In recent years, the introduction of two important pieces of legislation has created new opportunities for the integration of substance abuse treatment into primary care1-3. First, the 2008 Mental Health Parity and Addiction Equity Act requires that health insurance companies impose similar financial requirements and treatments caps for mental health and substance abuse treatments as for general health benefits4. The 2010 Affordable Care Act built on this by seeking to provide affordable health insurance to all US residents, including access to mental health and substance abuse treatment2,3. The requirement that health insurance companies provide substance abuse treatment is especially relevant regarding treatment for opioid dependence, which has reached epidemic levels in the USA5,6. In 2010, more than 12 million individuals used prescription opioids for non-medical reasons, and there are currently over 100 drug overdose deaths daily7,8. Effective treatments for opioid dependence exist, but currently fewer than 20% of individuals with opioid dependence are able to access them9.
Buprenorphine is a partial agonist with a very high binding affinity at the μ-opioid receptors10. This allows buprenorphine to both prevent opioid withdrawal and block the euphoria associated with opioid use. Maintenance therapy with buprenorphine, known as buprenorphine maintenance therapy (BMT), has been shown to reduce opioid use and overdose death in opioid-dependent populations11-15. Agonist therapy with a pharmacologic agent such as buprenorphine is considered first-line treatment for opioid dependence as it has been shown to reduce deaths by 30%, decrease HIV infections, and leads to lower rates of criminal activity16-22. Buprenorphine maintenance therapy is designed for office-based use and has been shown to be safe and effective in non-research primary care settings, even those with limited resources23,24. Offering BMT in primary care settings has the potential to reduce overall costs of care, decrease medical morbidity associated with opioid dependence, increase substance abuse treatment capacity, and improve treatment outcomes of this patient population1,25-29. Yet, only 17% of those requiring treatment for any type of substance abuse receive it, and buprenorphine remains an example of an evidenced-based, but underutilized, treatment7,30-32.
Legislative changes have increased the number of individuals receiving substance abuse treatment, yet access to BMT, especially in rural areas, remains limited3,7,30-34. Family medicine is the specialty with the largest number of physicians involved in patient care, and is an important factor in the future success of the legislative campaign to increasing access to substance abuse treatment such as BMT35. Legal restrictions, geographic obstacles, and patient attributes have all been examined to better understand patterns of buprenorphine adoption, but the family physician perspective is not well-understood30,31,36-38. In other physician populations, access to counseling, a lack of time, and perceived knowledge deficits have been associated with decreased willingness to provide BMT39-44. In this article, prior research is expanded upon to quantify and further explore barriers and facilitators to buprenorphine adoption among family physicians in New Hampshire and Vermont, two largely rural states in New England.
Survey development
The questionnaire was designed adhering to groundwork and principles of rigorous survey design45. Survey development consisted of three stages. First, the literature on barriers to prescribing buprenorphine among physicians was reviewed34,40,46. Second, based on this literature, a preliminary version of the survey was designed. This version of the survey was shared with three experienced individuals in the content area for feedback. An updated version was shared with the same experts, who established expert validity of the survey instrument. Third, the survey was piloted to establish construct validity (n=13).
Domains assessed by the survey included factors influencing buprenorphine adoption and opinions of opioids, addiction, and treatment options. Specific factors assessed included physician factors, physicians' understanding of patient factors, and logistical issues. Respondents were asked to rate their opinion of 20 statements using a five-point Likert scale. Three open-ended questions allowed respondents to share additional information. Limited demographic information was collected.
Study design
Both quantitative and qualitative survey questions were utilized. Data collection consisted of anonymous surveys. This decision was based on feedback from the pilot that indicated physicians were reluctant to acknowledge a lack of familiarity with opioid addiction or state negative opinions about addiction to researchers. It was determined that physicians would be more forthcoming via an anonymous survey as compared to more traditional qualitative methods such as interviews or focus groups. This approach has been successful in mixed-methods analysis of other value-laden topics47,48. The survey included open-ended questions, such as, 'What are the major barriers to providing buprenorphine at your clinic?' Space for additional comments was provided.
