The healthcare implications of a population aging with multiple chronic illnesses1 has placed palliative care high on the research agenda2. In Canada, a number of recent policy documents have provided roadmaps for developing palliative care. The goal is to provide high quality care closer to home, while reducing unnecessary hospitalization and emergency room visits in the final weeks and months of life3-5. Achieving this goal requires better integration of healthcare services, delivery models that bridge the gap between chronic illness management and palliative care; a person-centered approach that includes advance care planning and quality of life assessment6, family caregiver (FCG) support so that persons can be cared for at home in a way that does not compromise the health and wellbeing of FCGs, and community capacity-building strategies that include recognition of the importance of the volunteer network to high quality care3,4,7,8.
Realizing this goal for rural populations, who are older and aging proportionately faster9 with a higher burden of chronic illness10 than their urban counterparts, is challenging. These challenges vary depending on geographic location. In Canada, palliative care in rural communities is complicated by limited healthcare services, poor access to specialized palliative services and expertise, healthcare provider shortages, harsh weather conditions that make travel challenging, and geographic dispersion of the population4,11,12. Within the province of British Columbia, where this research project is being implemented, it has been estimated that over 600 000 residents live beyond a 60-minute drive to specialized palliative services and that services deteriorate as one moves farther from the south-west urban core of the province13. This means that rural older persons often endure complex and challenging transitions near end of life14. Rural citizens experience more transitions in care settings and undergo more procedures in the last year of life than their urban counterparts15. In Canada, a higher percentage of rural cancer patients are admitted to acute care multiple times during their last 28 days of life (30% rural versus 20% urban)16. This is particularly important in light of a recent Canadian survey suggesting that the rural population has the highest percentage of persons who prefer to die at home17. Rural FCGs have difficulty getting the information and tangible support they need18,19 and report greater use of family physicians, the emergency room and pharmacists than their urban counterparts20. Although there are significant healthcare gaps in rural palliative care, studies have illustrated how healthcare providers and the community seek to overcome those gaps through shared values and flexible responsibilities21-23.
There are important principles to consider regarding the delivery of rural palliative care. A comparative case study conducted in one province in Canada suggested that successful delivery of rural palliative care has common characteristics: an advanced practice nurse; 24-hour access to palliative expertise; clearly designated roles and accountabilities; and palliative-specific planning, coordination, and documentation24. The approach should be driven by local leadership in consultation with local stakeholders and should build upon and develop currently existing capacity25. Being cared for in their home community by those whom they know is important for rural citizens21,26, particularly in light of the relationship that exists between rural citizens' sense of belonging and health27,28. Building upon these principles, a community capacity-building model of rural palliative care has been constructed29,30, a toolkit has been created to translate the knowledge of how to use this model31, and a case study illustrating the model has been published32. Further, spatial models have been constructed of the relevant factors to consider when situating palliative services in rural areas33-35.
The purpose of this article is to describe the feasibility of implementing a rural palliative supportive service (RPaSS) using a community-based research approach. RPaSS differs from typical rural palliative care delivery in four ways: (1) the palliative expertise resides within the rural community; (2) individuals are identified early in the palliative trajectory thus facilitating a seamless transition between chronic disease management care and palliative care; (3) a nurse coordinator visits regularly in the home with a clinical team to provide backup; and (4) the service is guided by a community-based advisory committee of relevant stakeholders.
Purpose
The purpose of this community-based research project is to test the feasibility and identify potential outcomes of implementing an RPaSS for older adults living with life-limiting chronic illness and their family caregiver in the community. This article reports on the feasibility aspects of the study.
Definitions
In Canada, a rural community is outside the commuting zone of larger urban centers with populations of 10 000 or more36. Palliative care is supportive care provided through the primary care network and specialized services that begins with the identification of chronic life-limiting illness and continues through into the bereavement period3. A chronic life-limiting illness is any chronic illness (eg solid organ failure, cancer, neurodegenerative diseases) in which death could reasonably be expected to occur within the foreseeable future3.
Study period
RPaSS is a 3-year pilot study (2012-2014). The first year (2012) was dedicated to service planning. Recruitment began in January 2013.
