Context
Injection safety has improved in Africa largely due to efforts to prevent HIV transmission. In some rural clinics and remote pharmacies, however, disposable syringes are still re-used, although heat sterilization damages the plastic ring at the base of the needle and is limited by the availability of fuel. A study of HIV transmission risks in Mbeya, Tanzania revealed that 33% of sterilized equipment prepared for re-use was contaminated with bacteria, introduced by handling the needle in preparing the next injection1. This finding has serious implications for patient safety when IV injections are administered for the treatment of malaria. In a prospective study of patients receiving venous injections with catheters that were culture positive on removal, 61% developed a bloodstream infection2. However, low recognition of resulting secondary infections is to be expected in rural areas, because sepsis and malaria are indistinguishable by clinical examination; and WHO guidelines for managing childhood illnesses typically fail to identify 50% of bacteremia cases in Africa3. The purpose of this review was to explore the clinical significance of a traditional proscription of injections ('I know if I take my convulsed child to hospital they may inject him and this practice is known to kill children') in treating cerebral malaria (known as degedege in Tanzania4). The hypothesis of this review was that the belief that injections will kill a child with degedege arises from the occurrence of injection associated infections resulting in fatal sepsis, and that recognizing and redressing problems with infection control may improve parents' and traditional healers' willingness to refer sick children for quinine injections.
Issue
A systematic search of the peer reviewed literature was completed in January 2010 using Medline and the search terms 'sepsis', 'septicemia', 'bacteremia', 'catheter', 'nosocomial' and 'degedege', together with the search term 'Tanzania'. All English-language articles published since 1995 were screened (167 records), and the reference lists of relevant articles were reviewed, identifying a further 9 articles. Only 25 articles met the inclusion criteria by providing quantitative or qualitative data on the incidence of nosocomial bloodstream infections or the nature of degedege. The authors of one article with an aberrant description of degedege were contacted for clarification. Five articles that did not meet the inclusion criteria were deemed relevant because they provide information on the management of malaria and diseases misdiagnosed as malaria in rural Tanzania. Three additional searches were performed to assess the incidence of bacteremia in African patients receiving treatment via an IV line, the incidence of bacteremia in vaccine trials, and the mortality rate for nosocomial bacteremia in developing country hospitals; and to review all records on nosocomial bacteremia in developing countries, and adverse events in vaccine trials in Africa.
Currently, oral medication is prescribed for uncomplicated malaria in Tanzania, but severe malaria still requires parenteral therapy because convulsions are often accompanied by weakness or vomiting. This presents a particular problem in Tanzania because a fever with convulsions is popularly understood in rural areas as degedege, believed to be a condition that must never be treated by injection5,6. The defining symptoms of degedege are those of cerebral malaria, with additional symptoms that include rolling eyes, a stiff body, lethargy, no urine, and no stool, cold feet, babbling, shouting, and frothing at the mouth7,8. These additional symptoms are mentioned only by some traditional healers, however, and all the symptoms universally attributed to degedege according to traditional definitions are the symptoms of cerebral malaria.
Degedege is considered to be a spiritual malady associated with the spirit of a bird, and it is not traditionally associated with malaria in Tanzania, for which biomedical treatment is usually sought promptly by the same parents who would go first to a traditional healer to treat a child with convulsions9,10. A similar proscription of treating a child with convulsions by injection exists in Burkina Faso6; whereas in Ghana and Nigeria, cerebral malaria with convulsions is also attributed to the spirit of a bird, but no similar proscription of injections exists6. In Cameroon degedege ('Idalgdalgui') is understood to be malaria and hospital treatment is believed to be effective11.
Tanzanian mothers interviewed in some studies testified to the belief that injections will kill a child with degedege, and attributed this to their own experience8. Individuals who had not witnessed a sudden death in hospital following antimalarial injections had heard of the occurrence. One mother said, 'I could not take her to hospital as in the hospital they treat by injecting drugs, the practice which results to sudden death of children under such treatment'12. A traditional birth attendant who would refer patients with uncomplicated malaria for biomedical care reported that she would not refer a child with convulsions, because she believed that injections are fatal for this condition12. In other Tanzanian settings, some traditional healers and rural mothers have been persuaded that malaria causes degedege and that antimalarials should be a first-line treatment9. That this transition has been difficult to achieve has been attributed to an irrational fear arising from the disappointment of parents who brought their child to the hospital too late, only to see the child die on arrival8.
