value).
Ethics approval
Ethics approval for the original study was gained from all the thirteen New Zealand Regional Health Ethics Committees. Informed written consent was received from all parents or guardians of participating children.
Results
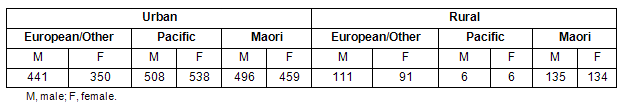
The proportion of differing ethnic groups in the sample population are shown (Table 1). An over-sampling technique produced approximately equal numbers of urban Maori, Pacific and European/other children, but did not sample equal numbers of Polynesian children compared with other ethnicities in rural areas. For this reason Pacific children were excluded from ethnicity comparisons of urban and rural groups.
Table 1: Ethnicity characteristics of sample population (subject number in each category)
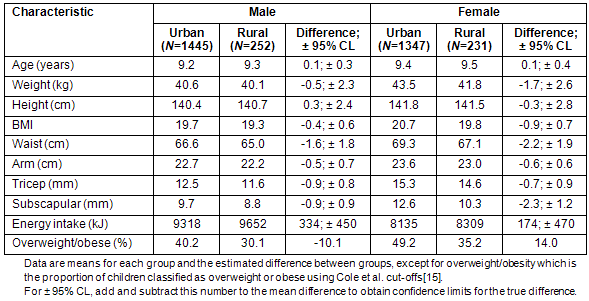
The physical characteristics of the urban and rural groups in this study are presented (Table 2). Although no significant effects were found with geographical location in age, weight, height or energy intake, significant differences were found in BMI, circumferences and skinfolds. Using well-established international cut-offs15 urban boys were found 1.3 times more likely to be overweight or obese than rural boys (95% confidence limits 1.1-1.6, p <0.01) and urban girls were 1.4 times more likely to be overweight or obese than rural girls (95% CL 1.2-1.7, p <0.01).
Table 2: Participant characteristics
Differences in body composition (sum of triceps and subscapular skinfolds), physical activity, screen time (television and computer/video game usage) and energy intake between the two geographical groups is shown (Table 3). Urban children had significantly higher levels of subcutaneous fat than rural children, which is evident in both males (approximately 8% higher) and females (approximately 14% higher). Children from rural areas tended to have lower subcutaneous fat levels regardless of ethnicity, age or socioeconomic status. Indeed, when the analysis was adjusted to account for differences in ethnic make-up and socioeconomic status of children from the two groups, the difference in the overall sum of skinfolds between the groups remained (urban 23.9 mm, rural 19.9 mm, difference -4.0 mm, p = 0.01). Significant interactions were found between sum of skinfolds, ethnicity (p = 0.004) and socioeconomic status (p = 0.05).
Overall weekly physical activity was similar in rural and urban children; however, differences were found between children in the various subgroups. Rural children identified as European/other, or from higher socioeconomic families were more active than their urban counterparts. Conversely, urban Maori children were more active than rural Maori children. Accounting for ethnicity or socioeconomic status in the model had little effect on weekly physical activity with no significant interactions found. Overall screen time tended to be lower in the rural children; however, this was only statistically significant in children aged 5-7 years. Accounting for ethnicity and socioeconomic status decreased the total screen time in the rural group to 2.3 but had little effect on the urban groups' screen time (2.8). Therefore, after accounting for these confounders it was found that rural children watched significantly less total screen time than urban children (p = 0.02).