Design
The research approach for this study is community-based research, an approach whereby community members are involved with identifying the problem and research questions and providing input into the applicability of the proposed research methods37,38.
Setting
The context for RPaSS is two co-located rural communities, each with a population of approximately 10 000 persons. These communities are located 30 minutes from one another and 4 hours by car from a specialist palliative treatment centre, a commute that entails navigating three mountain passes that often have difficult driving conditions due to weather. The communities were chosen because of their inherent capacity and unique needs. The capacity resides in a number of community-based palliative champions who have a high degree of expertise. The unique needs are related to the closure of the local hospital in one community, so those residents have to commute to the other community for many of their healthcare services. Palliative patients who require acute intervention must leave their local community and healthcare providers for care. The mandate of RPaSS is to serve both communities, with the intent of supporting residents to remain in their home community for care. One of the challenges of maintaining dedicated palliative services in rural communities is a fluctuating census that makes it difficult to justify the expenditures. The delivery of services across two communities has the added benefit of producing adequate census for the service.
Participants
Recruitment occurs through healthcare providers and by self-referral for those living with life-limiting chronic illness. To be eligible for the service, participants must have a chronic life-limiting illness and be living within a 1-hour commute by car from the communities under study. Exclusion criteria include those who are non-English speaking and those with dementia. The nature of the care provided by the nurse coordinator and the use of self-reported outcome measures require non-compromised cognitive function and English language skills; translation services are not readily available in the community. As these communities are largely English speaking this criterion does not exclude many individuals. FCGs also participate in the research, but their participation is not required. FCGs are eligible if they are the primary caregiver of the participant and if they speak English and do not have cognitive impairment.
A number of strategies are used to determine whether participants have a chronic life-limiting illness. The iPal tool, developed by Providence Health (see http://ipalapp.com), is provided in a pocket-sized laminated form to physicians to facilitate their ability to identify appropriate candidates for the study. For other healthcare providers who may not have access to the diagnostic information required of the iPal, the surprise question39 is used as an indicator that individuals might be eligible for the service. The question is, 'Would you be surprised to see this individual die within the next year?' If the answer is 'no', then they are eligible for study screening. Participants who self-refer are shown a model of a typical chronic illness trajectory40 and are asked to indicate where they see themselves on that trajectory. If they indicate being in a phase of increasing illness burden, they are considered eligible. If after the initial screening the nurse coordinator is still unclear about eligibility, participants are asked to sign a pre-consent to allow him or her to consult with their family physician regarding their chronic illness status.
Intervention
This service is based within the rural community (eg no link to urban specialist teams) and is structured around a nurse coordinator who visits persons living with chronic life-limiting illness in the home bi-weekly. The focus of these interventions includes symptom management, teaching, referrals, psychosocial and spiritual support, advance care planning, mobilizing community support for practical tasks, and telephone-based support for individuals who must commute outside of the rural community for care. The nurse coordinator role can be likened to that of the nurse navigator role described in the cancer management literature whereby the nurse facilitates continuity of care, promotes a patient- and family-centered approach, and helps individuals navigate the resources available through healthcare and the community41,42. The role differs from that performed by community health nurses or case managers in terms of intensity of care (eg caseload), continuity of care throughout all transitions in sites of care (including commuting outside of the community),and the emphasis on assisting participants to navigate both healthcare and informal community-based resources (eg mobilizing support from neighbors). The nurse coordinator is supported by a community-based clinical team that includes a general practitioner and nurse practitioner who have additional palliative training. However, registrants on the service remain under the care of their family physician; RPaSS is designed to augment the primary care network. A quarterly fax is sent to the participant's primary care physician outlining the nature of the care provided by RPaSS to ensure continuity of care. RPaSS has a toll-free number that is monitored by the nurse coordinator and nurse practitioner in case additional support is required between visits. Participants are followed until death. FCGs receive three bereavement visits: immediately after death and at 1 and 3 months.