In reality, sudden illness from an injection is a probability in unsterile conditions; the incidence of nosocomial bacteremia in patients receiving treatment via an IV line is reported to be as high as 54% in prospective studies of African hospitals, and of these infections 71% were matched to a culture from the IV catheter tip at the hospital recording the highest incidence. Table 1 presents the incidence of catheter-associated infections in five studies conducted in African hospitals. In many rural Tanzanian communities where injections are proscribed for degedege, the first line treatment for fever and convulsions is an IV infusion of quinine. Bloodstream infections in the developing world are usually associated with an IV line12,13. In the absence of any other source of infection, a clinical bloodstream infection after admission in a patient receiving IV therapy is, by definition, an injection associated infection14. In this instance the definition is unhelpful, because the clinical signs of a bloodstream infection are indistinguishable from the presenting condition of cerebral malaria. The causal association between IV lines and nosocomial bacteremia has been established in risk factor analyses, however. The unadjusted relative risk for a bloodstream infection in patients with a peripheral venous catheter in US hospitals was reported as 10.9 (95% CI 10.8-11.1)15. A similar study of risk factors for cephalosporin-resistant bacteremia in hospitalized patients in Madagascar found an unadjusted odds ratio of 7.5 (95% CI 1.0-59) for the presence of a venous catheter16.
Table 1: Incidence of catheter-associated nosocomial bacteremia in African hospitals17-21
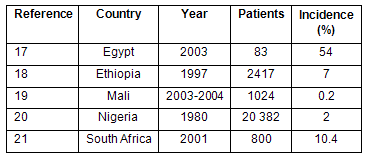
The incidence of nosocomial bacteremia is high in Tanzanian hospitals that have the facilities to diagnose bloodstream infections, explaining 6-8% of fevers at the national referral hospital in Dar es Salaam3,22,23. Nosocomial bloodstream infections develop in 1.5% of admissions to the intensive care unit24. In 2006, bacteremia caused more deaths in children than malaria at this hospital3.
The association between an IV line and secondary infection is captured in the experience of Temeke Hospital in Dar es Salaam, where the auto-disable syringe was introduced in 2005, ending the practice of routine syringe re-use. In a film produced as an informal intervention assessment, the chief medical officer reported that infections prevented by the auto-disable syringe saved the hospital approximately US$280 for a 7 day course of antibiotics, bed charges and intensive care to manage the symptoms of a serious systemic infection25. The average length of stay for non-surgical inpatients was significantly reduced by the injection safety intervention alone. Remarkably, before the injection safety intervention's impact was immediately apparent, injections were not considered a serious risk factor for infection at this hospital, as the background rate of secondary infections was not then appreciated.
The infection risk from an IV line is greater than from an IV injection, because the device remains in place for days at a time. The risk from an IV line is comparable to that from a contaminated central venous catheter (61% probability of infection), and can be adjusted by the rate ratio of infections per catheter-day for peripheral venous catheters, compared with central venous catheters (0.5:2.7)2,26. This adjustment gives an 11.3% probability of infection if injection equipment is contaminated with bacteria. From the contamination rate observed in Mbeya, Tanzania when injection equipment was being boiled and re-used, the incidence of nosocomial bacteremia should be 3.7% for malaria patients with an IV line, where injection equipment re-use is practiced1. This estimate is somewhat higher than the average observed incidence of catheter-associated nosocomial bacteremia in African hospitals (2.9%).
An ordinary IV injection should be less likely to lead to infection than an IV line. The incidence of HIV in drug users who inject intravenously has been used to estimate the risk of nosocomial HIV transmission in an unsafe medical injection27. The per-injection risk of a serious bacterial infection can also be estimated from the experience of injecting drug users, who learn aseptic technique to avoid a 'dirty hit,' a febrile infection felt by most users immediately after an injection27. Among febrile injecting drug users admitted to a hospital in Baltimore, USA, 25% had culture-confirmed bloodstream infections28. Among 94 cohorts of injecting drug users interviewed from 1999 to 2008 in Australia, Ireland, and Viet Nam using the Illicit Drug Reporting System, the average incidence of a 'dirty hit' over one month of daily injecting was 21% (range 14-54%). These reports support a per-injection risk of 0.2% in unsafe IV injections.
