Rural general practitioners who care for Aboriginal patients in Australia must be aware of strongyloidiasis because this chronic parasitic disease commonly affects these patients, particularly those in rural and remote tropical communities1. Other groups in Australia at high risk of strongyloidiasis are immigrants from endemic countries and military personnel who have served in endemic areas. Elimination programs for enteric parasites in rural Australian Indigenous communities are so important that a nationally coordinated approach has been advocated2. Wisely used cost-effective diagnostic tests are a critical component of an elimination program.
Strongyloidiasis diagnosis has relied on laboratory examination because the only pathognomic clinical sign of strongyloidiasis is larva currens, a rapidly moving erythematous, serpinginous skin lesion similar to cutaneous larval migrans, but moving much faster, often at rates of 10 cm per day. Diagnostic tests include: (i) serology to detect anti-strongyloides antibody; and (ii) faecal examination. In this article, comments are confined to strongyloides serology and its role in the diagnosis and management of strongyloidiasis.
Strongyloides ELISA
The ELISA test to detect serum antibodies against the pathogenic gut nematode, Strongyloides stercoralis, complements tests to detect larvae, mainly in the stools of infected individuals. Recent research has confirmed that the detection of positive cases is improved when both tests are used3. This is not surprising because complementary diagnostic tests used simultaneously (ie, in parallel) maximize diagnostic sensitivity4. Nevertheless, unlike many other serological diagnostic tests for microorganisms, application of strongyloides ELISA is frequently challenged on the grounds of uncertain usefulness5. Humans, including clinicians, crave certainty, but in biological systems certainty is an illusion and a relentless crusade for certainty may retard the application of useful but imperfect measures to improve human health6. The degree of uncertainty inherently characterising a diagnostic or screening test is expressed as its sensitivity and specificity. These are conditional probabilities that are properties of the test per se. When the test is used in a particular patient population, its value also depends on the prevalence of the disease in that population. It is worth emphasising that all diagnostic and screening tests and subsequent interventions based on test results have benefits and costs, and the value of a particular test depends on how much the benefits exceed the costs in the specific population. This article examines the role of the strongyloides ELISA in diagnosing and managing human strongyloidiasis, and discusses what additional data is needed to better define this role. The Australian context is ideal for this exercise because it provides populations at both ends of the prevalence spectrum.
In Australia the strongyloides ELISA is standardised, and has a sensitivity and specificity of 93% and 95% respectively, originally determined in a group of former prisoners of war from Singapore7. This test performance is comparable to well-established serological tests for other diseases7,8. Sackett and Haynes9 proposed that in evaluating the usefulness of diagnostic tests, four phases of questions should be answered. The strongyloides ELISA has progressed to Phase II only ('Are patients with positive strongyloides ELISA more likely to have the target disorder than patients with a negative or borderline result?'), providing the current values for sensitivity and specificity7. The Phase III question ('Does the test distinguish patients with and without strongyloidiasis among patients in whom it is clinically reasonable to suspect that the disease is present?') needs to be answered for the various target groups. Independent, blind comparison with a gold standard of diagnosis will be required for these studies9. A major constraint is that there is no available gold standard for diagnosis10-11 because the strongyloides ELISA has a greater sensitivity than the examination of faeces for larvae of strongyloides3,8,12,13.
Benefits of treating strongyloidiasis
Although there are clearly no perfect tests currently available for strongyloides diagnosis, there are definite psychological, economic, morbidity/mortality alleviation and epidemiological benefits to diagnosing and correctly treating strongyloidiasis. Available diagnostic tests, including the strongyloides ELISA should be optimally utilised until better alternatives are available. Most people with strongyloidiasis have symptoms, and infected individuals occasionally progress to severe disease and death. The knowledge that the parasite has been eradicated from a patient's gut is reassuring both for the treating clinician and the worried symptomatic patient. Economic benefits include those that directly accrue to the health service through preventing severe disease and recurrent health service presentations, and the costs to the individual patient and their family, in terms of healthcare costs and lost productivity due to chronic ill health. If a patient is liable to contaminate the soil with faeces, then effective treatment will also reduce transmission in the patient's community, with attendant epidemiological benefits.