Data collection
Mixed-method data collection strategies are used. During the home visits the nurse coordinator conducts a functional performance assessment using the Palliative Performance Scale (PPS)43,44, and administers paper-based versions of the McGill Quality of Life Questionnaire45,46 to the participant and the Caregiver Support Needs Survey to the FCG. The Caregiver Support Needs Survey was adapted by Dumont et al47 from the Home Caregiver Need Survey originally developed by Hileman, Lackey and Hassanein48. The PPS scores individuals between 0 and 100 based upon ambulation, activity and evidence of disease, self-care, intake and level of consciousness. The McGill Quality of Life Questionnaire requires the participant to rate overall perceived quality of life on a scale of 0-10; identify three of the most troublesome symptoms and rate them on a 10-point scale; and rate various physical, psychosocial and existential dimensions on a scale of 1-10. A qualitative question enquires about the factors that have the greatest effect on quality of life. The Caregiver Support Needs Survey asks participants to rate both the importance and the degree of met need on 25 common caregiving tasks.
The nurse coordinator also collects data on healthcare utilization since the previous visit (eg number of hospitalizations and emergency room visits, length of stay, physician visits, number and length of respite stays, length of time on palliative benefits, home support involvement, and other healthcare provider visits/usage). This has been collected through each participant's story of 'how things have gone' since the previous visit. Qualitative data around healthcare utilization is captured to facilitate an understanding of the appropriateness of the use. For example, preventable emergency room usage or hospitalization is evaluated. At the 1-month and 3-month bereavement visits the nurse coordinator administers the Texas Revised Inventory of Grief49, primarily as a means to assess and intervene around bereavement. In addition, the research assistant conducts a semi-structured interview with the FCG about satisfaction with RPaSS at approximately 2 months into the bereavement period.
The nurse coordinator keeps a journal about the process of implementing the service including who declines participation and why, challenges that arise with the use of outcome instruments, explanations of modifications to the visit protocol, engagements with the community around the service and other relevant observations. She also documents visit statistics that include the nature and length of the visit (eg telephone versus in person) and the interventions to address the identified needs.
Satisfaction with care, grief and quality of life instruments were evaluated for this rural population using cognitive interviewing prior to study implementation50. Individuals were asked to fill out the instruments and reflect on what they were thinking as they filled them out. During the cognitive interview participants recorded high satisfaction scores on instruments even as they told stories that suggested a strong degree of dissatisfaction. They were reluctant to complain 'in writing' because, as one interviewee put it, 'I might be needing care soon'. As a result of the cognitive interviewing the satisfaction-with-care measures were augmented with a telephone interview about satisfaction, conducted by the research assistant with the FCG after the death of the participant.
Ethics approval
This study underwent a harmonized ethical review process by the Behavioural Research Boards of the University of British Columbia and Interior Health (H12-00786). Confidentiality of participants is protected through assigning unique study identification numbers. Only the investigative and clinical team have access to the participant names. The community advisory committee is provided with aggregated and anonymized data only.
Results described here are from the first year where planning was conducted in consultation with the community advisory committee (2012) and the first 17 months of RPaSS implementation (January 2013 to May 2014). No bereavement data is provided due to the small amount of bereavement data collected during this period.
Using a community-based research approach
Figure 1 provides a timeline of the partnership between the investigative team and community-based champions, illustrating the length of time that can be required to know a community well and to build the credibility required for community-based research methods37. The idea of RPaSS was derived from findings of several studies that explored the state of palliative care in these communities through stakeholder interviews and participant observation18,21,23. In keeping with community-based research methods, the investigative team worked closely with champions from these communities to determine the needs and to develop and implement RPaSS as a response to those needs.
Change with community-based research is achieved largely through community capacity-building approaches such as engaging individuals, building networks, responding to perceived needs, and supporting local knowledge and skills51. As such, a first step in implementing this service was identifying members for an advisory committee and establishing terms of reference. The 21-member community-based advisory committee included members from stakeholder groups such as the health authority, hospice, local government, and residential care; from disciplinary groups such as physicians, nurses, social workers, pharmacists and chaplains; and from relevant interest groups such as First Nations and FCGs. The mandate of the advisory committee was to provide guidance for the service, to communicate information back to their stakeholders about the service, and to monitor the quality of care provided by the service.