The search for records of sepsis in vaccine trials anywhere in Africa identified only two studies that reported cases of fatal sepsis classified as vaccine-related adverse events. One rotavirus trial reported two cases of fatal sepsis (0.06% incidence)29. One trial in a review of trials of preventive treatment for malaria recorded a 0.6% incidence of fatal sepsis but did not report the nature of other hospitalizations (12% of infants were hospitalized within 8 weeks of treatment) that may have been related to the intervention30. These findings suggest that IV injections for the treatment of malaria carry a much greater risk of secondary infection than ordinary medical injections.
Table 2: Crude and attributable mortality from nosocomial bacteremia in developing country hospitals22,31-35
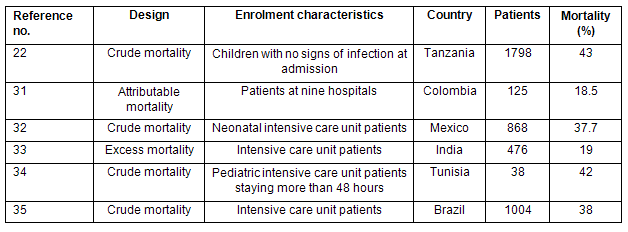
The US CDC estimates that 15% of episodes of bacteremia are fatal, but the attributable mortality rate for nosocomial bacteremia in developing countries may be somewhat higher36. Prospective studies into the mortality rate of patients with nosocomial bacteremia in developing countries are presented (Table 2). The crude mortality rate is high even in pediatric patients with no signs of infection on admission.
Lessons learned
The findings of this review suggest that the prevention of secondary infections through the prevention of injection equipment re-use may substantially improve the safety and acceptability of life saving injections for the treatment of malaria in rural Tanzania. The prevention of injection equipment re-use by informal-sector injection providers poses a special challenge. Outbreak investigations in the USA have shown that re-use of injection equipment without sterilization usually occurs when health workers mistakenly believe37 that:
- it is safe to reuse a syringe after changing the needle or after changing the syringe
- it is safe to reuse a needle or syringe on the same patient: for re-entering a multi-dose vial or saline bag; or when accessing an IV port separated from the patient by intervening IV tubing or heparin locks or valves
- it is safe to re-use finger-stick devices among different patients without sterilization
- cleaning injection equipment or shared devices is only necessary when blood or other contamination is visible.
These misconceptions are likely to be common among injection providers who have no formal training in injection safety.
Informal-sector health care plays a prominent role in the treatment of malaria in Tanzania. Treatment seeking for malaria was comprehensively reviewed in one household survey of mothers and guardians in an endemic area of Tanzania38. Of all individuals interviewed, 16% reported their children had experienced malaria in the previous 2 weeks, and 85% had sought treatment, most at a formal health facility but 27% at a drug store, and 30% at a general shop. The definition of malaria used in this survey included a traditional term sometimes understood to include a broader range of symptoms, such as respiratory or gastrointestinal illness (homa). The actual incidence of malaria is likely to be much lower than reported; however, the reliability of reported treatment-seeking behavior is considered good in retrospective surveys with a short recall interval.
The intensity of informal-sector treatment for malaria in Tanzania is evident from the high levels of chloroquine in the blood of patients presenting with malaria at hospitals and clinics in rural areas. Two studies evaluated levels of chloroquine already present in the blood of patients diagnosed with malaria. One study of primary healthcare facilities in eastern Tanzania (40 km north-west of Dar es Salaam) found that 98% of febrile children already had chloroquine in their blood when they presented for treatment39. A follow-up study at public health facilities in the same region found that of sick children given chloroquine at the health facilities, 85% had recently taken chloroquine40. Another study in the northern Highlands found that 51% of febrile patients seeking a malaria test had already taken antimalarials within the past 2 weeks.
