Routine mammography screening and treatment advances have resulted in substantial reductions in breast cancer mortality1-3. With the notable exception of the United States Preventive Services Task Force (USPSTF)4, most major professional organizations, including the American Cancer Society, National Comprehensive Cancer Network, American College of Radiology, and American Congress of Obstetricians and Gynecologists, suggest annual mammogram screening in women starting at 40 years of age5-8. Additionally, it was recently demonstrated that missing even one annual mammogram increases the risk of being diagnosed at a later stage9. Despite the prognostic significance of early detection10,11, rates of mammography screening in eligible women in the USA have reached no higher than 80% and rarely exceed 50% in most localities12. Nationwide, the average rate of mammography screening for women older than 40 years has remained between 50% and 80% since 200013. Several patient characteristics, including physician access, past screening behavior, socioeconomic barriers, racial and ethnic differences, and age, have been identified as factors that influence mammogram utilization14. The authors recently identified travel time as an additional important factor, and the issues of geographical access and the influence of climate were further explored9.
In rural regions, access to mammography screening can be problematic, and several studies suggest that longer travel distances adversely affect early detection of breast cancer in rural populations15-17. Other studies have reported mixed results18-21. Additionally, Oh et al have suggested seasonal variation in breast cancer diagnosis, specifically noting a trough in breast cancer diagnosis in the winter months22. Studies of mammography access by location are improving as geographic information systems (GIS) technology is refined to more accurately determine point-to-point distance and travel time via road networks. The goal of the present study was to utilize modern GIS technology to determine the association between geographic proximity to mammogram centers and breast cancer stage at diagnosis in a single healthcare system that serves the majority of residents in north-central Wisconsin, USA, a predominantly rural agricultural region. In light of the potential for inclement winter weather to affect travel conditions in the studied service area, seasonal mammogram variability was also explored.
Patient data
Female patients diagnosed with primary breast cancer at any of the over 50 system-wide healthcare facilities between 1 January 2002 and 30 December 2008 were identified electronically through the local cancer registry23. Data regarding number of medical encounters during the study period, mammograms within the 5 years before breast cancer diagnosis, and breast cancer stage at diagnosis were abstracted electronically and by manual chart abstraction when necessary. Manual validation was performed on 10% of records. Patients diagnosed at facilities outside the healthcare system, male breast cancer patients, and patients whose physical address of residence was located outside of Wisconsin and neighboring Illinois, Michigan, and Minnesota were excluded.
Comprehensive capture of mammogram screening information was achieved via electronic search for mammogram-related procedure codes (ICD-9 codes 793.8X, V76.11, V76.12) and appointment types. Procedure codes included codes for computer mammogram add-on, radiograph of mammary duct(s), mammogram (one breast, both breasts, or screening), computer-aided detection of diagnostic or screening mammography, mammary ductogram or galactogram multiple, mammography (unilaterial or bilateral), screening mammography (bilateral or digital), and diagnostic mammography digital. Appointment types included 31 mammography-related codes. Most mammograms were identified by procedure and radiology and/or appointment type (89.7%). All others were manually adjudicated to ensure accuracy. Diagnostic mammograms were excluded from analyses when done subsequent to a screening mammogram.
Screening mammograms were annualized so that number of missed mammograms could be determined for each patient. For analysis purposes, the number of missed mammograms is defined as the number of 1-year (365 days) periods subjects went without having at least one mammogram before breast cancer diagnosis based on the recommendations of most major professional organizations5-8 and recent findings demonstrating that missing even one annual mammogram results in an increased risk for later stage at diagnosis9. For analysis of stage, patients were dichotomized into early- and late-stage categories. Patients with stages 0-2 breast cancer were considered early stage and patients with stages 3-4 breast cancer were considered late stage.
Geocoding facility locations
A total of 22 facilities in Wisconsin were included. Facility data included name, physical street address, city, state, ZIP code, and status as either a fixed screening location or mobile screening unit. Facility locations were geocoded as point features based on the physical street address of each facility. Mobile facilities were geocoded at the location where the mammogram screening was conducted. In analyses, no distinction was made between fixed and mobile locations, as mobile locations are consistent on a recurrent basis.