Table 3: Differences in body composition, physical activity, screen time and energy intake between urban and rural children
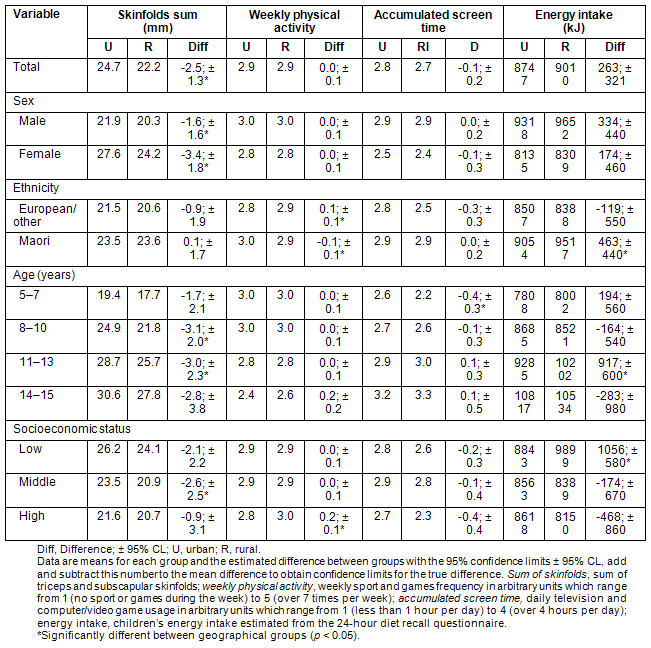
Energy intake tended to be similar between the two groups, apart from rural Maori, rural children aged 11-13 years, and children from rural low socioeconomic families who all had significantly higher energy intakes than their urban counterparts. Accounting for the confounding variables of ethnicity and socioeconomic status had little effect on total energy intakes between the groups with no significant interactions found.
Discussion
Obesity and body composition
The current study reports rural and urban differences in body composition, energy intake, physical activity and screen time for New Zealand children. The study found that rural children had a significantly lower BMI, smaller waist circumferences and thinner skinfold measurements than urban children. This finding is similar to a previous New Zealand report that found rural children had lower percent body fat in 5 year-olds and smaller waist circumferences and BMIs in 10 year-olds compared with their urban counterparts14. This finding is also similar to a cross-sectional study of body composition in Turkish children, which found that urban children had higher tricep, subscapular and supralliac skinfold thicknesses than rural children, as well as being significantly taller and heavier16. In addition, a study reporting secular trends in China and Brazil has reported a higher prevalence of overweight in children in urban areas, although these findings may reflect the changes in economic development in these countries17.
There is, however, research that shows a different relationship. Lewis et al. studied the prevalence and extent of obesity in school age children from Georgia, USA, and found that children in rural areas had a higher prevalence of overweight than those in the suburban and urban areas18. Similarly, McMurray et al. found that rural children have a significantly higher BMI, sum of skinfolds and a greater proportion of rural children were obese19. Studies outside the USA have also generally found that rural children had a higher prevalence of obesity than urban children12,17,20-25.
The disparity between the results of the current study and other reports could be related to the demographic differences in rural and urban populations in the different studies. This is because socioeconomic status and ethnicity are correlates of obesity26 and it is possible that the rural-urban differences found are reflections of the demographics of the populations. Despite controlling for ethnicity and socioeconomic status, the current study found that rural children were leaner than their urban counterparts. Other studies report the converse finding, despite controlling for socioeconomic status11,12 and ethnicity11. Other explanations for the reported rural-urban differences in body composition could be differences in the physical or social environment12. These potential environmental differences warrant further investigation because this may provide useful insights in terms of obesity interventions.
Physical activity
The current study did not find any differences in the frequency of bouts of physical activity but it was found that rural children had lower screen time. However, some studies have found physical activity differences. In 1996, urban dwelling Cameroon children tended to be more sedentary and total physical activity scores were two times higher in the rural children27. These results contrast with an Icelandic study that found rural students were involved in less strenuous and more sedentary leisure time activities28.
These conflicting results have a number of possible explanations. First, differences could be due to demographic differences such as socioeconomic status or ethnicity, as mentioned previously. This highlights the importance of controlling for these variables in studies of rural and urban differences in obesity or physical activity. Second, in large population studies like the present one, physical activity is usually measured by self-report. Self-reports are an easy and efficient method of data collection but may lack accuracy. An issue to bear in mind when interpreting self-reported physical activity data for urban and rural residents is that urban and rural people are likely to interpret physical activity differently. While physical activity is often seen as a leisure or recreation time activity for urban residents, it is often associated with work for rural residents (eg farm work) and, therefore, may not be reported as physical activity29.
