Skin diseases are a common cause of morbidity, especially among school children, worldwide. Although skin disease is rarely lethal, it can have a significant impact in terms of treatment cost, days absent from school, and psychological distress1,2. It is often stated that overcrowding3 and poor living conditions favor the development of many skin diseases4,5. However, when assessing risk factors for skin disease there are many other ecological and environmental considerations. Children are often exposed to climatic and social conditions that predispose them to develop skin infections and suffer from minor skin injuries6. A proper epidemiological approach to pediatric dermatoses in a community should include not only prevalence and incidence studies, but also sociocultural-economic correlates. This is especially important in the control of infectious dermatoses.
Although skin disorders are commonly observed conditions in children, only a few population based epidemiological studies exist that measure the prevalence of skin diseases in school children7-10. Previous studies conducted in the Kingdom of Saudi Arabia (KSA) have merely highlighted the high prevalence of skin disease among school children11; however, there are very few detailed epidemiological studies that focus on both prevalence and underlying socio-demographic/personal hygiene/cultural factors that might be responsible for the development of these lesions. The goal of the present study was to define the prevalent skin disorders, both transmissible and non-transmissible, among male primary school children in Al Hassa, KSA, and also to detect possible socio-demographic risk factors implicated in their development, with the hope of subsequently developing a better community approach to the management of pediatric dermatoses.
Setting
The study was carried out in Al-Hassa Governorate located in the Eastern Province of Saudi Arabia which has a total number of 168 public primary schools, of which 25 schools were located in 'Hegar' areas (consisting of scattered Bedouin communities), 90 in urban and 53 in other rural areas. The total number of students enrolled in these schools was 45 631 (Al Hassa Directorate of School Health records, 2009). Hegar schools were excluded from the sample frame for logistical reasons (chiefly transportation issues). Urban schools were located in two districts (Hofuf and Mubaraz), while rural schools were mainly located in 6 major villages.
Study design and sampling
For this cross-sectional study design the following considerations were employed for sample size calculation: the total recorded population for the academic year 2009 was 45 631 (Al Hassa Directorate of School Health records, 2009). Assuming a prevalence of skin lesions of 50%11 and the worst acceptable prevalence of 47%, applying a margin of error of 5% (95% confidence), the sample size would be 592. A design effect of 2 was considered in employing the cluster method of sampling, hence the sample size accounted to 1184. A 20% contingency factor was added, taking into account non-responders. Thus, the final sample size was 1421 school children. A proportionate sampling method was applied with regard to the rural-urban distribution using an appropriate sampling fraction. An updated list of all public primary schools was used as the sample frame, and 16 schools were randomly selected from the list: 6 rural and 10 urban. From each school, one class from each grade was randomly chosen and all its students were included (the study was restricted to male students due to local cultural restrictions).
Data collection tools and techniques
The following tools were used for collecting parents'/guardians' data, assessing children's hygiene habits and screening for skin disorders.
Parents'/guardians' data collection format: Socio-demographic data were gathered, including: age in years, nationality, residence, educational and occupational status of the parents, family income, family size, birth order of the child, type of current residence, pets in the house, presence of skin lesions among family members and their nature (physician-based diagnoses). Questionaires were sent with the selected children to their parents to be completed one week before the dermatological screening phase, along with a covering letter and consent form. The covering letter provided adequate orientation regarding study objectives and methods, and referral for treatment.
Hygiene habits of the children: The hygiene habits of the included children were assessed through a personal interview with the children on the day of their screening examination, using a structured form for gathering data regarding personal habits, including: bathing frequency, towel sharing, use of soap and other cleansing materials while bathing, frequency of underwear changing, nail trimming , and contact with pets/other animals.
Clinical screening for skin disorders: Dermatological examination was carried out on all school children upon receiving parental approval and signed consent forms. A complete head-to-foot examination was performed in a private room in each school, using day light. Examination was carried out by a single male dermatology consultant. Skin disorders were classified according to the International Diseases Classification modified 10th revision (ICD 10)12. Atopic eczema was defined using the UK Working Party Criteria for Atopic Dermatitis13.
Pilot testing
The data collection tools were pre-tested on 158 male primary school children from nearby schools beyond the sample size to ensure readability and proper administration of the data collection forms dedicated to both children and their guardians. A reliability coefficient was calculated for enquiries regarding hygiene habits (Cronbach's alpha = .78) and for parental data (=.81).
Lesions diagnosed by the field dermatologist were re-evaluated by a consultant at a tertiary care hospital, with a total percent agreement of 89.1% (100% for acne and pediculosis, 95.6% for dandruff, 88.2% alopecia, 82.7% for eczema, and 77.8% for fungal infections).