ESRI ArcGIS Desktop v10.0, ArcInfo license, Business Analyst extension, and StreetMap Premium using data from TeleAtlas 2010 were used to geocode point features (http://www.esri.com/software/arcgis). Business Analyst Store Setup wizard was used to determine facility locations andArcGIS Geocoding toolbar, Review/Rematch Addresses tool was used to substantiate geocoded results.
Geocoded facility locations were validated by comparing geocoded point features to full-color orthophotographs acquired in 2010 by the National Agricultural Inventory Program. Visual inspection verified existence of the facility locations. One geocoded facility was found to be misplaced. Two distinct geocoded locations identified the same address, but in two different counties. The spatial feature was moved based on the correct address match.
Geocoding patient residence
Patient residences were geocoded as point features based on the physical street address noted in the electronic medical record (EMR). ESRI ArcGIS Desktop version 10.0, ArcInfo license, Business Analyst extension, and StreetMap Premium using data from TeleAtlas 2010 was used to geocode point features. Business Analyst, Customer Setup wizard was used to initially determine patient residence location. ArcGIS Geocoding toolbar, Review/Rematch Addresses tool was used to substantiate geocoded results, with a match score of 100 indicating a best match. A best match is attained when an address in the project database matches perfectly with an address in the streets. Lower scores indicate inconsistency between an address in the two databases. Inconsistencies frequently occur due to misspellings, abbreviations (e.g., St., Street, or Str.), and street direction (left or right side of the street). Other inconsistencies may result from errors in city, state, and ZIP code information.
Geocoded features with a score <100 (n=424) were manually investigated. Parcel data from Wisconsin counties were used to identify and correct addresses based on the comparison between the parcel's address and the geocoded address. The TerraServer online imagery service (http://www.terraserver.com) was used to verify an address and acquire latitude and longitude coordinates. Latitude and longitude coordinates were used as a substitute for a positive address match (ie score of 100).
Patient residences describing a post office box or rural route, or with missing or unidentifiable addresses that could not be geocoded, were geolocated to the centroid of the patient's ZIP code using the Mean Center tool and ArcToolbox. Geocoded residences for 77 patients were documented this way.
A random sample of 100 patient addresses with a geocoded score of 100 was inspected further by comparison to orthophotographic images, as described above. There were 96 point features found to have a residence within 500 ft (152.4 m) of the geocoded address. Four geocoded addresses had a residence identified at a distance >500 ft and two were within 1200 ft (365.8 m) of the actual location. The remaining two addresses did not have an identifiable residence within close proximity to the geolocated address.
Distance to closest facility analysis
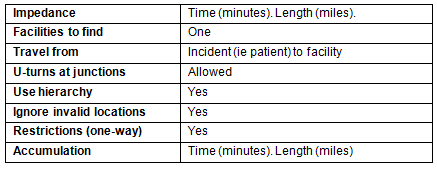
The distance to closest facility analysis measured distance and time along a road network between a patient and their closest mammogram facility, which may or may not have been the facility at which they received cancer screening services. ESRI ArcGIS Desktop v10, ArcInfo license, and the Network Analyst Extension and Toolbar were used to conduct this analysis. Parameters shown in Table 1 were applied using Network Analyst's New Closest Facility analysis. The author institution's fixed and mobile facility point features (22 features) were loaded as the Facilities layer. The result of the New Closest Facility analysis created a time and distance attribute for each record loaded in the Incidents layer.
Table 1: Network Analyst's New Closest Facility analysis
parameters for distance to nearest/visited facility
Distance to visited facility analysis
The distance to visited facility analysis measured distance and time along a road network between a patient and the facility where they received cancer screening services, as recorded in the EMR. Network Analyst's New Closest Facility analysis was conducted 22 times, once for each fixed and mobile facility. Patient records (1421) were extracted into 22 separate feature classes based on facility visited. Parameters shown in Table 2 were applied using Network Analyst's New Closest Facility analysis. The author institution's fixed and mobile facility point features were loaded individually as the Facilities layer. Patient residence location point features that visited the facility were loaded as the Incidents layer. The result of the New Closest Facility analysis created a time and distance attribute for each record loaded in the 22 Incidents layers.
Of 1421 patients, 482 did not have data recording the facility visited for cancer screening services; they were assumed to have visited the clinic in the central location, based on project data revealing that 746 of 939 (79.4%) patients with a recorded facility address visited the facility in the central location in Wisconsin.