The investigative team worked with the community advisory committee over a 1-year period (2012) to plan RPaSS. Several representatives from the committee worked closely with the investigative team and the committee as a whole met regularly to provide input into the planning. During this planning phase a logic model, assessment framework and communication plan were constructed to support RPaSS. Logic models make explicit the goals that a program seeks to achieve and so can be useful for planning and evaluating practice-based research initiatives52,53. The logic model located in Figure 2 describes the resources, activities, outcomes, and goals RPaSS seeks to achieve. At the bottom of the diagram are listed the foundational values and principles, derived from research, upon which RPaSS is designed. RPaSS has four primary goals: to enhance quality of life and quality of care for dying persons; to attenuate potential negative sequelae, such as depression and burden, for FCGs; to support appropriate use of the healthcare resources available in rural communities; and to promote a sense of community pride in the palliative care provided to citizens. These four primary goals are supported by a variety of short-term goals that are suitable for measurement.
An evidence-informed program assessment framework was constructed to evaluate RPaSS. Although there are many published frameworks to assess the quality of palliative care, none are suitable to the rural context. This context is important because of the differences between urban and rural contexts in available services. For example, many frameworks assume the existence of a multidisciplinary team dedicated to palliative care. Rural communities rarely have such teams available. Further, quality frameworks are usually designed to evaluate the overall delivery of healthcare services, while RPaSS was designed to augment what is available through the primary care network. As such, it was important to have evaluative strategies that would be sensitive to the unique contributions of RPaSS. This framework has been published elsewhere54.
A communication plan was designed to promote common messages about the service. Common messages are particularly important because of the unique nature of the service. Community members are not familiar with a service that comes into their home early in the disease trajectory. In these communities, home services are typically only provided when persons have task-related needs that require skilled nursing care. In addition to providing key messages about RPaSS, the communication plan provides details of the who, what, when and how of communication between stakeholders. For example, the nurse coordinator has a plan for communicating with the research participants, the research co-investigators, the primary healthcare providers, the community and the RPaSS clinical and advisory team. These are accompanied by standard flow sheets. An important part of this plan is a media strategy designed to keep the public informed about RPaSS and to recruit participants. Information is difficult to disseminate in rural communities where much information is passed through word of mouth. The challenge in the first-year planning phase was to educate the community in anticipation of the service while not losing community interest because participants were not yet being recruited. Strategies included stories in the media, talking to local community organizations, presenting at town councils, and visiting offices of healthcare providers. The nurse coordinator began this public information strategy about 4 months prior to service implementation. Despite a carefully crafted communication plan, the plan was labor-intensive and over time it has been difficult for the nurse coordinator to conduct the clinical visits and keep RPaSS visible in the communities.
Overall, this community-based research approach was successful in building networks, engaging local citizens, and obtaining essential advice for the implementation of RPaSS. The inclusion of a wide variety of stakeholders for the community advisory committee was a particularly effective strategy in solving challenges and disseminating information. Even though meetings typically included only half of the members, most members attended at various times. The detailed communication plan was less useful, although the construction of key messages was an essential part of the public information strategy.
Using this community-based approach also had challenges. Tensions between what the investigative team believed was the best science and what the advisory committee members believed was acceptable required negotiation. Examples included suitability of structured instruments, visit schedules, service capacity and the use of technology. The 1-year planning cycle was essential to ensuring that community members were sufficiently involved to provide input into RPaSS and its evaluation.
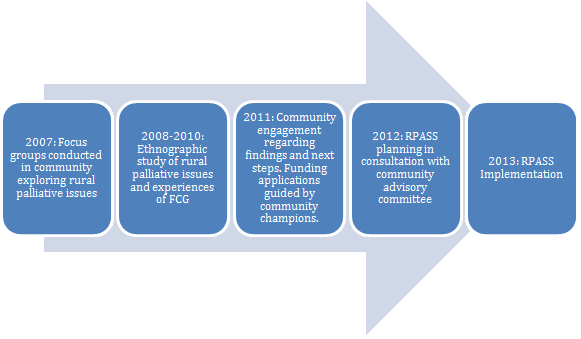 Figure 1: Timeline of community-based research approach.