Those studies that found urban children to have higher levels of physical activity may have had easier access to interventions such as community sports programs or sports facilities28. Those studies that found rural children to be more active may be due to an increased opportunity to spend time outdoors for rural children29,30.
There are a number of limitations to this study. The study population is not representative, in that there were high proportions of Maori and Pacific (particularly urban Pacific) children sampled. To overcome this non-representation, differences between urban and rural children were controlled for ethnicity. In addition, rural children form a relatively small percentage (13.7%) of the sample and they could have been oversampled. The study also excluded very small schools and very remote areas and this may have influenced the results. The study used only two skinfold measurements and a self-reported measure of physical activity frequency. Differences between the rural and urban children may have been revealed if activity frequency and duration had been measured.
Conclusion
Differences were found in body composition, with rural children being leaner than urban children (despite no differences in energy intakes and frequency of physical activity). This New Zealand finding is different from other Western countries and may be due to differences in the physical and social environment. More research is required to understand these potential environmental rural-urban differences because this may provide useful insights in terms of obesity interventions.
Acknowledgements
The authors thank the Ministry of Health for approving the use of this data for this research report. The New Zealand Health Research Council funded a Summer Research Studentship for Finn Peters to complete this project. Thanks also to Neil Pearce, Director, Centre for Public Health Research, Massey University for his supervision of Emma Hodgkin.
References
1. Ministry of Health. Tracking the obesity epidemic: New Zealand 1977-2003. Wellington: Ministry of Health, 2004.
2. Dawson KA, Hamlin MJ, Ross JJ, Duffy DF. Trends in the health-related physical fitness of 10-14 year old New Zealand children. Journal of Physical Education New Zealand 2001; 34(1): 26-39.
3. Albon HM, Hamlin MJ, Ross JJ. Secular trends and distributional changes in health and fitness performance variables of 10-14 year old New Zealand children between 1991 and 2003. British Journal of Sports Medicine 2010; 44(4): 256-262.
4. Parnell W, Scragg R, Wilson N, Schaaf D, Fitzgerald E. NZ Food: NZ Children: Key Results of the 2002 National Children's Nutrition Survey. Wellington: Ministry of Health, 2003.
5. Bauman A. Updating the evidence that physical activity is good for health: an epidemiological review 2000-2003. Journal of Science and Medicine in Sport 2004; 7(1): 6-19.
6. Van Der Horst K, Chin MJ, Paw A, Twisk JWR, Van Mechelen W. A brief review on correlates of physical activity and sedentariness in youth. Medicine and Science in Sports and Exercise 2007; 39(8): 1241-1250.
7. Biddle SJH, Whitehead SH, O'Donovan TM, Nevill ME. Correlates of participation in physical activity for adolescent girls: a systematic review of recent literature. Journal of Physical Activity and Health 2005; 2(4): 423-434.
8. Parks SE, Housemann RA, Brownson RC. Differential correlates of physical activity in urban and rural adults of various socioeconomic backgrounds in the United States. Journal of Epidemiology and Community Health 2003; 57(1): 9-35.
9. Patterson PD, Moore CG, Probst JC, Shinogle JA. Obesity and physical inactivity in rural America. Journal of Rural Health 2004; 20(2): 151-159.
10. Martin SL, Kirkner GJ, Mayo K, Matthews CE, Durstine JL, Herbert JR. Urban, rural, and regional variations in physical activity. Journal of Rural Health 2005; 21(3): 239-244.
11. Lui J, Bennett KJ, Harun N, Probst JC. Urban-rural differences in overweight status and physical inactivity among US children aged 10-17 years. Journal of Rural Health 2008; 24(4): 407-415.