Data collection
This survey was distributed via the New Hampshire and Vermont chapters of the American Academy of Family Physicians (AAFP). As each state chapter of the AAFP maintains confidential membership lists, it was not possible to determine how many family physicians received the survey invitation. The estimate of the clinically active family physicians for both states is 714 (430 in New Hampshire; 284 in Vermont)49-51, although only those who were active in the online state chapters of the AAFP received the survey invitation. The decision to distribute the survey entirely online was consistent with the anonymous survey design, which was based on feedback from the pilot.
Survey distribution was web-based and practicalities of using Survey Monkey® were tested prior to administration. Once a state chapter agreed to participate, the state representative emailed a unique website address to their members. The survey link was active for 2 months and a reminder was sent 4-6 weeks after the initial email. Individuals receiving the survey were allowed to complete the survey once. Participants were eligible to complete the survey if they were a family physician in either Vermont or New Hampshire. Eligibility was determined by a series of demographic questions. Data collection occurred between September 2012 and March 2013.
Data analysis
Survey responses were analyzed using STATA v12.1 (StataCorp; http://www.stata.com) and ATLAS.ti v7.0 (http://atlasti.com). Descriptive statistics summarized respondent's quantitative survey responses, collapsing the five-point Likert scale into dichotomous outcomes. Comparisons between respondents who identified as buprenorphine prescribers and those who did not were made using the χ2 statistical test. Barriers/facilitators were correlated using a Pearson's r statistical test. Respondents also had the opportunity to share their thoughts in open-ended questions. These written comments were analyzed using an inductive analysis technique. Based on the patterns and categories of written comments, a coding scheme was developed and applied to the transcript of all written comments by one author (SR). The coded transcripts were then reviewed, developing a consensus by the authors (JD, SR) on the major ideas expressed in this section.
Ethics approval
This study was granted exemption status by the Dartmouth-Hitchcock Medical Center's Committee for the Protection of Human Subjects.
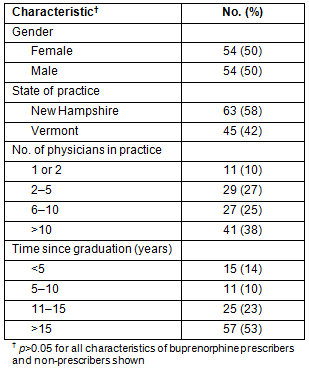
A total of 108 completed surveys were received from family physicians, with greater than 75% responding to at least one open-ended question. Gender of respondents was evenly distributed, and 10% were buprenorphine prescribers. Respondents in New Hampshire did not differ significantly from respondents in Vermont in demographics. Participant characteristics are displayed in Table 1.
Table 1: Demographics of all survey participants (prescribers and non-prescribers)
Perception of addiction and addiction treatment
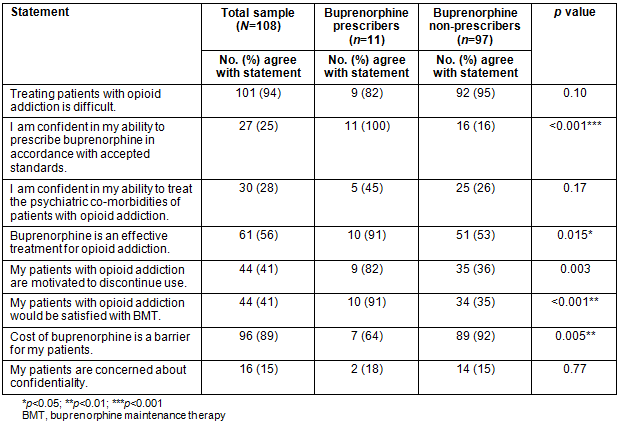
Greater than 80% of family physicians reported they regularly saw patients addicted to opioids. The vast majority (94%) stated that treating this patient population was difficult. Nearly three-quarters (73%) reported that they felt a personal responsibility, as family physicians, to treat opioid addiction. Of family physicians not prescribing buprenorphine, 10% were interested in becoming a BMT prescriber. Nearly one-third of non-prescribers reported they would be more likely to prescribe if they could have phone access to an addiction expert. This correlated with interest in becoming a BMT prescriber (r=0.60, p<0.001).