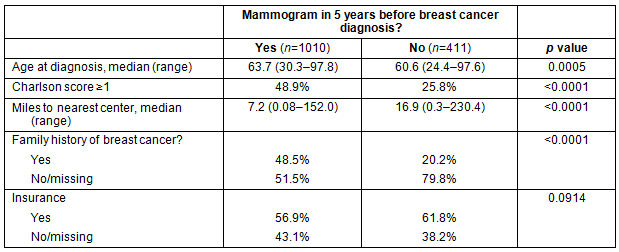
Table 2: Characteristics of women with and without mammogram in 5 years before breast cancer diagnosis
Seasonal variation
Seasons were defined using northern hemisphere calendar dates of equinoxes and solstices: spring (20 March - 19 June), summer (20 June - 20 September), autumn/fall (21 September - 20 December), and winter (21 December - 19 March). The Wisconsin State Climatology Office reports mean winter temperature ranges in Wisconsin of 0-20ºF (-18ºC to -7ºC), with an average monthly snowfall range of 8-14 inches (20-35 cm) and the potential for 10 or more major winter storms per season24.
Statistical analyses
The Wilcoxon rank-sum test was used to analyze differences in time to nearest facility in subjects by number of missed mammograms and breast cancer stage. Multinomial logit analysis was performed to analyze the relationship between later stage diagnosis of breast cancer and time to nearest facility for all subjects, or time to visited facility for those subjects who had had at least one mammogram. To account for factors with the potential to influence mammogram utilization or stage at diagnosis, multinomial logit analyses were adjusted for age, insurance status, number of medical encounters, comorbidities, family history of breast cancer, and calendar year. In addition, χ2 testing was used to determine whether seasonal mammogram distribution was even and if there was an association between season and distance to the nearest facility. All analyses were conducted using Statistical Analysis System v9.2 software (SAS; http://www.sas.com) and p values <0.05 were considered significant.
Ethics approval
Institutional Review Board approval was given, with waiver of informed consent; ethics approval number ONI11609.
A total of 1428 patients with breast cancer were identified systemwide for review, of which seven were excluded for incomplete mammography dates. Demographic characteristics for patients with and without at least one mammogram in the 5 years before breast cancer diagnosis are summarized in Table 2, and suggest that women who had at least one mammogram were more likely to be older, have more comorbidities, live closer to a mammogram facility, and to have a known family history of breast cancer.
Mammograms were annualized, and number of missed mammograms was assessed with respect to time to the nearest facility (Table 3). The difference was significant, and women who missed none of their five annual mammograms lived a median of 15 minutes from the nearest facility, while those who missed five of their past five annual mammograms lived nearly twice as far, with a median travel time of 27 minutes to the nearest facility (p<0.0001) (Table 3). Similarly, a trend in the relationship between travel time to nearest mammogram facility and stage of breast cancer at diagnosis was observed, with travel time increasing as cancer stage increased, from a median 17 minutes for subjects with stage 0 breast cancer to 24 minutes for subjects with stage 4 breast cancer (p=0.0586; Fig1).
Despite the trend toward increasing travel time with increasing cancer stage, the odds ratio (OR) of being diagnosed with breast cancer at a later stage (stages 3-4) was not significantly increased with increasing time to nearest facility or time to visited facility (Table 4). However, there was a trend toward increasing OR with increasing time to visited facility, reaching 1.63 in women who travelled 60 minutes or longer to a mammogram facility compared to those who travelled less than 5 minutes.
Analysis of seasonal mammogram distribution revealed significantly fewer screening mammograms performed during the winter months (Table 5). To investigate the possibility that this was related to travel difficulties, seasonal mammogram distribution was compared in women with distances of 30 mi (48.3 km) or less to the nearest mammogram facility to those with greater than 30 mi to travel. A significant trend toward decreased mammography in the winter months in women with more than 30 mi to travel was noted (Table 5), suggesting that winter weather may affect geographical access in such women. As healthcare providers often recommend annual mammogram screening coinciding with birthday, the number of missed mammograms by month and season of birth was also assessed. No difference was found based on birth month (data not shown), suggesting that date of annual mammogram may not be tied to season of birth in the system.