12. Bruner MW, Lawson J, Pickett W, Boyce W, Janssen I. Rural Canadian adolescents are more likely to be obese compared with urban adolescents. International Journal of Pediatric Obesity 2008; 3(4): 205-211.
13. Booth ML, Okely AD, Chey T, Bauman AE, Macaskill P. Epidemiology of physical activity participation among New South Wales school students. Australian and New Zealand Journal of Public Health 2002; 26(4): 371-374.
14. Graham D, Reed P, Ayers K, Amey J, Clinton J, Appleton S et al. Project Energise. Happy healthy children of all shapes and sizes. Hamilton: Waikato District Health Board, 2008.
15. Cole TJ, Bellizzi M, Flegal K, Dietz WH. Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 2000; 320(7244): 1240-1243.
16. Ozdirenc M, Ozcan A, Akin F, Gelecek N. Physical fitness in rural children compared with urban children in Turkey. Pediatrics International 2005; 47(1): 26-31.
17. Wang Y, Monteiro C, Popkins BM. Trends of obesity and underweight in older children and adolescents in the United States, Brazil, China and Russia. American Journal of Clinical Nutrition 2002; 75(6): 971-977.
18. Lewis RD, Meyer MC, Lehman SC, Trowbridge FL, Bason JJ, Yurman KH et al. Prevalence and degree of childhood and adolescent overweight in rural, urban, and suburban Georgia. Journal of School Health 2006; 76(4): 126-132.
19. McMurray RG, Harrell JS, Bangdiwala SI, Deng S. Cardiovascular disease risk factors and obesity of rural and urban elementary school children. Journal of Rural Health1999; 15(4): 65-374.
20. Borders TF, Rohrer JE, Cardarelli KM. Gender-specific disparities in obesity. Journal of Community Health 2006; 31(1): 57-68.
21. Jackson JE, Doescher MP, Jerant AF, Hart LG. A national study of obesity prevalence and trends by type of rural county. Journal of Rural Health 2005; 21(2): 140-148.
22. Kettle SM, Roebotha BV, West R. Prevalence of specific cardiovascular disease risk factors in young Newfoundland and Labrador adults living in urban and rural communities. Canadian Journal of Rural Medicine 2005; 10(2): 81-85.
23. Rasmussen F, Johansson M, Hansen HO. Trends in overweight and obesity among 18-year-old males in Sweden between 1971 and 1995. Acta Paediatrica 1999; 88(4): 431-437.
24. Reeder BA, Chen Y, Macdonald SM, Angel A, Sweet L. Regional and rural-urban differences in obesity in Canada. Canadian Heart Health Surveys Research Group. Canadian Medical Association Journal1997; 157(Suppl1): S10-16.
25. Woodward DR, Cumming FJ, Ball PJ, Williams HM, Hornsby H, Boon JA. Urban-rural differences in dietary habits and influences among Australian adolescents. Ecology of Food and Nutrition 2000; 39(4): 271-292.
26. Wang Y, Beydoun MA. The obesity epidemic in the United States-gender, age, socioeconomic, racial/ethnic, and geographic characteristics: a systematic review and meta-regression analysis. Epidemiologic Reviews 2007; 29(1): 6-28.
27. Proctor MH, Moore LL, Singer MR, Hood MY, Nguyen US, Ellison RC. Risk profiles for non-communicable diseases in rural and urban schoolchildren in the Republic of Cameroon. Ethnicity & Disease 1996; 6(3-4): 235-243.
28. Kristjansdottir G, Vilhjalmsson R. Sociodemographic differences in patterns of sedentary and physically active behaviour in older children and adolescents. Acta Paediatrica 2001; 90(4): 429-435.
29. Potvin L, Gauvin L, Nguyen NM. Prevalence of stages of change for physical activity in rural, suburban and inner-city communities. Journal of Community Health 1997; 22(1): 1-13.
30. Loucaides CA, Chedzoy SM, Bennett N. Differences in physical activity levels between urban and rural school children in Cyprus. Health Education Research 2004; 19(2): 138-147.