Data analysis
Out of 1695 children sent consent forms, 1421 agreed to participate (83.8% response rate). Those who refused to participate did not differ significantly from those sampled regarding socio-demographic characteristics. For the sake of data validity, 74 subjects were excluded due to missing parental socio-demographic forms; thus, the final number of participants was 1337 males. Those excluded from analysis due to missing data were not significantly different from those included with regard to the distribution and frequency of skin lesions.
Data entry and data processing was carried out using SPSS V16.0 software (SPSS Inc; Chicago IL, USA; www.spss.com). Both descriptive and inferential data analyses were applied using the appropriate statistical tests of significance including χ², Z-test for proportions and t-test. Categorical variables were expressed in percentages with 95% confidence intervals. Pearson's correlation coefficient was used when appropriate. A multivariate binary logistic regression model was generated by the inclusion of significant variables at univariate analysis, rating various socio-demographics and personal hygiene habits against the most commonly encountered skin lesions as dependent variables. Confidence interval of 95% and significance level of p £0.05 were applied.
Ethical considerations
Permissions and ethical clearances were obtained from the local School Health and Education Directorates, as well as our Institutional Ethics Committee. Parental data collection forms were appended with a detailed 'orientation document' regarding the objectives of the study and procedures involved; it also informed about their child's skin problem, and whether there was any need for treatment or referral to higher level of care (when indicated). Written consent (from the parents) and the agreement of the student were required to be enrolled in the study. Prior orientation of the teaching and administrative staff at the selected schools was carried out. Data confidentiality and subjects' privacy were maintained throughout the study.
The age of included children ranged from 6 to 13 years with a mean of 10.37 (standard deviation = 1.41 years). Saudi children comprised 76.1% of the subjects, while the remainder consisted of other nationalities (Egyptians, Syrians, Jordanians and Sudanese).
Socio-demographic features and family history of skin related conditions
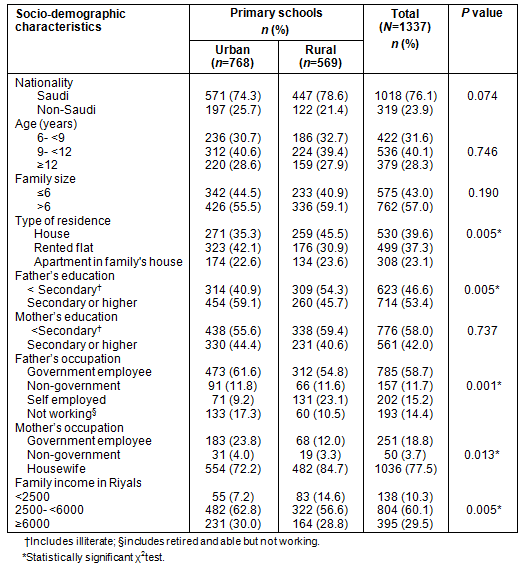
The socio-demographic characteristics of the included children are provided (Table 1). Urban males constituted 57.4% of the children. The number of family members of the included children ranged from 4 to 22 persons with mean of 7.81 ± 2.9 and a median of 7 with no significant difference between urban and rural families (7.91 ± 3.09 for urban vs 7.63 ± 2.82 for rural families, p = 0.088).
The type of current housing varied, with rural children being more likely to reside in apartments within the family house, compared with the urban sample where more children resided in rented flats (p = 0.005).
Paternal educational status was higher in urban compared with rural children, while this difference was not found when comparing maternal educational status. Only 22.2% of children's mothers were employed. Family income was higher in urban families.
Table 1: Socio-demographics of the included male primary school children, Al Hassa, in relation to urban or rural type of school
History of skin disorders and related conditions in the family
Acne vulgaris was reported in other family members in 132 (9.9%), eczema in 176 (13.2%), dandruff in 138 (10.3%), fungal infections in 23 (1.7%) and other non-specific skin lesions was reported among siblings and parents in 164 subjects (12.3%).
Positive history of bronchial asthma was reported among siblings or parents in 137 children (10.2%). Chronic diseases among the sample, as reported by guardians, included sickle cell disease and other blood diseases in 13 children (1%), bronchial asthma in 174 children (13.0%), diabetes mellitus in 11 (0.8%) and congenital heart disease in 2 children.
Hygiene and domestic habits
Pet ownership was reported by 30.7% of subjects, more in the rural (198; 34.9%) than urban (212; 27.6%) children (p = 0.004). Cats, rabbits and poultry were most frequently mentioned overall, with cats more in urban, and rabbits, poultry, goat and sheep in rural children.
Among those with pet ownership, regular and frequent contact was reported among 118/212 (55.7%) of urban children, compared with 153/198 (77.3%) in rural children.