Costs
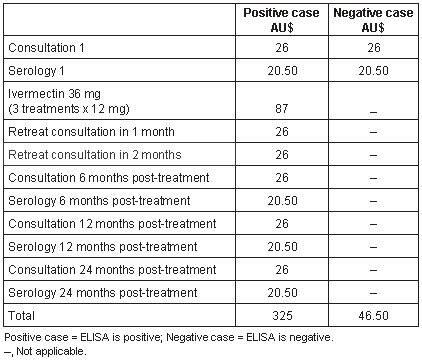
What are the costs associated with strongyloides serology? The direct costs of diagnosis include a consultation involving a venepuncture and the costs of the ELISA test (Table 1), approximately $46.50 using the Australian Medicare benefits for a standard consultation and laboratory test (Table 2). Of course, these costs can be higher or lower depending on whether cases are managed through private GPs, Aboriginal health services or through health departments. The costs used provide a starting point allowing comparison between groups. Further costs are negligible if the test is negative, while if the test is positive, direct costs include those for therapy and subsequent monitoring. The ideal monitoring regimen post-treatment has not been determined. We would recommend six-monthly monitoring for 1 year and if the 12 month test is negative, a final serological test at 2 years; that is, three repeat serological tests14. Because the most effective drug, ivermectin, has a very low incidence of side effects15-17, the indirect costs associated with adverse events are probably negligible.
Table 1: Components comprising costs of positive and false negative strongyloides ELISA
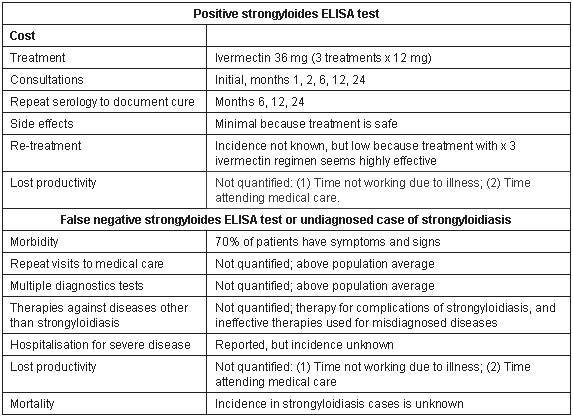
Table 2: Estimate of direct cost of diagnosing, treating and monitoring patients using the strongyloides ELISA. Consultation costs are the Australian Medicare benefit for standard consultation (Item 23) and Australian Medicare benefit for the strongyloides ELISA (Item 69384)
Role of strongyloides ELISA in Management of strongyloidiasis
Although it has been postulated that the ELISA might demonstrate greater sensitivity in people chronically exposed to S. stercoralis18, this was not proven until recently when infected adults in the UK were shown to be more likely to have a negative ELISA than those with more long-standing strongyloidiasis3. Young age per se does not guarantee seronegativity19. In Australia, positive serology was demonstrated in immigrant children as young as one year of age from Indochina20. Five per cent of children who were aged between one and two years gave positive reactions, and the positive reaction rate in this population increased by approximately 7% per year until the age of seven to eight years at which point it leveled off20. The time necessary for seroconversion is unknown for human strongyloidiasis. Dogs experimentally infected with S. stercoralis had a rise in anti-strongyloides IgM at one week and in IgG soon after21. Although acute infections may be seronegative in humans, the window period in humans is unknown. This needs to be determined by studying short-term travelers of all ages exposed to high risk situations.
The role of the strongyloides ELISA in the management of strongyloidiasis is equally important. Although the ELISA detects anti-strongyloides IgG, the titre falls after effective treatment7-8,22. This makes the test potentially valuable as a monitoring tool. A failure of the titre to fall can thus be used by clinicians to determine whether retreatment is required. This remains controversial, with those arguing against the usefulness of serology for monitoring patient response often basing their arguments on trials using inadequate treatment regimens, in most cases a single treatment with albendazole or ivermectin3. Single treatment failure is not uncommon owing to two unique aspects of the biology of S. stercoralis. First, S. stercoralis has autoinfective larvae, which are more difficult to kill with either albendazole or ivermectin7,23. Second, the parasitic female, the only adult parasitic stage, reproduces by parthenogenesis, and one worm is a patent infection. Parthenogenesis is a feature of the genus while production of autoinfective larvae has been identified in only two species of strongyloides, S. stercoralis and S. felis; the latter being a parasite of cats in India and Australia24-25. Even if all adult S. stercoralis in the small intestine mucosa/wall are killed by a single dose of anthelmintic medication, autoinfective larvae may not be killed, and can return to the gut to mature to adulthood and produce larvae, including autoinfective larvae. This maintains infection or even steadily increases the intensity of infection. Hence, before a study can be accepted as providing evidence of the inadequacy of the strongyloides ELISA as a monitoring tool, it must make use of highly effective treatment. Currently this should entail repeated doses of ivermectin26. The optimal retreatment period is unknown but, on biological grounds, monthly retreatment may be preferable because the only experimental study to date showed that the prepatent period after infective larvae penetrated the skin was 28 days27. Retreatment on at least two occasions with one month intervals after the initial dose (ie, three monthly doses) will theoretically kill adults derived from autoinfective larvae, hopefully before they produce autoinfective larvae themselves. A limited number of studies that have adopted this approach to treatment have shown that strongyloides ELISA will revert to negative in the majority of patients13. However, additional studies are required to confirm the utility of strongyloides ELISA as a monitoring tool after effective treatment.
