Introduction
Tuberculosis (TB) causes approximately 3 million deaths annually, worldwide. The disease is endemic in developing countries (DC), particularly among those most vulnerable, and presents a great public health problem1.
According to the World Health Organization (WHO), the incidence of TB in Ethiopia is 533/100 000 persons2. Extra-pulmonary forms comprise 10-20% of all TB cases and, of these, the cutaneous forms comprise 1.5%3,4. Cutaneous TB (CT) is a re-emerging pathology, following an initial decrease due to the diffusion of antitubercular treatment, mainly in countries with a high incidence of HIV infection and multidrug-resistant strains of TB. Contributing issues in DC include shortages of healthcare facilities with appropriate diagnostic equipment, reduced access to treatment, and poor treatment compliance among patients who often resort to traditional medicine5.
Most patients attending at the Italian Dermatological Centre (IDC) in Mekelle, the capital city of Tigray (Northern Ethiopia), are from remote areas with no access to health services. For this reason many patients use traditional medicine initially for health problems and present to hospital at advanced stages of disease.
Cutaneous TB represents 1% of all dermatological diseases seen in the IDC outpatients unit, with scrofuloderma being the most commonly observed form. The literature reports lupus vulgaris as the most common form of CT, particularly in patients with pulmonary involvement who have not had or have prematurely ceased systemic therapy.
The present case highlights the importance of establishing a patient's medical history and their physical examination, particularly in rural areas of DC where access to technical diagnostic equipment is unlikely.
Case report
A 70 year-old woman attended the IDC outpatient unit complaining of two ulcers on the left region of her face and neck. The disease was of 2 months' duration and commenced with the eruption of two painful, red-brown papules on the left side of the face and neck which gradually increased in size and eroded. The patient had undergone traditional topical treatment with herbs and this provoked a worsening of the lesions. The patient's medical history revealed a hospital admission, 5 months previously, for a productive cough, hemophtysis, weight loss and weakness. Pulmonary TB was diagnosed and confirmed by smear microscopy and culture, and chest X-ray, followed by systemic treatment that ceased after 1 month. Treatment discontinuation was due to difficulty of access to the treating hospital (distance from her home). No other noteworthy illness was reported.
On physical examination the skin ulcers were well demarcated, irregularly bordered and disfiguring; they were covered with slough and foul smelling with a purulent discharge present (Fig1). Full body examination detected no other lesions or suspicious lymph nodes.
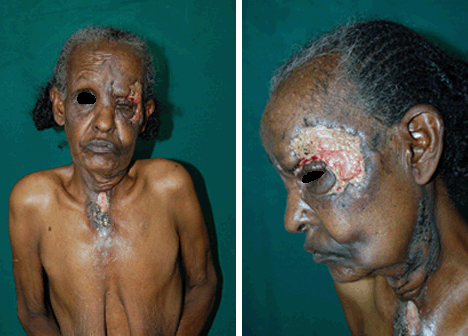
Figure 1: Skin ulcers on the face and neck of a 70 year-old woman with pulmonary TB.
Haemato-chemical tests were normal; testing for HIV was negative; chest X-ray showed cavities in the upper lungs, confirming the diagnosis of active pulmonary infection. Cytological examination of a needle aspiration from the neck lesion showed a non-specific bacterial superinfection, which was negative for acid-fast bacilli on Ziehl-Nielsen stain. It was decided to avoid biopsy for histology and culture and to immediately start recommence anti-tubercular treatment on the basis of clinical suspicion of CT. A significant improvement in the cutaneous lesions was noted after approximately 40 days, with a reduction in size, granulating bases and limited secretions (Fig2).
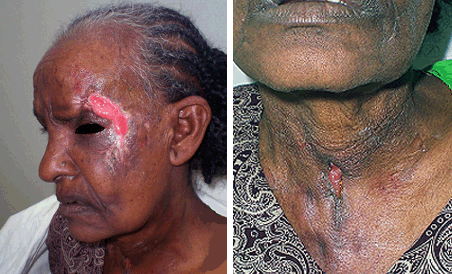
Figure 2: Improvement of skin lesions after 40 days of treatment.
Discussion
Cutaneous TB is a widespread in Ethiopia2,6, with several varieties, some of which are due to direct infection of the skin with Mycobacterium tuberculosis of external origin (tuberculous chancre, TB verrucosa cutis and sometimes lupus vulgaris), and others caused by spread from an internal source (scrofuloderma, miliaria, gumma, lupus vulgaris, orificial TB). Lupus vulgaris is the most frequent form in DC, where 10-20% of the patients have pulmonary or bone TB. Other forms of CT are localised immune reactions (delayed-type hypersensitivity) to AFB (tuberculids, including papulonecrotic tuberculid, Erythema induratum, Lichen scrofulosorum)7-13.