'Frequency of showering per week' showed a median result of 3 (times per week), towel sharing was stated by 600 children (44.9%), frequency of change in underwear was at a median of 5 times/week, and frequency of changing clothes a median of 4 times/week. There was no significant urban-rural difference with regard to hygiene habits.
Prevalence of transmissible skin disorders
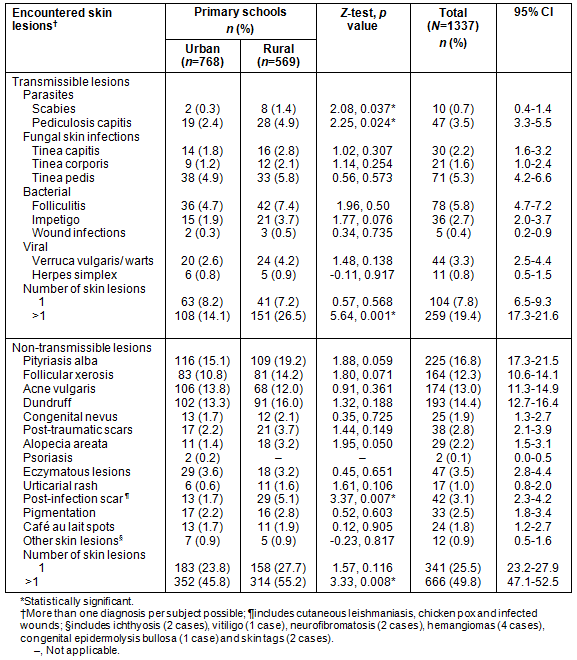
The prevalence of diagnosed transmissible and non-transmissible skin lesions encountered among the school children according to location is provided (Table 2). The overall prevalence of transmissible skin lesions was 27.2% (CI=24.8-29.6); in rural children it was 33.7% (CI= 29.9-37.6), compared with 22.3% (CI=19.5-25.3) in urban.
Single transmissible dermatosis were diagnosed in 104/133 (7.8%) subjects, while 259 (19.4%) had more than one disorders, with the incidence of multiple dermatoses being more common among rural school children. Ectoparasitic skin infections, namely scabies and pediculosis capitis, were more prevalent among rural children.
Bacterial skin infections, namely folliculitis and impetigo, were diagnosed among those of relatively younger age (8.71 ± 1.29 years). Children diagnosed with viral skin infections, mainly in the form of warts, were aged 9.72 ± 1.61 years.
Regarding the multiplicity of skin disorders, those affected by fungal skin infections showed frequent associations with other transmissible skin disorders: 16/47 children (34%) with pediculosis and 12/36 cases of impetigo had co-existing fungal skin infections.
Table 2: Distribution and prevalence of encountered skin lesions among the included male primary school children in Al Hassa
Non-transmissible disorders
Non-transmissible disorders were diagnosed in 1007 children (75.3%, CI=72.9-77.6); 341 (25.5%) with single dermatosis and 666 (49.8%) with multiple dermatoses, the incidence of multiple dermatoses being more among rural children.
The most frequently encountered dermatoses were pityriasis alba followed by keratosis pilaris, dandruff/seborrheic dermatitis, acne vulgaris, atopic dermatitis and other non-specific eczematous lesions.
Children with pityriasis alba had an average age of 11.1 ± 1.2 years, while for keratosis pilaris it was 11.3 ± 1.5 years. These two conditions were more likely among rural school children but this was without statistically significant difference. Both acne vulgaris and dandruff affected older children (12.7 ± 1.51 years for acne and 11.5 ± 1.38 years for dandruff, p = 0.001). Acne vulgaris lesions were correlated with the age of subjects, with 10 cases of those aged 10 years, 31 at 11 years, 69 at 12 years and 64 cases among those aged 13 years or older (correlation coefficient=.209, p = 0.001). Alopecia areata was more common among rural children (3.2%) compared with urban (1.4%) but this was without statistical significance.
Post-infectious scars were significantly more prevalent among rural children and they included of 23 cases of cutaneous leishmaniasis (18 rural and 5 urban), infected wounds in 11 cases, and chicken pox in 12 cases.
For eczematous skin lesions (including atopic dermatitis), the total number of subjects affected were 47 (3.5%). Affected children had a mean age of 10.12 ± 0.98 years, 11 cases were less than 9 years of age, 27 cases were 9-10 years, and another 9 cases were older than 10 years. Most of the affected children were Saudi nationals, 44/47(93.6%). Atopic dermatitis was associated with other skin lesions such as pityriasis alba in 41 out of 47 children, and with bronchial asthma in 31 out of 47 children.
Socio-demographic correlates of skin disorders
Tables 3 and 4 demonstrate univariate and multivariate logistic regression analyses of the two most frequently encountered transmissible skin diagnoses, pediculosis and tinea, in relation to possible socio-demographic and hygiene correlates.