Confidence in personal ability to treat addiction
Regarding self-perception of ability to treat patients with opioid addiction, a quarter of respondents endorsed confidence in their ability to prescribe buprenorphine in accordance with accepted standards (Table 2). Slightly more respondents (28%) expressed confidence in their ability to treat psychiatric comorbidities. Current buprenorphine prescribers did not have more confidence in their ability to treat psychiatric comorbidities than did non-prescribers (p=0.17).
Perception of patient barriers
A minority (41%) felt their patients with opioid addiction were motivated to discontinue illegal use and 41% agreed that these patients would be satisfied with BMT. Prescribers were more likely than non-prescribers to report perceived patient satisfaction with BMT (91% vs 35%, p<0.001). Few non-prescribers felt BMT was affordable for patients, and non-prescribers were significantly more likely than prescribers to feel that cost was a barrier (92% vs 64%%, p=0.005).
Table 2: Physician perception of personal and patient barriers to buprenorphine adoption
Perception of logistic barriers
Among non-prescribers, perceived logistic barriers to buprenorphine adoption included inadequately trained staff (88%), insufficient time (80%), inadequate office space (49%), and cumbersome regulations (37%).
Remuneration
Approximately half (52%) of family physicians felt that there should be special remuneration for prescribing buprenorphine. Many of these respondents wrote that they were 'not sure' what an appropriate reimbursement should be, but felt that a fee should 'cover the cost of office procedures and staff training' or be used specifically to 'pay for counseling time' or 'pay nurses to do urines, pill counts, etc.' Others stated a specific amount of money, ranging from $100 to $500 per visit, or a set monthly amount per patient.
Qualitative responses
Survey respondents were invited to share their comments in three open-ended questions. Over 75% of participants responded to at least one of the open-ended questions. The comments were designed to elicit what participants perceived as barriers to buprenorphine adoption by them personally, in their clinic or community. The most commonly occurring themes in the comments included lack of knowledge, time, or interest; mistrust; and difficulty treating patients with addiction.
Lack of knowledge, time, or interest: Comments frequently referred to a lack of knowledge, time and/or interest. Lack of knowledge referred both to family physicians ('[I] lack understanding of needs of patients with addiction') and their staff ('lack of staff knowledge about opioid addiction'). A lack of time suggested that family physicians felt their practices were already overburdened ('Practice is already full with waiting list for primary care') and a lack of time in daily schedule ('It takes a lot'). In fact, 'time' was the most frequently reported barrier in the comments section of the survey, cited by 25 of the 108 survey participants. The participants expressing concern about 'time' were much more likely to support additional remuneration for BMT (72% vs 46%, p=0.02). Lack of interest was also a significant barrier to the provision of buprenorphine. Some family physicians (19%) stated they simply don't want to provide buprenorphine ('I do not wish to prescribe it [buprenorphine]'; 'not a provider by choice'.)
Mistrust: Written comments conveyed a high level of mistrust, both of patients with addiction and of buprenorphine as a treatment. Concerning patients with addiction, family physicians indicated concern working with people who had a history of addiction. For example, one respondent stated he or she did not want to 'deal with addicts who lie - sell half of what they get from the pharmacy' and another stated that their office was 'not set up to function as a police agency'. One physician stated that a major barrier to prescribing buprenorphine for opioid replacement therapy was their own 'very negative attitude about treating a messy problem in a messy population'. Several comments indicated that family physicians would prefer if patients went elsewhere for addiction services ('I want these patients to go to a special center for this therapy rather than my clinic').
Mistrust concerning the diversion of buprenorphine was also expressed ('[a] significant amount of buprenorphine ... ends up in the community and schools'). Family physicians stated that they did not want to be known as a buprenorphine prescriber ('I don't want to become a local source for this'). Other comments indicated a lack of confidence in the philosophical basis of opioid maintenance therapy. There was a general sentiment that patients were substituting addictions ('trading one addiction for another') and skepticism that buprenorphine is an effective treatment long-term ('works long term').