Cost-benefit of strongyloides ELISA
The cost-benefit of a diagnostic test is related to its positive predictive value (ie, yield) and thus the particular disease prevalence in specific populations. This is well illustrated using the Australian situation. In Australia, strongyloidiasis is a problem restricted to rural and remote Aboriginal communities in the tropics1,28,29, immigrants from high endemicity countries particularly Southeast Asia19,30, returned prisoners of war from Southeast Asia31,32, and military personnel that have served in Southeast Asia33. Strongyloidiasis is uncommon in Australian urban communities because faeces are safely deposited in toilets and the life cycle cannot continue34. In some Australian Aboriginal communities the prevalence of strongyloidiasis in cross-sectional faecal surveys is 15% or greater, implying a higher true infection rate because faecal examination is a less sensitive diagnostic technique28,35-37. For illustrative purposes, the present study will use a prevalence of 20%.
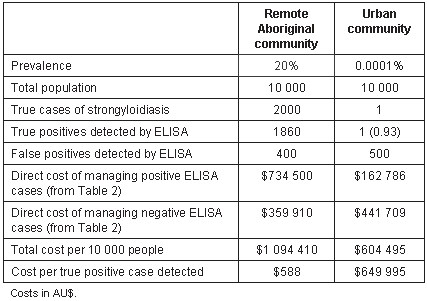
In the urban Australian community, strongyloidiasis is a very rare disease; the present study will use an arbitrary prevalence of 1/10 000, which is probably an overestimate. What is the cost-benefit of screening with the strongyloides ELISA in each community? Since humans, including clinicians feel more comfortable interpreting natural frequencies rather than probabilities, the present study will use the former38. Of 10 000 members of the rural Aboriginal community with a prevalence of 20%, 2000 can be expected to have strongyloidiasis. If these individuals are tested with the Australian strongyloides ELISA with a sensitivity and specificity of 93% and 95% respectively7, 1860 true positives will be detected and 400 false positives. Of the urban group of 10 000, approximately one true positive (actual value = 0.93) and 500 false positives will be detected. Thus, the number needed to test to detect one true positive will be 5.4 and 10 753 for the rural Aboriginal and urban groups, respectively.
Another useful perspective is to consider the number of people who test positive who are true positives. This is a particularly useful perspective for individual patients from these communities, because there are considerable costs, both psychological and in terms of exposure to further investigation or treatment, if a person is falsely 'diagnosed' as infected6. For every 1000 rural Aboriginal Australians who have a positive strongyloides ELISA, 823 will be true positives; while for every 1000 urban people with a positive test, only 2 (actual value = 1.99) will have strongyloidiasis. Clearly, for the urban group with its low prevalence, the costs of testing exceed the benefits, while for high prevalence rural Aboriginal communities, the benefits are much greater. Using figures for direct costs of detection and treatment only (Table 2), every true positive case detected in the remote Aboriginal community costs AU$588 to manage, while every true positive case detected in the urban community costs AU$649 995 (Table 3). If the focus was further refined to individuals within Aboriginal communities who were symptomatic, with respiratory, gastrointestinal or cutaneous symptoms/signs, the cost per positive case detected would decrease further3,9,39.
Table 3: Estimate of direct cost of detecting a true positive using the strongyloides ELISA (sensitivity 93%, specificity 95%) in remote northern Aboriginal community and urban community in Australia. Natural frequencies are used36 with a base population of 10 000 chosen to give whole integers in urban population.