The logistic regression model revealed that large family size was a predictor for infection with both lesions, while rural residence was an additional positive predictor for pediculosis. However, higher maternal educational status was found to be possibly protective against the development of both lesions. Additional determinants revealed for superficial fungal infection included the protective effect of non-sharing of towels (OR=0.69, C.I=0.43-0.97, p = 0.038). For pediculosis capitis, frequent showering and high family income were both negative predictors for the development of infection.
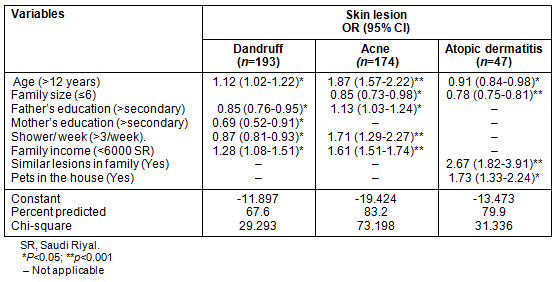
The results are given of univariate and logistic regression analyses of the possible socio-demographic determinants for the three most commonly encountered non-transmissible skin disorders: acne, dandruff and eczema (Tables 5,6). Logistic regression showed that older age of the child was positively associated with dandruff and acne but negatively associated with atopic dermatitis. Higher paternal education was a negative predictor for dandruff, while it was a positive correlate for the development of acne.
Frequent showering was a negative predictor for dandruff but was found to be a positive correlate to acne. High family income was a risk factor for the development of dandruff and acne. Similar lesions among family members and pet ownership were positive predictors for the development of atopic dermatitis.
Table 3: Univariate analysis of commonly encountered transmissible skin disorders in relation to socio-demographic and hygienic habits of male primary school children, Al Hassa
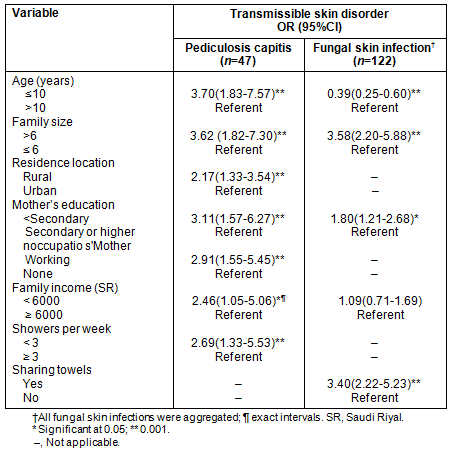
Table 4: Logistic regression models for common transmissible skin disorders among the included male primary school children, Al Hassa
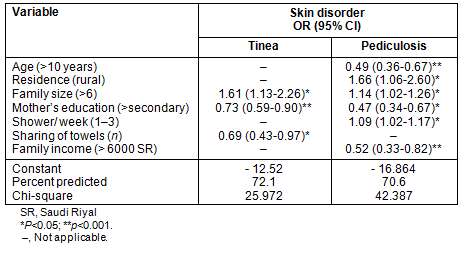
Table 5: Univariate analysis of commonly encountered non-transmissible skin disorders in relation to socio-demographic and hygienic habits among male primary school children, Al Hassa
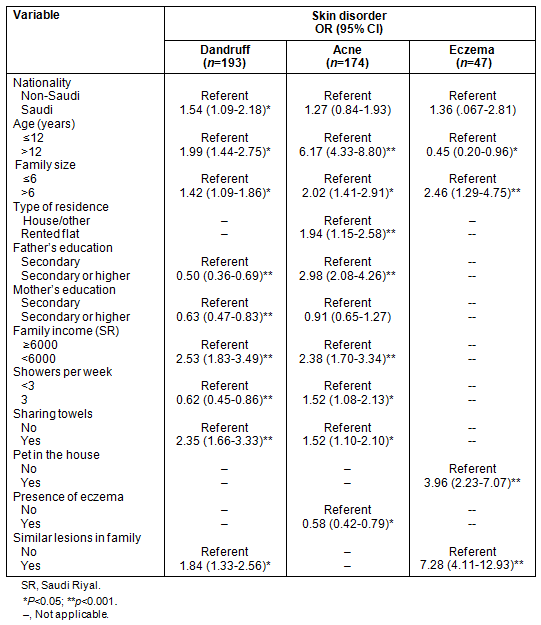
Table 6: Logistic regression analysis of the non-transmissible skin lesions among the included male primary school children
Discussion
The prevalence of skin diseases in the pediatric age group is very high. Different studies have shown cross-sectional prevalence rates ranging from 22.8 to 96.8%6-8,14-16. Skin diseases are considered to be the second most common cause for medical consultation for children in rural communities16. It has also been shown that in regions with a poorer socioeconomic environment, morbidity rates, especially regarding infectious diseases, are higher17. It is therefore important from the point of view of community health to assess not only the prevalence of skin disease, but also to identify possible associated socioeconomic factors.