In support of the Ministry of Health and Social Welfare policy on safe injections and healthcare waste management (effective from 1 May 2010) the Tanzania Food and Drugs Authority will no longer issue a permit for the importation of standard disposable syringes of 10 mL or less, unless these are meant for specific procedures, including blood drawing. Instead, only auto-disable syringes will be used for medical injections. The transition to importing only auto-disable syringes for ordinary medical injections is expected to improve injection safety at public, private and faith-based healthcare facilities, as well as in the informal sector.
Although fear of injections is not necessarily the most important barrier to seeking health care for fever with convulsions in Tanzania, injection fears in rural areas may reflect real risks associated with unsterile injections that can be redressed by improving infection control in the hospital. Other obstacles to receiving appropriate treatment for cerebral malaria must also be addressed. One study of treatment-seeking behavior in a rural area found that the most common explanation for a child's death due to delay in taking the child to a healthcare facility, was the perception that it was 'only ordinary fever', and not a severe episode of illness41. Other obstacles to obtaining timely treatment are a lack of access to transport and the high cost of professional medical care38.
Conclusion
The clinical significance of the traditional proscription of treating degedege with injections is comparable to parents' fears of injection-provoked paralysis, a rare but avoidable adverse event that can be prevented by safe injection technique. The estimated probability of infection when an IV injection is unsafe is 0.2%, and the estimated probability of infection when an IV infusion is unsafe is 3.7%1,2,26. The observed average incidence of catheter-related nosocomial bacteremia in African hospitals supports a per-injection risk of 2.9%17-21. Vaccines carry a much lower risk of secondary infections29,30. Nosocomial bacteremia has an attributable mortality rate of 19% in developing country hospitals, and accounted for more deaths in children than malaria at the national referral hospital in Tanzania in 20063,31,33. Low recognition of secondary bacterial infections at rural hospitals and a focus on HIV prevention in injection safety interventions have led to a general neglect of these serious adverse events, despite parents' fears that IV injections may kill a child. Attention to aseptic technique should be paramount where HIV transmission risks have been eliminated, as bacterial secondary infections can also be fatal. Bloodstream infections are a major burden on Tanzanian hospitals and their prevention through the use of auto-disable syringes may improve malaria treatment referrals in rural areas.
References
1. Hoelscher M, Riedner G, Hemed Y, Wagner H, Korte R, von Sonnenburg F. Estimating the number of HIV transmission through reused syringes and needles in the Mbeya Region, Tanzania. AIDS 1994; 8: 1609-1615.
2. Park K-H, Kim S-H, Song EH, Jang E-Y, Lee EJ, Chong YP et al. Development of bacteremia of fungaemia after removal of colonized central venous catheters in patients with negative concomitant blood cultures. Clinical Microbiology and Infection 2010; 16(6): 747-746.
3. Blomberg B, Manji K, Urassa W, Tamim B, Mwakagile D, Jureen R, Msangi V et al. Antimicrobial resistance predicts death in Tanzanian children with bloodstream infections: a prospective cohort study. BMC Infectious Diseases 2007; 7: 43.
4. Muela S, Ribera J, Mushi A, Tanner M. Medical syncretism with reference to malaria in a Tanzanian community. Social Science & Medicine 2002; 55: 403-413.
5. Adongo P, Kirkwood B, Kendall C. How local community knowledge about malaria affects insecticide-treated net use in northern Ghana. Tropical Medicine and International Health 2005; 10(4): 366-378.
6. Beiersmann C, Sanou A, Wladarsch E, De Allegri M, Kouyate B, Muller O. Malaria in rural Burkina Faso: local illness concepts, patterns of traditional treatment and influence on health-seeking behaviour. Malaria Journal 2007; 6: 106.
7. Minja H, Schellenberg J, Mukasa O, Nathan R, Abdulla S, Mponda H et al. Introducing insecticide-treated nets in the Kilombero Valley, Tanzania: the relevance of local knowledge and practice for an Information, Education and Communication (IEC) campaign. Tropical Medicine and International Health 2001; 6(8): 614-623.