This is a simplistic analysis that assumes 100% success in treatment and does not allow for indirect costs. It is only a beginning. From a public health perspective other costs must be known before decisions on screening can be undertaken, in particular the medical impact of untreated chronic strongyloidiasis. Chronic strongyloidiasis is symptomatic in approximately 70% of cases3,39, but the cost of ongoing illness has not been calculated. Avoidance of episodes of severe disease by prompt treatment of chronic strongyloidiasis and prevention of progression to hyperinfection must be factored in, as any intensive hospital treatment immensely increases cost. One episode of hyperinfection treated in an intensive care unit may cost US$80 000 in the USA40. This would appear to be an overestimate of costs in the Australian system, but no figures are available for Australia. Severely ill patients in isolated, remote northern Australian Aboriginal communities are evacuated to major hospitals by air at a cost of $6 per km (B. McGuire, Royal Flying Doctor Service, pers. comm., 2004). Thus, an emergency evacuation may cost from $3000 to $10 000 depending on the distance flown. Severe strongyloidiasis occurs in remote Aboriginal communities36,41-44, but the incidence of severe strongyloidiasis, including death, is not known. To obtain a comprehensive cost-benefit analysis prior to screening communities with strongyloides ELISA, data is needed on the cost of untreated strongyloidiasis and severe strongyloidiasis, as well as reasonably accurate estimates of the prevalence of strongyloidiasis in the communities of interest. Additional cost data is needed for communities with different prevalences in order to construct a cost-benefit algorithm. Health departments also need to decide how much they are willing to spend on detecting and treating a positive case. Consultation costs for detecting strongyloidiasis could be decreased in rural and remote tropical Indigenous communities by including serological screening for strongyloidiasis in well-person health checks with the other chronic diseases currently included.
Conclusion
In summary, although a new strongyloides ELISA or a new test with higher sensitivity and specificity would be ideal, the current ELISA is a useful tool for diagnosing strongyloidiasis and for monitoring response to adequate treatment. It is of particular value in communities and subgroups hyperendemic for strongyloidiasis where the cost, per positive case detected, is considerably lower. Phase III trials9 should be carried out for subgroups selected on the probability of having strongyloidiasis. To better define the role of the strongyloides ELISA, we require a more complete understanding of the length of the window period to seroconversion after infection, and whether failure of the titre to fall to negative values always indicates treatment failure. To make fully informed decisions about screening endemic communities, data on the prevalence and costs of chronic and severe strongyloidiasis are needed. Although community-wide interventions for strongyloidiasis has been advocated17,45 and implemented on a limited number of occasions46, comprehensive data are not available on the most effective approach in the Australian situation. Any intervention program should be evaluated in terms of epidemiological parameters (prevalence, intensity and transmission dynamics of infection), impact on general health (clinic attendances, hospitalisations, time off work, quality of life), and direct and indirect economic costs and benefits to allow development of appropriate cost-benefit algorithms.
References
1. Adams M, Page W, Speare R. Strongyloidiasis: an issue in Aboriginal communities. Rural and Remote Health 3: 152. (Online), 2003. Available: http://rrh.org.au (Accessed 28 Sept 2004).
2. McCarthy JS, Garrow SC. Parasite elimination programs: at home and away. Medical Journal of Australia 2002; 176: 456-457.
3. Sudarshi S, Stümpfle R, Armstrong M, et al. Clinical presentation and diagnostic sensitivity of laboratory tests for Strongyloides stercoralis in travelers compared with immigrants in a non-endemic country. Tropical Medicine and International Health 2003; 8: 728-732.
4. Sackett DL, Haynes RB, Guyatt GH, Tugwell P. Clinical epidemiology, a basic science for clinical medicine. 2nd edn. Boston: Little Brown, 1991.
5. Siddiqui A, Berk S. Diagnosis of Strongyloides stercoralis infection. Clinical Infectious Diseases 2001; 33: 1040-1047.
6. Gigerenzer G. Reckoning with risk: learning to live with uncertainty. London: Penguin Books, 2002.
7. Grove DI. Diagnosis. In: DI Grove (ED.). Strongyloidiasis: an important roundworm infection of man. London: Taylor Francis, 1989; 175-197.