As far as prevalence of skin disease is concerned, the present study results are similar to many previous school-based studies in the sense that the major groups of dermatosis identified were essentially the same. These include: eczematous dermatosis (such as pityriasis alba/keratosis pilaris), infectious dermatosis (superficial fungal and bacterial infections, warts etc), infestations (scabies, pediculosis), and others such as acne, pigmentary disorders/nevi. As mentioned previously, broad studies have been made into the prevalence of dermatological problems in school children. The prevalence rates of many transmissible and non-transmissible diseases in the present study are similar to some previous studies on the same age group14,17.
However there are very few studies which have attempted to highlight the socioeconomic-cultural correlates, especially with regard to rural versus urban populations. Two previous similar studies were conducted in Saudi Arabia: the study by Zimmo et al showed an overall prevalence of skin disorders among male primary school children in Jeddah, KSA, of 19.23 per 100 school children18. High prevalence rates were reported for nevi (12.9%), head lice (11%), pityriasis alba (8%) and alopecia (6.1%). The prevalence rates for acne vulgaris and warts were similar to our study (2.9% each). The prevalence rate for atopic eczema was 2.1%, while the prevalence rates for tinea capitis, and tinea versicolor were relatively low (0.25%, 0.82%, respectively)18. The second study by Al-Saeed et al reported a prevalence of skin disease among female school children in Al-Khobar City, KSA, of 98.6%. The most common condition was pigmentary disorders (91.6%), followed by a group of dermatitis/eczema and related conditions (26.7%), and disorders of skin appendages (25.3%)19.
The prevalence of non-transmissible disease in our study is similar to studies in more developed nations but the prevalence of transmissible disease was higher. However, the prevalence of transmissible disease was lower than that shown in studies from less-developed countries6-8,15,16.
Transmissible dermatoses
In general, transmissible dermatoses were more prevalent in the rural population compared with the urban population in the present study. This is so for most common transmissible skin diseases, including ectoparasitic infestations, and superficial fungal, viral and bacterial infections.
Ectoparasitic infestations - scabies and pediculosis: Pediculosis capitis and scabies are common parasitic skin diseases, especially in resource-poor communities, but data on epidemiology and morbidity are scarce. Heukelbach et al conducted a population-based study on the epidemiology and morbidity of scabies and pediculosis capitis in resource-poor communities in Brazil20. Important results from the study were that the prevalence of pediculosis capitis was found to be 43.4% in urban slum areas. Scabies was present in 8% of the population in the urban slums, without any consistent pattern in relation to age distribution. Multivariate analysis showed that being less than 15 years, being of female sex and living in an urban slum were independent factors contributing to simultaneous co-infestation with pediculosis capitis and scabies20. Another study on pediculosis capitis from Australia by Speare et al showed a prevalence of 33.7 %. This study also found a clustering of cases by school class, indicating the classroom was the main source of infestation21. A study of Argentinean school children gave preference to massive, complete, and simultaneous treatment of the whole school as opposed to the treatment of single cases22.
There are very few studies which have tackled the socioeconomic-cultural factors governing the epidemiology of scabies. In a study by Karim et al in Bangladesh23, of 492 children included 98% of children had scabies, of which 71% had been re-infected (96% during the winter). In all, 74% of children were living in poorly ventilated buildings with overcrowded sleeping arrangements and poor personal hygiene. Disease severity and re-infection were significantly associated with infrequent washing of clothes (p = <0.001) and bed linen (p = <0.001), overcrowded sleeping arrangements (p = <0.001) and infrequent bathing (p = <0.001) with soap (p = <0.001). This was further related to household income (p = <0.001 for both infrequent bathing and soap use)23. To conclude, ectoparasitic infestations are more prevalent in the rural population and specific sociocultural-economic factors may contribute significantly to their development, and this must be addressed for proper epidemiological control.
Superficial fungal infections: Fung et al found that the prevalence of superficial fungal infections in a school-based study was 2.2%. Tinea cruris was the most common type among the group of superficial mycotic infections (72.7%), tinea pedis (9.1%) was next most common, followed by tinea versicolor (4.5%), chronic paronychia (4.5%), intertrigo (4.5%), and tinea corporis (4.5%)24. The study by Dogra et al showed a prevalence of 2.9% while Inanir et al showed a prevalence of 0.75%14,17. One of the largest prevalence studies of tinea capitis from Slovenia showed a prevalence of 3.9 % (n = 33974). A total of 91.1% of tinea capitis cases in this study were caused by Microsporum canis and cats were considered to be the main source of infection. The author suggested that the transmission of M. canis from stray cats to domestic cats lead to the mycotic infection of young children playing with these kittens25. According to Ginter-Hanselmayer et al, children (aged 3-7 years with no predilection for gender) remain the most commonly affected26. The importance of pets/animal contact with regards to superficial skin infections needs to be emphasized, especially in the context of a rural populace.