8. Comoro C, Nsimba S, Warsame M, Tomson G. Local understanding, perceptions and reported practices of mothers/guardians and health workers on childhood malaria in a Tanzanian district - implications for malaria control. Acta Tropica 2003; 87: 305-313.
9. Dillip A, Hetzel M, Gosoniu D, Kessy F, Lengeler C, Mayumana I et al. Socio-cultural factors explaining timely and appropriate use of health facilities for degedege in south-eastern Tanzania. Malaria Journal 2009; 8: 144.
10. Muela S. Community understanding of malaria, and treatment-seeking behaviour, in a holoendemic area of southeastern Tanzania (PhD thesis). Basel, Switzerland: Universität Basel; 2000.
11. Einterz E. Perceptions of malaria transmission, presentation and management in northern Cameroon. Transactions of the Royal Society of Tropical Medicine and Hygiene 2003; 97(1): 51-52.
12. Makundi E, Malebo H, Mhame P, Kitua A, Warsame M. Role of traditional healers in the management of severe malaria among children below five years of age: the case of Kilosa and Handeni Districts, Tanzania. Malaria Journal 2006; 5: 58.
13. Zaidi A, Huskins W, Thaver D, Bhutta Z, Abbas Z, Goldmann D. Hospital-acquired neonatal infections in developing countries. Lancet 2005; 365: 1175-1188.
14. Bouza E, Alvarado N, Alcala L, Perez M, Rincon C, Munoz P. A randomized and prospective study of 3 procedures for the diagnosis of catheter-related bloodstream infection without catheter withdrawal. Clinical Infectious Diseases 2007; 44: 820-826.
15. Al-Rawajfah O, Stetzer F, Hewitt J. Incidence of and risk factors for nosocomial bloodstream infections in adults in the United States, 2003. Infection Control and Hospital Epidemiology 2009; 30(11): 1036-1044.
16. Randrianirina F, Vaillant L, Ramarokoto C, Rakotoarijaona A, Andriamanarivo M, Razafimahandry H et al. Antimicrobial resistance in pathogens causing nosocomial infections in surgery and intensive care wards in Antananarivo, Madagascar. Journal of Infection in Developing Countries 2010; 4(2): 74-82.
17. Bakr A. IV lines-related sepsis in newborn babies admitted to NICU in a developing country. Journal of Tropical Pediatrics 2003; 49(5): 295-297.
18. Ghiorghis B. Neonatal sepsis in Addis Ababa, Ethiopia: a review of 151 bacteremic neonates. Ethiopian Medical Journal 1997; 35(3): 169-176.
19. Koumare A, Sissoko F, Diop A, Ongoiba N, Maiga I, Bougoudogo F et al. [No title]. Mali Medicine 2008; 23(3): 44-46.
20. Anyiwo C, Daniel S, Ogunbi O, Aromolaran G. Nosocomial infections - a continuing danger to patients at Lagos University Teaching Hospital. Public Health 1980; 94: 229-234.
21. Van de Wetering M, Poole J, Friedland I, Caron H. Bacteremia in a paediatric oncology unit in South Africa. Medical and Pediatric Oncology 2001; 37(6): 525-531.
22. Blomberg B, Jureen R, Manji K, Tamim B, Mwakagile D, Urassa W et al. High rate of fatal cases of pediatric septicemia caused by gram-negative bacteria with extended-spectrum beta-lactamases in Dar es Salaam, Tanzania. Journal of Clinical Microbiology 2005; 43(2): 745-749.
23. Blomberg B, Mwakagile D, Urassa W, Maselle S, Mashurano M, Digranes A et al. Surveillance of antimicrobial resistance at a tertiary hospital in Tanzania. BMC Public Health 2004; 4: 45.
24. Ndugulile F, Jureen R, Harthug S, Urassa W, Langeland N. Extended spectrum β-Lactamases among Gram-negative bacteria of nosocomial origin from an intensive care unit of a tertiary health facility in Tanzania. BMC Infectious Diseases 2005; 5: 86.
25. Taylor S. Money saving movie. Safe Point Trust. (Online) 2005. Available: http://gallery.me.com/shauntaylor#100092 (Accessed 17 February 2010). (Film)
26. Maki D, Kluger D, Crnich C. The risk of bloodstream infection in adults with different intravascular devices: a systematic review of 200 published prospective studies. Mayo Clinic Proceedings 2006; 81(9): 1159-1171.