Table 3: Number of missed annual mammograms and time to nearest facility
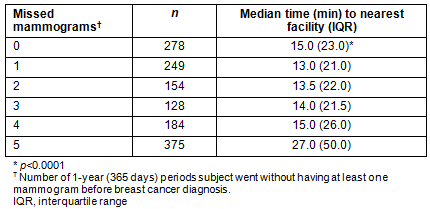
Table 4: Odds ratio of cancer diagnosis at late stage (stage 3-4) by time to nearest or visited facility
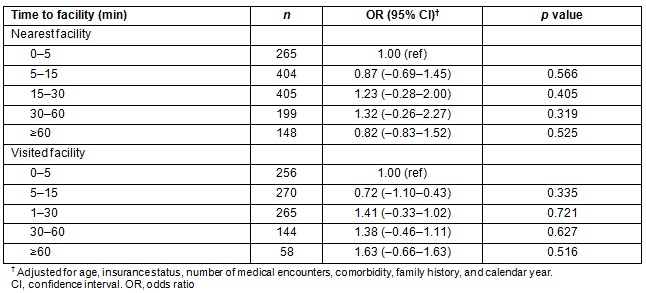
Table 5: Seasonal mammogram distribution
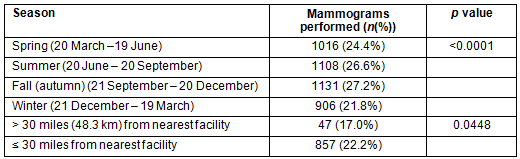
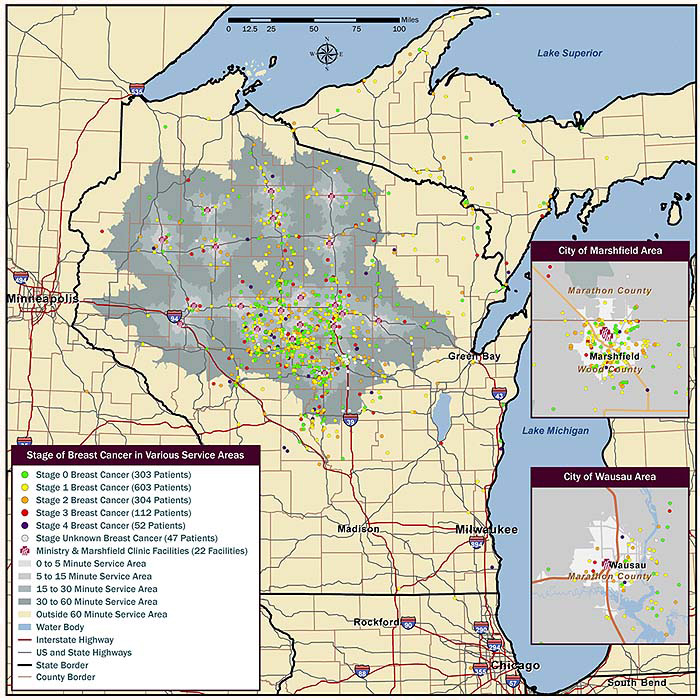
Figure 1: Stage of breast cancer and distance between patient and nearest mammogram facility. Map portrays the stage
of breast cancer at diagnosis, as indicated by colored dot, in comparison to physical distance, by street network,
between place of residence and the closest mammogram facility. Travel time in minutes is indicated by shading.
Discussion
Routine mammography provides opportunities for earlier breast cancer diagnosis. It has been demonstrated that missed mammograms, even 1 year before diagnosis, increase risk of later stage breast cancer9,25. Evidence suggests that regular mammography screening may be limited by geographical access to mammography centers, and that longer travel distances adversely affect early detection of breast cancer in rural populations15-17, although results are mixed18-21. The data presented here support the notion that longer travel time may be related to missed mammograms and later stage at diagnosis of breast cancer. A novel effect of season, whereby winter weather appears to result in reduced mammogram utilization, is also shown here.
In a recent, large-scale analysis of 161 619 women diagnosed with breast cancer around the USA, Henry et al found no relationship between travel time to mammogram facility and stage of breast cancer at diagnosis26. However, consistent with the findings presented here, several other region-specific studies have noted effects of travel time or distance on breast cancer stage. Huang et al examined distance between residence and nearest mammogram facility as a risk factor for advanced stage diagnosis in rural Kentucky, finding that women with advanced stage cancer had longer average travel distances than those diagnosed at an early stage17. Women who traveled 15 mi (24.1 km) or more were 1.5 times more likely to be diagnosed with late stage disease than those who lived within 5 mi (8.0 km) of a mammogram center. Additional studies in Kansas and the UK have demonstrated a similar relationship between shorter travel distance and early detection15,16. Huang et al hypothesized that geographic access may be affected by factors other than distance and road networks, including topography (eg mountainous terrain) and climate (eg heavy snow, icy conditions)17. Geographic area may also be associated with other, often unmeasured or unquantifiable, variables related to population in residence, quality of facilities available, structure of healthcare delivery systems, local public health measures, and availability of transportation. Therefore, studies that carefully examine a given service area, as opposed to nationwide analyses, may be warranted for improvement of mammogram utilization.