8. Grove DI. Human strongyloidiasis. Advances in Parasitology 1996; 38: 251-309.
9. Sackett DL, Haynes RB. The architecture of diagnostic research. BMJ 2002; 324: 539-541.
10. Black MA, Craig BA. Estimating disease prevalence in the absence of a gold standard. Statistical Medicine 2002; 21: 2653-2669.
11. Johnson WO, Gastwirth JL, Pearson LM. Screening without a 'gold standard': the Hui-Walter paradigm revisited. American Journal of Epidemiology 2001; 153: 921-924.
12. Genta RM. Global prevalence of strongyloidiasis: critical review with epidemiological insights into the prevention of disseminated disease. Review of Infectious Disease 1989; 11: 755-767.
13. Sato Y, Kobayashi J, Toma H, Shiroma Y. Efficacy of stool examination for detection of Strongyloides infection. American Journal of Tropical Medicine and Hygiene 1995; 53: 248-250.
14. First National Workshop on Strongyloidiasis. Recommendations of the First National Workshop on Strongyloidiasis. Nhulunbuy, East Arnhem Land. (Online) 2001. Available: http://www.jcu.edu.au/school/phtm/PHTM/ss/1rec.htm (Accessed 28 Sept 2004).
15. Elgart GW, Meinking TL. Ivermectin. Dermatology Clinics 2003; 21: 277-282.
16. Adenusi AA, Oke AO, Adenusi AO. Comparison of ivermectin and thiabendazole in the treatment of uncomplicated human Strongyloides stercoralis infection. African Journal of Biotechnology 2003; 2: 465-469.
17. Speare R, Durrheim D. Mass treatment with ivermectin: an underutilized public health strategy. Bulletin of the World Health Organization 2004; 8: 562.
18. Bailey JW. A serological test for the diagnosis of strongyloides antibodies in ex Far East prisoners of war. Annals of Tropical Medicine and Parasitology 1989; 83: 241-247.
19. Atkins NS, Lindo JF, Lee MG et al. Humoral responses in human strongyloidiasis: correlations with infection chronicity. Transactions of the Royal Society of Tropical Medicine and Hygiene 1997; 91: 609-613.
20. Sampson IA, Grove DI. Strongyloidiasis is endemic in another Australian population group: Indochinese immigrants. Medical Journal of Australia 1987; 146: 580-582.
21. Grove DI, Northern C. Infection and immunity in dogs infected with a human strain of Strongyloides stercoralis. Transactions of the Royal Society of Tropical Medicine and Hygiene 1982; 76: 833-838.
22. Kobayashi J, Sato Y, Toma H, Takara M, Shiroma Y. Application of enzyme immunoassay for post chemotherapy evaluation of human strongyloidiasis. Diagnostic Microbiology and Infectious Diseases 1994; 18: 19-23.
23. Grove DI, Lumsden J, Northern C. Efficacy of albendazole against Strongyloides ratti and S. stercoralis in vitro, in mice, and in normal and immunosuppressed dogs. Journal of Antimicrobial Chemotherapy 1988; 21: 75-84.
24. Speare R. Identification of species of Strongyloides. In: DI Grove (Ed.). Strongyloidiasis: an important roundworm infection of man. London: Taylor Francis, 1989; 11-82.
25. Speare R, Tinsley DJ. Strongyloides felis: an 'old' worm rediscovered in Australian cats. Australian Veterinary Practitioner 1986; 16: 10-18.
26. Zaha O, Hirata T, Kinjo F, Saito A, Fukuhara H. Efficacy of ivermectin for chronic strongyloidiasis: two single doses given 2 weeks apart. Journal of Infection and Chemotherapy 2002; 8: 94-98.
27. Tanaka H. Experimental and epidemiological studies on strongyloidiasis of Amami Oshima Island. Japanese Journal of Experimental Medicine 1958; 28: 159-182.
28. Kukuruzovic RH, Robins Browne RM, Anstey N, Brewster DR. Enteric pathogens, intestinal permeability and nitric oxide production in childhood diarrheal diseases. Paediatric Infectious Disease Journal 2002; 21: 730-739.
29. Prociv P, Luke R. Observations on strongyloidiasis in Queensland aboriginal communities. Medical Journal of Australia 1993; 158: 160-163.
30. de Silva S, Saykao P, Kelly H et al. Chronic Strongyloides stercoralis infection in Laotian immigrants and refugees 7-20 years after resettlement in Australia. Epidemiology and Infection 2002; 128: 439-444.