Difficulty treating patients with addiction: Family physicians frequently cited the difficulty of treating patients with addiction issues, stating patients with addiction are a 'difficult population to manage'. Patients with drug addiction were referred to as 'high-maintenance', 'stressful', and 'challenging'. The difficulty of caring for patients with addiction was compounded by a perceived 'lack of easy access to high-quality, well-coordinated psychiatric care for comorbidities'. Family physicians stated that in order for them to consider treating patients with buprenorphine, they would 'need easier access to pain management and ... psychiatric care, as most of these patients seem to have comorbid depression, anxiety, bipolar, or personality traits/disorders'.
Discussion
This study is the largest survey of family physicians about buprenorphine known to the authors, including more than 100 respondents, with 10% reporting first-hand experience with BMT. It investigates family physicians' attitudes towards BMT with the goal of quantifying perceived barriers and facilitators to providing treatment. The results suggest family physicians are excellent candidates to provide BMT for two key reasons: one, they report regularly seeing opioid-addicted patients, and two, most believe that treating opioid addiction is their responsibility.
The most commonly cited barrier to providing BMT was a lack of staff preparedness. While many family physicians were concerned about their personal ability to meet the needs of opioid addicted patients - expressing concerns about following regulations or treating psychiatric comorbidities - they were far more concerned about their staff's ability to do so. Training modules directed at alleviating this concern are freely available through a joint effort by the National Institute on Drug Abuse and Substance Abuse and Mental Health Services Administration52. Their buprenorphine awareness training products are specifically targeted at non-physicians, which may help physicians feel their clinical teams are prepared. Physicians themselves have access to free and low-cost online buprenorphine licensing courses, which also provide continuing medical education credits. Approximately 10% of family physicians reported an interest in prescribing buprenorphine, and stated that having phone access to an addiction expert would increase their willingness to prescribe. This intervention currently exists, as the Physician Clinical Support System-Buprenorphine (PCSS-B), a federally funded national mentorship network which includes phone support53. Increasing awareness of these free training opportunities and PCSS-B among the family physician community could increase the number of providers prescribing buprenorphine.
After staff preparedness, the main concern of non-prescribers was about having sufficient time to prescribe buprenorphine. If prescribing BMT was compensated at a higher rate, it is likely that family physicians would be able to allocate more time for these patients. This is supported by the fact that those citing 'time' concerns in their comments were most likely to feel additional remuneration was appropriate. This would be commensurate with the fact that prescribing buprenorphine requires additional training and a special license. Additional survey results on cost and reimbursement suggest that family physicians believe patients cannot afford treatment. However, a survey of current buprenorphine prescribers in rural Washington State found that over 75% of patients receiving buprenorphine were insured. Furthermore, these family physicians indicated that patients 'always pay their bills' as 'it is worth it to them34.' Given the demand for buprenorphine, this may indicate an opportunity for family physicians to consider direct cash payments for providing BMT that would make it 'worth' their time. As a comparison, the current average charge for BMT in the USA is $250-450 for an initial visit, with an additional monthly charge of $150-250 for follow-up medical care54. This may differ depending on the practice and region, but education about possible compensation models for BMT may help family physicians see this as a viable option for their practice.
Another challenge remains in the perceived efficacy of buprenorphine: family physicians with first-hand experience prescribing buprenorphine had a much more optimistic view of its efficacy than did family physicians with no BMT experience. Furthermore, many used the open-ended questions to express concern that BMT was simply 'substituting' addictions. This commonly expressed view is outside the realm of evidence-based medicine, as research has shown that few individuals with opioid addiction successfully abstain from use without opioid replacement therapy11. A Cochrane review in 2008 found that medium and high doses of buprenorphine not only increased retention in treatment, but also suppressed heroin use significantly compared to placebo13. In contrast, behavioral interventions to treat opioid abuse are inadequate, as more than 80% of patients resume drug use within 2 years of intensive residential treatment55. Regarding concerns of diversion of buprenorphine, lessons can be learned from countries with successful buprenorphine use. For example, in France, nearly 20 years of unregulated buprenorphine prescriptions have resulted in increased treatment for opioid addiction, decreased mortality from opioid-related overdoses, few buprenorphine-related medical complications, and low diversion56.