The healthcare system described in this study is a multicenter, multi-specialty system in north central Wisconsin, USA that serves the medical needs of the predominately rural community in which it resides. In the present study, an inverse relationship between distance from the nearest mammogram facility and mammogram utilization as well as breast cancer stage at diagnosis was observed. Interestingly, this relationship was not linear. The median time for women with 0-4 missed mammograms in the 5 years before breast cancer diagnosis ranged from approximately 13-15 minutes. However, when all five mammograms were missed, median time nearly doubled. The reasons for this lack of linearity are currently unclear, but may suggest that a threshold exists somewhere between 15 and 30 minutes of travel time, or that the group that never had a mammogram in the 5 years before diagnosis may be more heterogeneous with respect to mammogram frequency than those in the other groups. Considering mammography in the 5 years before breast cancer diagnosis as a dichotomous variable (ie 'yes' or 'no') may be of interest in this case, whereby women who had at least one mammogram during this time period had a median travel time of approximately 14 minutes, whereas those without had a median travel time of approximately 27 minutes.
An interesting seasonal distribution of mammogram utilization that illustrates the important role climate and weather patterns may play in modifying geographical access was also noted. Mammogram use was lower in the winter than in other months of the year, and this seasonal discrepancy was even more prominent in women who lived 30 minutes or more from the nearest mammogram center. Oh et al recently reported globally consistent seasonal patterns of breast cancer incidence22. In nearly 3 million cases of breast cancer worldwide, diagnoses were significantly more common during the spring and autumn/fall in both the northern and southern hemispheres. This pattern became more prominent as distance from the equator increased, and latitude dependence was most pronounced in women living in rural areas22. Oh et al proposed a relationship between diagnosis and local seasonal changes in the length of the day and effects on vitamin D and melatonin levels22. The results reported here suggest that impaired travel as a result of inclement weather may impede use of mammogram services during the winter months and may account for some of the trough in breast cancer diagnoses observed during the winter in areas further from the equator. Seasonality was more pronounced in women living more than 30 minutes from the nearest mammogram facility in the present study, consistent with Oh et al's finding that patterns were more pronounced in women living in rural areas22. A 2006 study by Celaya et al found winter weather to have a negative impact on breast cancer treatment in rural New Hampshire, USA27. The same group later demonstrated a borderline association between diagnosis during the winter months and later stage28; however, the present study is the first to describe a potential impact of weather on screening behaviors.
Many mammogram reminder programs incorporate reminder phone calls or mailed reminder materials coinciding with a woman's birthday29,30. To assess whether such practices influenced receipt of mammograms in women born during the winter, the effect of month and season of birth of mammogram utilization was examined. In the population examined here, there was no association between month or season of birth and mammogram utilization. However, in systems where birthdays or other anniversaries are heavily used as reminder triggers, particularly in geographical locations where winter weather may be an impediment for travel, season and the practice of using birthdays or other anniversaries for mammogram reminder programs deserve further consideration.
Most studies of travel time and breast cancer stage use diagnostic mammogram centers or assume routine mammography screening at the nearest facility. In the current study, the authors collected information regarding screening mammograms over a 5-year period and assessed the relationship between both travel time and breast cancer stage as well as travel time and routine mammography screening. Despite these strengths, limitations include those inherent to retrospective studies, namely use of data on hand and as reported. The small number of patients included likely explains the failure to reach statistical significance when assessing the OR of late stage cancer in patients by travel time, despite the trends observed. Also, the authors were unable to account for women who sought screening at other institutions. However, alternative mammography centers outside the authors' healthcare system are limited or non-existent in most of the service areas. Additionally, in women known to receive mammography services at other institutions, the radiology department at the author institution obtains reports and films for comparison. An additional factor worth consideration is access to transportation as a potentially important modifier, especially since public transportation is quite limited throughout the relatively rural service area. A final limiting factor of this study is related to diagnosis of breast cancer in women under the age of 40 years. Such women would not be expected to receive any screening mammograms prior to breast cancer diagnosis. In the current study, 56 of 1421 subjects received a diagnosis of breast cancer before they were 40 years old, but were included in the analyses. To determine whether this may have influenced study results, the authors examined the distribution of early- and late-stage breast cancer in women younger than 45 years and found that the distribution was similar to that of the entire study population, suggesting that the inclusion of these women was unlikely to have altered study outcomes.