Figure 1: Timeline of community-based research approach.
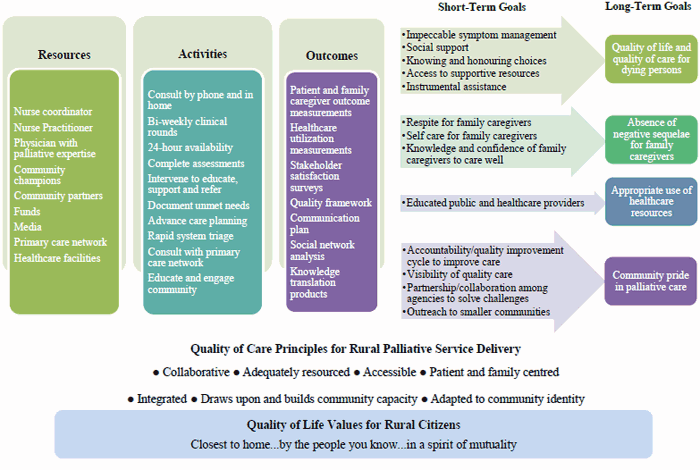 Figure 2: RPaSS resources, activities, outcomes, and goals.
Figure 2: RPaSS resources, activities, outcomes, and goals.
Recruitment patterns and challenges
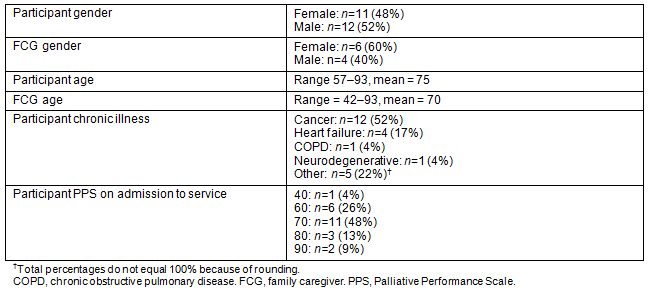
To date, 23 participants and 10 FCGs have been recruited to the service. Table 1 shows the demographic data for these participants. Recruitment increased steadily over the first months of the service, from two participants recruited in the first month to nine participants recruited in the eighth month at which point the nurse coordinator was unable to accommodate further participants. Recruitment was more challenging than originally anticipated. A number of factors may have influenced recruitment: the reluctance of individuals to identify themselves as having a life-limiting illness, the reluctance of physicians to refer to a project that is time-limited, a misunderstanding in the community that the service was already over-subscribed, and the challenges of making the service known in the community. To enhance recruitment efforts, the nurse coordinator visited physician offices, home care services, and hospital-based services to educate them about the service. Recruitment materials were placed on logo pens with flash drives so that healthcare providers had the necessary materials readily available. Flyers were placed at strategic locations throughout the community (eg pharmacies) and advertisements were run in local papers. The nurse coordinator and research team spoke at community forums and research assistants passed out flyers in public places such as grocery stores and churches. Recruitment flyers were modified to indicate 'serious' rather than 'life-limiting' illness. Advisory members played an important role in recruiting participants and designing strategies to enhance recruitment. The most effective strategies were those that entailed personal contact such as consulting with healthcare providers and attending local meetings. Media strategies were costly and less effective.
Another factor that may have influenced recruitment was the use of the quality of life instrument. As part of the informed consent process, potential participants were introduced to the instrument and at that point four persons declined participation in the study. Although it was not stated explicitly, the nurse coordinator had a sense that the personal nature of the questions (eg asking individuals to rate feelings of sadness) was a deterrent to participation.