Other relevant findings cast doubt on perceived barriers reported in the literature. Specifically, all family physicians sampled agreed that treating patients with addiction was challenging. Buprenorphine prescribers and non-prescribers had equal confidence in their ability to treat patients' psychiatric comorbidities. In addition, cost was seen as less of a barrier by current buprenorphine prescribers than non-prescribers. This may indicate these issues may not be true barriers to buprenorphine adoption.
There are a number of potential limitations to the study at hand. The study sample size is relatively small and consists of family physicians in New Hampshire and Vermont, which may not be generalizable to all family physicians. Other concerns include the fact that non-members of AAFP may have been inadvertently excluded and this survey was distributed online, which requires participants to have access to email and internet. However, a survey of family physicians from 10 years ago found that approximately 95% had internet access at that time57,58. Additionally, while it is not possible to calculate an accurate response rate, the demographics of survey respondents are similar to the demographics of the overall AAFP membership57,58. That key questions (for example about the effectiveness of buprenorphine, importance of offering buprenorphine treatment, and interest in becoming a prescriber) consisted mainly of responses between 2 and 4 on a scale of 1 to 5 (with few extreme responses of 1 or 5) suggests that bias is contained. Additionally, the authors' decision to identify current buprenorphine providers and analyze their responses independently minimizes the positive bias associated with buprenorphine. Lastly, only family physicians were surveyed, so physician perception of patient barriers (such as cost) may not reflect patient reality.
Despite the limitations, this study presents rich findings on barriers to buprenorphine adoption among family physicians, integrating quantitative and qualitative information. Results suggest BMT remains an opportunity for family physicians to make a significant contribution to the public health of rural communities. However, adoption of buprenorphine faces resistance due to a multitude of factors explored in this study. The level of physician knowledge and trust of BMT remains low, although frustration regarding patients with opioid addiction may indicate an increased readiness to adopt new practices such as BMT. Addressing specific concerns about buprenorphine effectiveness and relative safety, as well as combating the stigma surrounding addiction and patients with mental illness, may help increase the availability of BMT in rural areas. Organizations should seek to support adoption of BMT by family physicians by increasing access to addiction experts, providing staff training, and educating family physicians about possible compensation models.
Acknowledgements
The authors would like to thank the participants who took this survey. They also thank those who helped with this project at different stages of conception, including Matthew Duncan, Steven Elgert, Cathleen Morrow, Anthony Valdini, Catrina Watson, Donald West, and the staff and providers at Dartmouth-Hitchcock Family Medicine Clinic and Dartmouth Psychiatric Research Center for their support.
Stephanie A. Rolin received financial support from the Tenney Research Fellowship at the Geisel School of Medicine at Dartmouth.
References
1. Finkelstein R, Netherland J, Sylla L, Gourevitch MN, Cajina A, Cheever L. Policy implications of integrating buprenorphine/naloxone treatment and HIV care. Journal of Acquired Immune Deficiency Syndromes 2011; 56(Suppl 1): S98-S104.
2. Mojtabai R, Chen LY, Kaufmann CN, Crum RM. Comparing barriers to mental health treatment and substance use disorder treatment among individuals with comorbid major depression and substance use disorders. Journal of Substance Abuse Treatment 2014; 46(2): 268-273.
3. Wen H, Cummings JR, Hockenberry JM, Gaydos LM, Druss BG. State parity laws and access to treatment for substance use disorder in the United States: implications for federal parity legislation. JAMA Psychiatry 2013; 70(12): 1355-1362.
4. United States Department of Labor. The Mental Health Parity and Addiction Equity Act of 2008 (MHPAEA). Washington, DC: United States Department of Labor, 2010.