27. Dwyer R, Topp L, Maher L, Power R, Hellard M, Walsh N et al. Prevalences and correlates of non-viral injecting-related injuries and diseases in a convenience sample of Australian injecting drug users. Drug and Alcohol Dependence 2009; 100(1-2): 9-16.
28. Rothman R, Majmudar M, Kelen G, Madico G, Gaydos C, Walker T et al. Detection of bacteremia in emergency department patients at risk for infective endocarditis using universal 16S rRNA primers in a decontaminated polymerase chain reaction assay. Journal of Infectious Diseases 2002; 186: 1677-1681.
29. Madhi S, Cunliffe N, Steele D, Witte D, Kirsten M, Louw C et al. Effect of human rotavirus vaccine on severe diarrhea in African infants. New England Journal of Medicine 2010; 362(4): 289-298.
30. Aponte J, Schellenberg D, Egan A, Breckenridge A, Carneiro I, Critchley J et al. Efficacy and safety of intermittent preventive treatment with sulfadoxine-pyrimethamine for malaria in African infants: a pooled analysis of six randomized, placebo-controlled trials. Lancet 2009; 374: 1533-1542.
31. Moreno C, Rosenthal V, Olarte N, Gomez W, Sussmann O, Agudelo J et al. Device-associated infection rate and mortality in intensive care units of 9 Colombian hospitals: findings of the International Nosocomial Infection Control Consortium. Infection Control and Hospital Epidemiology 2006; 27(4): 349-356.
32. Perez-Gonzalez L, Ruiz-Gonzalez J, Noyola D. Nosocomial bacteremia in children: a 15-year experience at a general hospital in Mexico. Infection Control and Hospital Epidemiology 2007; 28(4): 418-422.
33. Mehta A, Rosenthal V, Mehta Y, Chakravarthy M, Todi S, Sen N et al. Device-associated nosocomial infection rates in intensive care units of seven Indian cities. Findings of the International Nosocomial Infection Control Consortium (INICC). Journal of Hospital Infection 2007; 67(2): 168-174.
34. Ben Jaballah N, Bouziri A, Mnif K, Hamdi A, Khaldi A et al. Epidemiology of hospital-acquired bloodstream infections in a Tunisian pediatric intensive care unit: a 2-year prospective study. American Journal of Infection Control 2007; 35(9): 613-618.
35. Girao E, Levin A, Basso M, Gobara S, Gomes L, Medeiros E et al. Trends and outcome of 1121 nosocomial bloodstream infections in intensive care units in a Brazilian hospital, 1999-2003. International Journal of Infectious Diseases 2008; 12(6): 145-146.
36. Wenzel R, Edmond M. The impact of hospital-acquired bloodstream infections. Emerging Infectious Diseases 2001; 7(2): 174-177.
37. Perz J, Thompson N, Schaefer M, Patel PR. US outbreak investigations highlight the need for safe injection practices and basic infection control. Clinics in Liver Disease 2010; 14: 137-151.
38. Njau J, Goodman C, Kachur S, Palmer N, Khatib R, Abdulla Set al. Fever treatment and household wealth: the challenge posed for rolling out combination therapy for malaria. Tropical Medicine and International Health 2006; 11(3): 299-313.
39. Nsimba S, Massele A, Eriksen J, Gustafsson L, Tomson G, Warsame M. Case management of malaria in under-fives at primary health care facilities in a Tanzanian district. Tropical Medicine and International Health 2002; 7(3): 201-209.
40. Mwanziva C, Shekalaghe S, Ndaro A, Mengerink B, Megiroo S, Mosha F et al. Overuse of artemisinin-combination therapy in Mto wa Mbu (river of mosquitoes), an area misinterpreted as high endemic for malaria. Malaria Journal 2008; 7: 22.
41. Kamat V. 'I thought it was only ordinary fever!' cultural knowledge and the micropolitics of therapy seeking for childhood febrile illness in Tanzania. Social Science & Medicine 2006; 62: 2945-2959.