In the rural north-central Wisconsin service area served by this healthcare system, travel time to the nearest mammogram center appears to be inversely related to regular mammography screening and breast cancer stage at diagnosis. Mammography screening is undertaken less frequently in the winter months, and this seasonal discrepancy is even more pronounced in women traveling 30 minutes or more to the nearest center. Healthcare delivery system-specific studies are warranted to investigate the factors of greatest importance within a given geographical service area. In rural areas, particularly in the northern USA, healthcare providers should ask female patients about geographic access to mammogram facilities, make recommendations for mammogram screening at every visit, and consolidate services when possible. Medical assistants and appointment coordinators should work to schedule patients living further from mammogram facilities at times of the year when travel is less likely to be impeded by inclement weather. When appointments are canceled or missed during the winter months, every effort should be made to contact patients and reschedule to ensure annual screening. Plans to increase mammogram coverage may be most effective if implemented during the non-winter months, and improved access to mammography services during the winter may require utilization of mobile mammogram units in remote areas. Finally, the practices of linking birthday or other anniversaries to mammogram reminders may be worth further consideration in areas where season may influence a patient's ability to access a facility.
Acknowledgements
The authors would like to acknowledge Marie Fleisner of the Marshfield Clinic Office of Scientific Writing and Publication for editorial assistance with this manuscript. Funding for this study was provided by the Marshfield Clinic Research Foundation's Disease Specific Funds.
References
1. Tabár L, Vitak B, Chen HH, Yen MF, Warwick J, Smith RA, et al. The Swedish Two-Country Trial twenty years later. Updated mortality results and new insights from long-term follow-up. Radiology Clinics of North America 2000; 38(4): 625-651.
2. Humphrey LL, Helfand M, Chan BK, Woolf SH. Breast cancer screening: a summary of the evidence for the US Preventive Services Task Force. Annals of Internal Medicine 2002; 137(5 Part 1): 347-360.
3. Tabár L, Yen MF, Vitak B, Chen HH, Smith RA, Duffy SW. Mammography service screening and mortality in breast cancer patients: 20-year follow-up before and after introduction of screening. Lancet 2003; 361(9367): 1405-1410.
4. U.S. Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Annals of Internal Medicine 2009; 151(10): 716-726.
5. American Cancer Society. Breast cancer: early detection. (Online) 2011. Available: www.cancer.org/acs/groups/cid/documents/webcontent/003165-pdf.pdf (Accessed 30 July 2012).
6. National Comprehensive Cancer Network. NCCB clinical practice guidelines in oncology (NCCN Guidelines): breast cancer screening and diagnosis, version 1. 2012. (Online) 2012. Available: http:www.NCCN.org (Accessed 28 September 2012).
7. American College of Radiology. ACR practice guidelines for the performance of screening and diagnosis mammography. In ACR practice guidelines and technical standards (Online) 2008. Available: http://www.acr.org (Accessed 26 February 2013).
8. American College of Obstetricians-Gynecologists. Practice bulletin no. 122: breast cancer screening. Obstetrics and Gynecology 2011; 118(2 Pt 1): 372-382.
9. Onitilo AA, Engel JM, Liang H, Stankowski RV, Miskowiak DA, Broton M et al. Mammography utilization: patient characteristics and breast cancer stage at diagnosis. American Journal of Roentgenology 2013; 201(5): 1057-1063.
10. Carter TI, Reilly JJ. Missing opportunities: clinical antecedents in the diagnosis of advanced breast cancer. Annals of Surgical Oncology 2012; 19(9): 2782-2785.
11. Kremer ME, Downs-Holmes C, Novak RD, Lyons JA, Silverman P, Pharm RM et al. Neglecting to screen women between the ages of 40 and 49 years with mammography: what is the impact on breast cancer diagnosis? American Journal of Roentgenology 2012; 198(5): 1218-1222.