5. Manchikanti L, Fellows B, Ailinani H, Pampati V. Therapeutic use, abuse, and nonmedical use of opioids: a ten-year perspective. Pain Physician 2010; 13(5): 401-435.
6. Warner M, Chen LH, Makuc DM, Anderson RN, Minino AM. Drug poisoning deaths in the United States, 1980-2008. Hyatsville, MD: National Center for Health Statistics, Centers for Disease Control and Prevention, 2011; 1-7.
7. Substance Abuse and Mental Health Services Administration. Results from the 2010 National Survey on Drug Use and Health: national findings. Rockville, MD: US Department of Health and Human Services, 2011.
8. Centers for Disease Control and Prevention. Vital signs: overdoses of prescription opioid pain relievers - United States, 1999-2008. Atlanta, GA: Centers for Disease Control and Prevention, 2011.
9. Federation of State Medical Boards. Model policy on DATA 2000 and treatment of opioid addiction in the medical office. Washington, DC: Federation of State Medical Boards, 2013.
10. Cowan A. Buprenorphine: the basic pharmacology revisited. Journal of Addiction Medicine. 2007; 1(2): 68-72.
11. Kakko J, Svanborg KD, Kreek MJ, Heilig M. 1-year retention and social function after buprenorphine-assisted relapse prevention treatment for heroin dependence in Sweden: a randomised, placebo-controlled trial. Lancet 2003; 361(9358): 662-668.
12. Ling W, Charuvastra C, Collins JF, Batki S, Brown LS, Jr, Kintaudi P, et al. Buprenorphine maintenance treatment of opiate dependence: a multicenter, randomized clinical trial. Addiction 1998; 93(4): 475-486.
13. Mattick RP, Kimber J, Breen C, Davoli M. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database of Systematic Reviews (Online) 2008, 2008(2):CD002207.
14. O'Connor PG, Oliveto AH, Shi JM, Triffleman EG, Carroll KM, Kosten TR, et al. A randomized trial of buprenorphine maintenance for heroin dependence in a primary care clinic for substance users versus a methadone clinic. American Journal of Medicine 1998; 105(2): 100-105.
15. Woody GE, Poole SA, Subramaniam G, Dugosh K, Bogenschutz M, Abbott P, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA 2008; 300(17): 2003-2011.
16. Sells SB, Simpson DD. Effectiveness of Treatment for drug abuse - evidence from the Darp Research Program in the United-States. Bulletin on Narcotics 1979; 31(1): 1-11.
17. Abbott PJ, Moore BA, Weller SB, Delaney HD. AIDS risk behavior in opioid dependent patients treated with community reinforcement approach and relationships with psychiatric disorders. Journal of addictive diseases. 1998; 17(4): 33-48.
18. Camacho LM, Bartholomew NG, Joe GW, Cloud MA, Simpson DD. Gender, cocaine and during-treatment HIV risk reduction among injection opioid users in methadone maintenance. Drug and Alcohol Dependence 1996; 41(1): 1-7.
19. Magura S, Rosenblum A, Rodriguez EM. Changes in HIV risk behaviors among cocaine-using methadone patients. Journal of Addictive Diseases 1998; 17(4): 71-90.
20. Novick DM, Joseph H, Croxson TS, Salsitz EA, Wang G, Richman BL, et al. Absence of antibody to human immunodeficiency virus in long-term, socially rehabilitated methadone maintenance patients. Archives of Internal Medicine 1990; 150(1): 97-99.
21. Grella CE, Anglin D, Annon JJ. HIV risk behaviors among women in methadone maintenance treatment. Substance Use & Misuse. 1996; 31(3): 277-301.
22. Strathdee SA, Galai N, Safaiean M, Celentano DD, Vlahov D, Johnson L, et al. Sex differences in risk factors for HIV seroconversion among injection drug users: a 10-year perspective. Archives of Internal Medicine 2001; 161(10): 1281-1288.