Currently, the diagnosis of skin ulcers is based on a careful clinical, laboratory and histopathological correlation, that considers the differential diagnoses of vascular ulcers, squamous cell carcinoma, leishmaniasis, rodent ulcer, fungal infection, Buruli ulcer, leprosy, atypical mycobacteriosis and other malignancy. The standard diagnostic approach is to take biopsies for cytology and Ziehl-Nielsen stain, culture and histology. However, in rural areas of DC, reduced access to diagnostic methods and advanced stage disease at presentation, encourages the use of empiric treatment on the basis of medical history and physical examination.
In IDC specimens for culture and histology are sent to Addis; however, in the present case it was decided to provide immediate empirical treatment on the basis of the clinical suspicion of CT. Contributing to this decision was the patient's history of defaulting TB treatment and the presence of a pulmonary focus, which alone would have necessitated TB treatment.
Conclusion
In rural areas of DC where diagnostic methods may not be available and advanced stages of disease such as CT are likely to be encountered, after the use of the most effective diagnostic tests available, empirical treatment on the basis of medical history and physical examination is suggested. Appropriate training of healthcare workers and public health education programs encouraging early presentation and improved patient treatment compliance are additional important preventative strategies.
Acknowledgement
The authors thank the patient in this report on for giving consent for publication, including the use of her clinical images.
References
1. Alianza Alto a la Tuberculosis y Organización Mundial de la Salud. Plan Mundial para Detener la Tuberculosis 2006-2015 (WHO/HTM/STB/2006,35). Ginebra: Organización Mundial de la Salud, 2006.
2. WHO. Global Tuberculosis Control. Geneva: WHO, 2009; 215.
3. Fariña MC, Gegundez MI, Piqué E, Esteban J, Martín L, Requena L et al. Cutaneous tuberculosis: a clinical, histopathologic, and bacteriologic study. Journal of the American Academy of Dermatology 1995; 33: 433-440.
4. Sàbat M, Ribera M, Casanova JM, Bielsa I, Fuente MJ, Ferrándiz C. Carcinoma epidermoide sobre lupus vulgar. Actas Dermo-Sifiliográficas 2003; 94: 616-619.
5. Handog EB, Gabriel TG, Pineda RTV. Management of cutaneous tuberculosis. Dermatologic Therapy 2008; 21: 54-161
6. Terranova M, Padovese V, Fornari U, Morrone A. Clinical and epidemiological study of cutaneous tuberculosis in Northern Ethiopia. Dermatology 2008; 217: 89-93.
7. Almaguer-Chávez J, Ocampo-Candiani J, Rendón A. Current panorama in the diagnosis of cutaneous tuberculosis. Actas Dermo-Sifiliográficas 2009; 100: 562-570.
8. Kivanc-Altunay I, Baysal Z, Ekmekci TR, Koslu A. Incidence of cutaneous tuberculosis in patients with organ tuberculosis. International Journal of Dermatology 2003; 42: 197-200.
9. Tappeiner G, Wolff K. Tuberculosis and other mycobacterial infections. In: IM Freedberg, AZ Eisen, K Wolff, AF Austen, LA Goldsmith, SI Katz (Eds). Fitzpatrick's dermatology in general medicine, 6th edn. Whitby, ON: McGraw-Hill, 2003; 1933-1949.
10. Ramos-e-Silva M, Ribeiro de Castro MC. Mycobacterial infections. In: JL Bolognia, JL Jorizzo, RP Rapini (Eds). Dermatology. Philadelphia, IL: Mosby, 2003; 1145-1164.
11. Tincopa Wong OW, Sánchez Saldana L. Tuberculosis cutánea. Dermatology Peru 2003; 13: 195-214.
12. Varas C, Eguino P, Gardeazabal J, Diaz-Pérez JL. Tuberculosis cutánea en cicatriz quirúrgica. Actas Dermo-Sifiliográficas 2003; 94: 412-413.
13. Serra Guillén C, Requena C, Alfaro A, Hueso L, Nagore E, Botella Estrada R et al. Lupus vulgar de 50 años de evolución. Actas Dermo-Sifiliográficas 2005; 96: 376-378.