An additional unanticipated recruitment factor was the prevalence of partners caring for one another as they both coped with chronic life-limiting illnesses. This made it challenging to identify who was the participant and who was the FCG. The decision was made to register both as participants to reduce participant fatigue from filling out multiple measurements. Six participants constitute three dyads with chronic illness caring for one another. This decision does provide 'clean' data by not conflating participants and FCGs, but important data about the realities of coping with chronic illness while being a caregiver is lost. To date, approximately half of the participants have been recruited through healthcare providers and half have self-referred. Seventeen eligible persons have declined participation after receiving further education about the study. A common reason was that a family member encouraged them to participate but they had no perceived need of the service. Eight potential participants were screened and deemed ineligible: two had dementia, three were too well, and three were too sick and rapidly declining.
Table 1: RPaSS participant (n=23) and FCG (n=10) demographics
Visit protocols and measures
Visit statistics for the first 17 months of the service are shown in Table 2. Based upon this experience, it is estimated the nurse coordinator can care for about 25 participants and their FCG using bi-weekly visits. This is an estimated number only and is influenced by several factors including the complexity and stability of participants (those with a declining functional status, for example, may require more support from the nurse coordinator). These visits typically take about 1 hour and this duration does not change over time on the service. When RPaSS initially began supporting participants, visits were scheduled on a weekly basis; however, due to the stability of the majority of participants over time, the visit schedule was changed to bi-weekly visits in September 2013. To date, there have been five participant deaths and the PPS of most participants has been stable. The goal of recruiting participants early on the palliative trajectory has been achieved; only one participant had a PPS of less than 60 on admission. Modifying the visit schedule from weekly to bi-weekly allowed for some expansion of the service, but it is important to provide flexibility for more frequent visits should participants' wellbeing decline and more support be required. This is essential in a rural context where relationships and availability are essential to the perceived quality of care21,23.
One of the indicators of acceptability of an intervention is the number of declined visits. Over 17 months and 393 in-person visits, there have only been 19 declined visits. Requests for additional visits based upon perceived need are another indicator of acceptability. Of the 101 unscheduled visits completed by the nurse coordinator, 38% were at the request of the participant or FCG. No participants have withdrawn from the study, although one participant did not receive a visit for several months, citing a reluctance to focus on his/her chronic illness. This individual has since resumed visits. Figure 3 illustrates the nature of the needs addressed for participants and FCGs; the most common are related to disease management and the attending physical symptoms. Interventions to meet these needs have been teaching, referral, support, and physical care. In some cases, the nurse coordinator arranges more frequent contact to ensure that identified needs are resolved.
The use of regular structured assessments to determine quality of life and FCG needs has not been an onerous task for participants or FCGs as it becomes a routine part of the communication with each visit. However, the use of the Quality of Life Scale has required some modification in the implementation phase. Some participants have been uncomfortable with the personal nature of the quality of life questions. In this situation the Edmonton Symptom Assessment Scale Revised is used instead55,56, which participants have viewed as minimally intrusive. However, in doing so, standardized assessment information about potentially important quality of life domains, such as existential wellbeing and social support, is limited for these participants. Using a single instrument repeatedly does require flexibility when participants are not feeling well enough to complete the measures.
Table 2: RPaSS visit statistics, January 2013 to May 2014
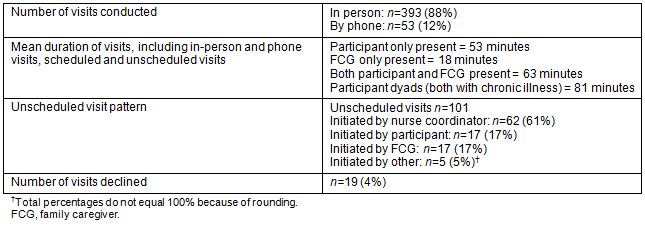
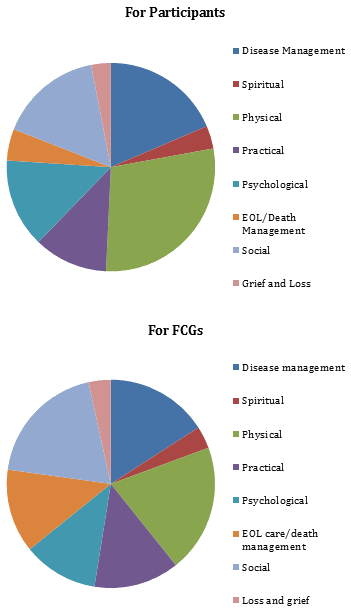 Figure 3: Distribution of needs identified and addressed by nurse coordinator.