23. Arfken CL, Johanson CE, di Menza S, Schuster CR. Expanding treatment capacity for opioid dependence with office-based treatment with buprenorphine: National surveys of physicians. Journal of substance abuse treatment. 2010;39(2):96-104. Epub 2010/07/06.
24. Mintzer IL, Eisenberg M, Terra M, MacVane C, Himmelstein DU, Woolhandler S. Treating opioid addiction with buprenorphine-naloxone in community-based primary care settings. Annals of Family Medicine. 2007; 5(2): 146-150.
25. Fiellin DA, Rosenheck RA, Kosten TR. Office-based treatment for opioid dependence: reaching new patient populations. American Journal of Psychiatry 2001; 158(8): 1200-1204.
26. Bates M, Pemberton DA. The effect of methadone prescribing in a clinic setting on the criminal activity of drug users. Scottish Medical Journal 1996; 41(6): 173-175.
27. Merrill J, Alterman A, Cacciola J, Rutherford M. Prior treatment history and its impact on criminal recidivism. Journal of Substance Abuse Treatment 1999; 17(4): 313-319.
28. Weinrich M, Stuart M. Provision of methadone treatment in primary care medical practices: review of the Scottish experience and implications for US policy. JAMA 2000; 283(10): 1343-1348.
29. Gossop M, Marsden J, Stewart D, Lehmann P, Strang J. Methadone treatment practices and outcome for opiate addicts treated in drug clinics and in general practice: results from the National Treatment Outcome Research Study. British Journal of General Practice 1999; 49(438): 31-34.
30. Knudsen HK, Ducharme LJ, Roman PM. Early adoption of buprenorphine in substance abuse treatment centers: data from the private and public sectors. Journal of Substance Abuse Treatment 2006; 30(4): 363-373.
31. Knudsen HK, Ducharme LJ, Roman PM. The adoption of medications in substance abuse treatment: associations with organizational characteristics and technology clusters. Drug and Alcohol Dependence 2007; 87(2-3): 164-174.
32. Gordon AJ, Liberto J, Granda S, Salmon-Cox S, Andree T, McNicholas L. Outcomes of DATA 2000 certification trainings for the provision of buprenorphine treatment in the Veterans Health Administration. American Journal on Addictions/American Academy of Psychiatrists in Alcoholism and Addictions 2008; 17(6): 459-462.
33. Stein BD, Gordon AJ, Sorbero M, Dick AW, Schuster J, Farmer C. The impact of buprenorphine on treatment of opioid dependence in a Medicaid population: recent service utilization trends in the use of buprenorphine and methadone. Drug and Alcohol Dependence 2012; 123(1-3): 72-78.
34. Quest TL, Merrill JO, Roll J, Saxon AJ, Rosenblatt RA. Buprenorphine therapy for opioid addiction in rural Washington: the experience of the early adopters. Journal of Opioid Management 2012; 8(1): 29-38.
35. Association of American Medical Colleges. 2012 Physician specialty data book. Washington, DC: Association of American Medical Colleges, 2012.
36. Cicero TJ, Surratt H, Inciardi JA, Munoz A. Relationship between therapeutic use and abuse of opioid analgesics in rural, suburban, and urban locations in the United States. Pharmacoepidemiology and Drug Safety 2007; 16(8): 827-840.
37. Ducharme LJ, Knudsen HK, Roman PM, Johnson JA. Innovation adoption in substance abuse treatment: exposure, trialability, and the Clinical Trials Network. Journal of Substance Abuse Treatment 2007; 32(4): 321-329.
38. Knudsen HK, Abraham AJ, Roman PM. Adoption and implementation of medications in addiction treatment programs. Journal of Addiction Medicine 2011; 5(1): 21-27.
39. Cunningham CO, Kunins HV, Roose RJ, Elam RT, Sohler NL. Barriers to obtaining waivers to prescribe buprenorphine for opioid addiction treatment among HIV physicians. Journal of General Internal Medicine 2007; 22(9): 1325-1329.
40. Cunningham CO, Sohler NL, McCoy K, Kunins HV. Attending physicians' and residents' attitudes and beliefs about prescribing buprenorphine at an urban teaching hospital. Family Medicine 2006; 38(5): 336-340.