Warts and other superficial cutaneous viral infections: The prevalence of superficial viral infections including warts was 3.8% in the study by Inanir et al; however, it was approximately 1% in the study by Dogra et al14,17. In both studies the commonest viral infection was warts. Warts are very common in primary school children. A large study from Netherlands by van Haalen et al found that 33% of the 1465 primary school children studied had warts. Parental questionnaires showed that environmental factors connected with barefoot activities, public showers or swimming pool visits were not related to the presence of warts. An increased risk of the presence of warts was found in children with a family member with warts, and in children where there was a high prevalence of warts in the school class27. While the role of socio-cultural factors and hygiene may not be very significant in the case of superficial viral infections such warts, factors like overcrowding and shared clothing must be taken into account while considering options for epidemiological control.
Superficial bacterial infections: Inanir et al, in their study from Turkey showed a prevalence of approximately 2% for superficial bacterial infections. Fung et al showed a prevalence of 0.3% and Dogra et al reported a higher prevalence of 7.34%14,17,24. A study by Ide et al from Jamaica revealed a significant correlation between pyodermas and a lower socioeconomic status. The same study, however, could not demonstrate the significant effect of other factors, such as nutritional status, size of family, bathing frequency, or water supply28. As in superficial fungal infections, the general prevalence for superficial bacterial infections is definitely greater in the rural population. Again, factors such as poor hygiene and overcrowding could be the major factors affecting the distribution of superficial bacterial skin infections.
Non-transmissible dermatoses
Non-transmissible dermatoses did not show a significant difference with respect to urban versus rural populations. In fact some conditions, such as acne, showed a higher prevalence in the urban group.
Eczemas/dermatitis (including atopic dermatitis, pityriasis alba, xerosis, follicular eczema/pilar keratosis, dandruff): The point prevalence of eczemas/dermatitis was 6.5% in the study of Dogra et al14. This figure is much lower than the reported prevalence of 11.4-22.3% in one population-based survey performed in the West29. Eczemas, including atopic dermatitis, have a higher prevalence in developed countries, being influenced by socioeconomic and environmental factors such as excessive hygiene, carpets, and central heating13.
Inanir et al reported that the eczema dermatitis group was the second most common dermatosis in their study. Keratosis pilaris, pityriasis alba and xerosis were the most common eczematous conditions and were observed in more than one-tenth of the children. Atopic dermatitis, dandruff, and discoid eczema were also frequent17. Fung et al have also demonstrated a high prevalence for eczematous conditions (endogenous eczema including atopic dermatitis 6.8%, keratosis pilaris 1.3%, pityriasis alba 1%, xerosis 0.2%). Some eczematous conditions, such as pityriasis alba, can be directly correlated with parameters reflecting low socioeconomic status. Some of these parameters were economic, such as family income, health insurance coverage, and housing conditions, while others were related to educational level. Pityriasis alba could also be correlated to the number of siblings in the household17.
Acne vulgaris: In a study from Iran, the overall acne prevalence was 93.3% (94.4% for boys and 92.0% for girls) in the 15-17 years age group. Acne severity risk increased with the number of family members with an acne history30. The reported prevalence is much less in other school based studies (Dogra et al 0.93%, Inanir et al 2.6%), and this is probably because acne is primarily a disease of older adolescents14,17.
Nevi, hamartomas and non-specific/nevoid pigmentation: Most school based studies, as expected, have demonstrated a high prevalence of nevoid conditions and pigmentation. Inanir et al found that almost one in five children had one of these nevoid lesions, while Fung et al reported a prevalence of approximately 10% and Dogra et al showed a lower prevalence of approximately 2%. The epidemiological importance is probably more in the context of melanocytic nevus with its risk of malignant transformation, which again would be more important for fair skinned populations14,17,24.
Papulo-squamous diseases: The papulo-squamous diseases category includes psoriasis, lichen planus and lichen nitidus, and it was relatively rare among the present study's subjects. This is similar to prevalence rates of 0-2% in various, similar studies8,14,16,17,24.
Autoimmune diseases: Of the primary cutaneous autoimmune diseases, alopecia areata was the most common in our study with prevalence of 1.4% and 3.2% in urban and rural schools, respectively. No cases of vitiligo were detected. The study by Inanir et had only one case of alopecia areata (0.13%) and no cases of vitiligo, while Dogra et al's study reported a prevalence of 2.2% for vitiligo14,17.