31. Grove DI. Strongyloidiasis in Allied ex-prisoners of war in southeast Asia. BMJ 1981; 280: 598-601.
32. Oliver NW, Rowbottom DJ, Sexton P et al. Chronic strongyloidiasis in Tasmanian veterans - clinical diagnosis by the use of a screening index. Australia New Zealand Journal of Medicine 1989; 19: 458-462.
33. Commonwealth Department of Veterans' Affairs. Morbidity of Vietnam Veterans. A study of the health of Australia's Vietnam Veteran Community. Canberra: Commonwealth Department of Veterans' Affairs, 1998.
34. Grove DI. Worms in Australia. Medical Journal of Australia 1993; 159: 464-466.
35. Aland K, Prociv P, Currie B, Jones H. Worm Project at Galiwin'ku. Working Together 1996; 6: 10.
36. Flannery G, White N. Immunological parameters in northeast Arnhem Land Aborigines: consequences of changing settlement and lifestyles. In: LM Schell, MT Smith, A Bilsborogh (Eds). Urban Ecology and Health in the Third World. Cambridge University Press, Cambridge, 1993.
37. Davis JS, Currie BJ, Fisher DA et al. Prevention of opportunistic infections in immunosuppressed patients in the tropical Top End of the Northern Territory. Northern Territory Disease Control Bulletin 2004; 11: 7-13.
38. Gigerenzer G, Edwards A. Simple tools for understanding risks: from innumeracy to insight. BMJ 2003; 327: 741-744.
39. Grove DI. Clinical manifestations. In: DI Grove (Ed.). Strongyloidiasis: an important roundworm infection of man. London: Taylor Francis, 1989: 155-173.
40. Nair D. Screening for Strongyloides infection among the institutionalized mentally disabled. Journal of the American Board of Family Practitioners 2001; 14: 51-53.
41. Mak DB. Recurrent bacterial meningitis associated with strongyloides hyperinfection. MJA 1993; 159: 354.
42. Fisher D, McCarry F, Curry B. Strongyloidiasis in the Northern Territory. Under-recognised and under-treated? Medical Journal of Australia 1993; 159: 88-90.
43. Walker AC, Blake G, Downing D. A syndrome of partial intestinal obstruction due to Strongyloides stercoralis. Medical Journal of Australia 1976; Suppl 1: 47-48.
44. Walker-Smith JA, McMillan B, Middleton AW, Robertson S, Hopcroft A. Strongyloidiasis causing small-bowel obstruction in an Aboriginal infant. Medical Journal of Australia 1969; 2: 1263-1265.
45. Conway DJ, Lindo JF, Robinson RD, Bundy DAP. Towards effective control of Strongyloides stercoralis. Parasitology Today 1995; 11: 421-424.
46. Toma H, Shimabukuro I, Kobayashi J, Tasaki T, Takara M, Sato Y. Community control studies on Strongyloides infection in a model island of Okinawa, Japan. Southeast Asian Journal of Tropical Medicine and Public Health 2000; 31: 383-387.
Abstract
General practitioners who care for Aboriginal patients in rural and remote communities from tropical Australia must be aware of strongyloidiasis. The prevalence of this parasitic infection is high and occasional cases can have a fatal outcome. Other groups in Australia at high risk of strongyloidiasis are immigrants from endemic countries, particularly from Southeast Asia, and military personnel who have served overseas in endemic areas. Elimination programs for enteric parasites in rural Australian Indigenous communities are so important that a nationally coordinated approach has been advocated. Wisely used, cost-effective diagnostic tests are a critical component of an elimination program. Strongyloidiasis must be confirmed by laboratory diagnosis and the strongyloides ELISA, although not ideal, is a useful test that can be used to diagnose strongyloidiasis and to monitor cure. In this article the value of the current strongyloides ELISA is discussed and a cost-benefit analysis is conducted using direct costs only. In a typical rural Aboriginal community in tropical Australia with prevalence of strongyloidiasis at 20% each true positive case detected by the strongyloides ELISA is estimated to cost approximately AU$590 to diagnose and manage until cure, proven by negative serology.
Key words: clinical epidemiology, cost-benefit, diagnosis, Indigenous Australians, serology, Strongyloides stercoralis.
You might also be interested in:
2009 - Education to improve cancer care in rural South Australia