41. Netherland J, Botsko M, Egan JE, Saxon AJ, Cunningham CO, Finkelstein R, et al. Factors affecting willingness to provide buprenorphine treatment. Journal of Substance Abuse Treatment 2009; 36(3): 244-251.
42. Turner BJ, Laine C, Lin YT, Lynch K. Barriers and facilitators to primary care or human immunodeficiency virus clinics providing methadone or buprenorphine for the management of opioid dependence. Archives of Internal Medicine 2005; 165(15): 1769-1776.
43. Walley AY, Alperen JK, Cheng DM, Botticelli M, Castro-Donlan C, Samet JH, et al. Office-based management of opioid dependence with buprenorphine: clinical practices and barriers. Journal of General Internal Medicine 2008; 23(9): 1393-1398.
44. West JC, Kosten TR, Wilk J, Svikis D, Triffleman E, Rae DS, et al. Challenges in increasing access to buprenorphine treatment for opiate addiction. American Journal on Addictions/American Academy of Psychiatrists in Alcoholism and Addictions 2004; 13(Suppl 1): S8-16.
45. Aday LA, Cornelius LJ. Designing and conducting health surveys: a comprehensive guide. San Francisco: Jossey-Bass, 2006.
46. Barry DT, Irwin KS, Jones ES, Becker WC, Tetrault JM, Sullivan LE, et al. Integrating buprenorphine treatment into office-based practice: a qualitative study. Journal of General Internal Medicine 2009; 24(2): 218-225.
47. Van Vorst RF, Araya-Guerra R, Felzien M, Fernald D, Elder N, Duclos C, et al. Rural community members' perceptions of harms from medical mistakes: A High Plains Research Network (HPRN) study. Journal of the American Board of Family Medicine 2007; 20: 135-143.
48. Wiese CH, Bartels UE, Ruppert DB, Graf BM, Hanekop GG. Prehospital emergency physicians' experiences with advance directives in Germany: a questionnaire-based multicenter study. Minerva Anestesiologica 2011; 77(2): 172-179.
49. Association of American Medical Colleges. State physician workforce data book. (Online) 2011. Available: https://www.aamc.org/data/workforce/reports (Accessed 2 April 2013).
50. Vermont Department of Health. 2010 physician survey statistical report. (Online) 2011. Available: http://healthvermont.gov/research/HlthCarePrvSrvys/documents/phys10bk.PDF (Accessed 2 April 2013).
51. Watson C, Executive Director of New Hampshire Academy of Family Physicians; personal communication with Kazal LA, 2 February 2013.
52. Addiction Technology Transfer Center Network. Buprenorphine treatment: training for multidisciplinary addiction professionals. Kansas City, MO: Addiction Technology Transfer Center Network, 2005.
53. Egan JE, Casadonte P, Gartenmann T, Martin J, McCance-Katz EF, Netherland J, et al. The Physician Clinical Support System-Buprenorphine (PCSS-B): a novel project to expand/improve buprenorphine treatment. Journal of General Internal Medicine 2010; 25(9): 936-941.
54. National Alliance of Advocates for Buprenorphine Treatment. Patient-matching system frequently asked questions (Online) 2013. Available: http://www.treatmentmatch.org/patients/patient_faq.cfm (Accessed 30 March 2013).
55. Bart G. Maintenance medication for opiate addiction: the foundation of recovery. Journal of Addictive Diseases 2012; 31(3): 207-225.
56. Fatseas M, Auriacombe M. Why buprenorphine is so successful in treating opiate addiction in France. Current Psychiatry Reports 2007; 9(5): 358-364.
57. American Academy of Family Physicians. Practice Management Profile: a survey of members of the American Academy of Family Physicians. (Online) 2003. Available: http://www.aaafp.org/fpm (Accessed 10 April 2013).
58. White B. The state of family medicine: family practice management. (Online) 2012. Available: http://www.aaafp.org/fpm (Accessed 10 April 2013).