Limitations
The results of this study must be considered with the following limitations. First, the cross-sectional data collection study design may imply biased information, especially regarding hygiene habits, with potential over-reporting of favorable behaviors, for which controlling was difficult. Second, the study included differing ethnic groups, although there was no significant difference in their hygiene habits and/or socio-demographics because they shared similar cultural and religious backgrounds; however, detecting and controlling this confounder was also difficult. Third, those who did not participate in the study might have had a different pattern/frequency of skin lesions and/or different socio-demographic and hygiene habits. Fourth, validation of the diagnosed lesions was carried out at the outset during pilot testing; however, due to logistical factors and a lack of parental cooperation it was not possible to validate all diagnosed cases.
In this cross sectional study, transmissible skin disorders were found to be quite common among primary school children in Saudi Arabia, affecting approximately one in four. The overall prevalence of transmissible skin disorders (27.2%) was slightly higher in the children from rural schools (33.7%) compared with that of the urban children (22.3%). One of the most salient results of the study was that the prevalence of all infectious/transmissible dermatoses was higher in the rural population. Also of epidemiological significance was the prevalence of most non-transmissible dermatoses being almost equal in urban and rural populations. It is of interest that some conditions, such as acne and eczematous dermatitis, showed a higher prevalence in urban populations. The vast majority of the included school children were affected by multiple skin lesions, while solitary transmissible skin disorders were diagnosed in 7.8% subjects. As revealed in the logistic regression model, large family size was a predictor for infection with pediculosis and tinea, while rural residence was a positive predictor for pediculosis. Higher maternal educational status may be protective against the development of both lesions. Frequent showering and high family income were both negative predictors for the development of infection.
It is important to determine not only the prevalence of skin disorders, but also possible underlying socio-demographic and hygiene factors, to ensure that any necessary educational programs and preventive measures can be properly planned and implemented in a culturally sensitive manner. It is therefore recommended that regular skin examination is performed in schools in order to identify children with skin disorders, and to help children to understand the importance of effective personal hygiene. Such school-based health programs should also include education for teachers and students' families about commonly observed skin diseases and their prevention.
Implications and recommendations for rural healthcare policy
This study underlines the need for definitive changes in health policy, especially in the context of the rural population, for better epidemiological control of transmissible skin diseases. Essential components of this policy change would include:
- Regular health education classes targeting both teachers and parents to provide a general idea of common pediatric dermatoses and related socioeconomic factors, especially effective personal hygiene. Government health agencies through primary healthcare centers should take the lead in this educational process
- Ensuring that routine school health check-ups give due importance to skin diseases, especially the early detection and treatment of transmissible disease such as scabies and pediculosis
- Ensuring adequate budgetary allocation for the epidemiological control of skin diseases in children, considering the significant burden that these may pose
- Indirectly encouraging positive governmental policy changes that involve improvements to rural housing and sanitation
- Ensuring the optimum number of students in classrooms, as per accepted universal guidelines, in order to prevent overcrowding.
Acknowledgement
The authors thank Dr Burhan Omar Zaza, the technical director of School Health Program in Al Al Hassa, Ministry of Education for his valuable assistance to accomplish this study.
References
1. Greaves MW. Dermatology. Practitioner 1976; 217: 585-590.
2. Clore ER, Longyear LA. Comprehensive pediculosis screening programs for elementary schools. Journal of School Health 1990; 60: 212-214.
3. Harris M, Nako D, Hopkins T, Powell DM, Kenny C, Carroll C et al. Skin infections in Tanna, Vanuatu in 1989. Papua New Guinea Medical Journal 1992; 35: 137-143.
4. Ebomoyi EW. Pediculosis capitis among urban school children in llorin, Nigeria. Journal of the National Medical Association I994; 86: 861-864.
5. Wegner Z, Racewicz M, Sta?czak J. Occurrence of pediculosis capitis in a population of children from Gda?sk, Sopot, Gdynia and the vicinities. Applied Parasitology 1994; 35: 219-225.
6. Kottenhahn RK, Heck JE. Prevalence of paediairic skin diseases in rural Honduras. Tropical Doctor 1994; 24: 87-88.
7. Popescu R, Popescu CM, Williams HC, Forsea D. The prevalence of skin conditions in Romanian school children. British Journal of Dermatology 1999; 140: 891-896.
8. Bechelli LM, Haddad N, Pimenta WP. Epidemiological survey of skin diseases in school children living in the Purus Valley (Acre State, Amazonia, Brazil). Dermatologica 1981; 163: 78-93.