12. Hirsch BR, Lyman GH. Breast cancer screening with mammography. Current Oncology Reports 2011; 13(1): 63-70.
13. National Center for Health Statistics. Health, United States, 2011: with special feature on socioeconomic status and health. (Online) 2012. Available: www.cdc.gov/nchs/data/hus/hus11.pdf (Accessed 13 February 2013).
14. Schuler KM, Chu PW, Smith-Bindman R. Factors associated with mammography utilization: a systematic review of the literature. Journal of Womens Health (Larchmt) 2008; 7(9): 1477-1498.
15. Engelman KK, Hawley DB, Gazaway R, Mosier MC, Ahluwalia JS, Ellerbeck EF. Impact of geographic barriers on the utilization of mammograms by older rural women. Journal of the American Geriatrics Society 2002; 50(1): 62-68.
16. Maheswaran R, Pearson T, Jordan H, Black D. Socioeconomic deprivation, travel distance, location of service, and uptake of breast cancer screening in North Derbyshire, UK. Journal of Epidemiology and Community Health 2006; 60(3): 208-212.
17. Huang B, Dignan M, Han D, Johnson O. Does distance matter? Distance to mammography facilities and stage at diagnosis of breast cancer in Kentucky. Journal of Rural Health 2009; 25(4): 366-371.
18. Kreher NE, Hickner JM, Ruffin MT 4th, Lin CS. Effect of distance and travel time on rural women's compliance with screening mammography: an UPRNet study. Upper Peninsula Research Network. Journal of Family Practice 1995; 40(2): 143-147.
19. Secker-Walker RH, Vacek PM, Hooper JG, Plante DA, Detsky AS. Screening for breast cancer: time, travel, and out-of-pocket expenses. Journal of the National Cancer Institute 1999; 91(8): 702-708.
20. Brustrom JE, Hunter DC. Going the distance: how far will women travel to undergo free mammography? Military Medicine 2001; 166(4): 347-349.
21. Schroen AT, Lohr ME. Travel distance to mammography and the early detection of breast cancer. Breast Journal 2009; 15(2): 216-217.
22. Oh EY, Ansell C, Nawaz H, Yang CH, Wood PA, Hrushesky WJ. Global breast cancer seasonality. Breast Cancer Research and Treatment 2010; 123(1): 233-243.
23. Onitilo AA, Engel JM, Greenlee RT, Mukesh BN. Breast cancer subtypes based on ER/PR and Her2 expression: comparison of clinicopathologic features and survival. Clinical Medicine & Research 2009; 7(1-2): 4-13.
24. Wisconsin State Climatology Office. Wisconsin winter climate (December, January, and February). (Online) 2012. Available: http://www.aos.wisc.edu/~sco/seasons/winter.html (Accessed 8 January 2013).
25. Norman SA, Localio AR, Zhou L, Weber AL, Coates RJ, Malone KE et al. Benefits of screening mammography in reducing the rate of late-stage breast cancer diagnoses (United States). Cancer Causes & Control 2006; 17(7): 921-929.
26. Henry KA, Boscoe FP, Johnson CJ, Goldberg DW, Sherman R, Cockburn M. Breast cancer stage at diagnosis: is travel time important? Journal of Community Health 2011; 36(6): 933-942.
27. Celaya MO, Rees JR, Gibson JJ, Riddle BL, Greenberg ER. Travel distance and season of diagnosis affect treatment choices for women with early-stage breast cancer in a predominately rural population (United States). Cancer Causes and Control 2006; 17(6): 851-856.
28. Celaya MO, Berke EM, Onega TL, Onega TL, Gui J, Riddle BL et al. Breast cancer stage at diagnosis and geographic access to mammography screening (New Hampshire, 1998-2004). Rural and Remote Health 2010; 10(2): 1361.
29. Davis NA, Nash E, Bailey C, Lewis MJ, Rimer BK, Koplan JP. Evaluation of three methods for improving mammography rates in a managed care plan. American Journal of Preventive Medicine 1997; 13(4): 298-302.
30. Romaire MA, Bowles EJ, Anderson ML, Buist DS. Comparative effectiveness of mailed reminder letters on mammography screening compliance. Preventive Medicine 2012; 55(2): 127-130.