9. Larrson PA, Liden S. Prevalence of skin diseases among adolescents 12-16 years of age. Acta Dermato-Venereologica 1980; 60: 415-423.
10. Porter MJ, Mack RW, Chaudhary MA. Pediatric skin disease in Pakistan. A study of three Punjab villages. International Journal of Dermatology 1984; 23: 613-616.
11. Abolfotouh MA, Abu-Zeid HA, Bahamdan K, Abdel Aziz M, Bassuni WA, Eid O. Skin disorders among male school children in the Asir region, southwestern Saudi Arabia. Annals of Saudi Medicine 1996; 16: 342-345.
12. WHO. International Statistical Classification of Diseases and Related Health Problems, 10th revision. Diseases of the skin and subcutaneous tissue, L00-L99. (Online) 2007. Available: http://apps.who.int/classifications/apps/icd/icd10online (Accessed 10 April 2010).
13. Williams HC. Epidemiology of skin diseases. In: RH Champion, JL Burton, DA Burns, SM Breathnach (Eds). Textbook of Dermatology, 6th edn. Oxford: Blackwell Science, 1998; 139-158.
14. Dogra S, Kumar B. Epidemiology of Skin Diseases in School Children: A Study from Northern India. Pediatric Dermatology 2003; 20: 470-473.
15. Mahe A, Prual A, Konate M, Bobin P. Skin diseases of children in Mali: a public health problem. Transactions of the Royal Society of Tropical Medicine and Hygiene 1995; 89: 467-470.
16. Figueroa JI, Hawranek T, Abraha A, Hay RJ. Prevalence of skin diseases in school children in rural and urban communities in the Illubabor province, south-western Ethiopia: a preliminary survey. Journal of the European Academy of Dermatology and Venereology 1997; 9: 142-148.
17. Inanir I, Sahin MT, Gündüz K, Dinç G, Türel A, Oztürkcan S. Prevalence of skin conditions in primary school children in Turkey: differences based on socioeconomic factors. Pediatric Dermatology 2002; 19: 307-311.
18. Zimmo SK, Qari MA, El-Gamal F, Mkordy MN. Prevalence of skin disorders among male primary school children in the City of Jeddah, Saudi Arabia. Saudi Medical Journal 1996; 17: 56-61.
19. Al-Saeed WY, Al-Dawood KM, Bukhari IA, Bahnassy AA. Prevalence and pattern of skin disorders among female school children in Eastern Saudi Arabia. Saudi Medical Journal 2006; 27: 227-234.
20. Heukelbach J, Wilcke T, Winter B, Feldmeier H. Epidemiology and morbidity of scabies and pediculosis capitis in resource-poor communities in Brazil. British Journal of Dermatology 2005; 153: 150-156.
21. Speare R, Buettner PG. Head lice in pupils of a primary school in Australia and implications for control. International Journal of Dermatology 1999; 38: 285-290.
22. Chouela E, Abeldao A, Cirigliano M, Ducard M, Neglia V, La Forgia M et al. Head louse infestations: epidemiologic survey and treatment evaluation in Argentinian school children. International Journal of Dermatology 1997; 36: 819-825.
23. Karim SA, Anwar KS, Khan MA, Mollah MA, Nahar N, Rahman HE et al. Socio-demographic characteristics of children infested with scabies in densely populated communities of residential madrashas (Islamic education institutes) in Dhaka, Bangladesh. Public Health 2007; 121: 923-934.
24. Fung WK, Lo KK. Prevalence of skin disease among school children and adolescents in a Student Health Service Center in Hong Kong. Pediatric Dermatology 2000; 17: 440-446.
25. Dolenc-Voljc M. Dermatophyte infections in the Ljubljana region, Slovenia, 1995-2002. Mycoses 2005; 48: 181-186.
26. Ginter-Hanselmayer G, Weger W, Ilkit M, Smolle J. Epidemiology of tinea capitis in Europe: current state and changing patterns. Mycoses 2007; 50(2): 6-13.
27. Van Haalen FM, Bruggink SC, Gussekloo J, Assendelft WJ, Eekhof JA. Warts in primary school children: prevalence and relation with environmental factors. British Journal of Dermatology 2009; 161: 148-152.
28. Ide A. The epidemiology of pyoderma in Jamaican children. Cutis 1989; 44: 321-324.
29. The International Study of Asthma and Allergies in Childhood (ISAAC) Steering Committee. Worldwide variation in prevalence of symptoms of asthma, allergic rhino-conjunctivitis and atopic eczema. Lancet 1998; 351: 1225-1232.
30. Ghodsi SZ, Orawa H, Zouboulis CC. Prevalence, severity, and severity risk factors of acne in high school pupils: a community-based study. Journal of Investigative Dermatology 2009; 129: 2136-2141.