Figure 3: Distribution of needs identified and addressed by nurse coordinator.
(Domains drawn from the Canadian Hospice Palliative Care's A model to guide hospice palliative care57.)
Discussion
The delivery of palliative care to rural populations requires innovative solutions that build upon the unique rural sociocultural context. This paper has described the feasibility of RPaSS as one potential solution. Several recommendations should be considered when initiating a similar service. Key to success of this initiative has been the community capacity-building approach whereby citizens of these communities have been involved in the design and implementation of RPaSS. This approach has required a continual process of negotiation between the 'ideal science' and the 'ideal service' as team members have sought to adapt the interventions and evaluation to best fit these community contexts. Allowing sufficient time to work through this process is essential. Although recruiting for the service was a slow process, after participants were on the service there were few declined visits and no attrition except through death. Focusing on educating physicians about the potential of RPaSS will facilitate recruitment efforts. Service capacity is approximately 25 participants through bi-weekly visits, which should be considered in light of sustainability. However, the nature of rural social connections and the relationships formed between the participants and the nurse coordinator are important to the success of the service; a less relationally oriented approach may change the nature of the acceptability of the intervention. The major focus of the nurse coordinator has been on supporting and teaching about chronic life-limiting disease management and the attending physical symptoms. The use of well-validated palliative care assessment instruments at the point of care has had multiple potential benefits including making visible patient and FCG insights into their healthcare experiences in a way that reduces observer bias, facilitating involvement in the plan of care, and systematically gathering data about patient and FCG experiences to increase accountability for the quality of care58-61.
Limitations
This study contributes important knowledge about the feasibility of a community-based clinical team led by a nurse coordinator to address the unmet needs of persons and families living with chronic life-limiting illness. However, claims cannot be made about the cost effectiveness of this service in relation to other models of care, in part because this is a population that is not currently receiving home-based services. Although many will receive home care later in the illness trajectory, the uniqueness of this service is the upstream approach to care. Further, despite the longitudinal collection of patient- and family-reported experiences and outcomes, it is not feasible to show statistically significant improvements in outcomes from the service because of the small sample size, the inability to isolate the effect of the service apart from the contributions of the primary care network, and the well-documented challenges of showing improvements in outcomes alongside the declines that are an inevitable part of progressive serious illness62. However, this model of care demonstrates the feasibility and utility of integrating a nurse coordinator led program with existing community services in a rural context.
Innovative solutions are required to meet the needs of rural populations aging with complex chronic illness. This article has described the feasibility of a service designed to provide an upstream palliative approach for those living in the community with chronic life-limiting illness. This model, led by a nurse coordinator, has the potential to smooth transitions and enhance quality of life along the disease trajectory and across locations of care by providing a consistent source of support and education. This type of continuity has the potential to foster the patient-and family-centered approach to care that is the ideal of a palliative approach. Further, the use of a rural community capacity-building approach may contribute to sustainability which is such an important part of rural health service delivery.
Acknowledgements
Funding for this study was provided by the Peter Wall Institute for Advanced Studies at the University of British Columbia (PW-11-020) and the Vancouver Foundation (UNR-12-0950).
Barbara Pesut is supported by a Canada Research Chair. Rick Sawatzky is supported by a Canada Research Chair. The authors would like to thank to the RPaSS Advisory Committee for their intellectual contributions to this project and to the research participants who have registered with the service.
References
1. Canadian Institute for Health Information. Healthcare in Canada 2011: a focus on seniors and aging. (Online) 2011. Available: https://secure.cihi.ca/free_products/HCIC_2011_seniors_report_en.pdf (Accessed 6 December 2013).
2. Canadian Institutes of Health Research Institute of Aging. Living longer, living better: Canadian Institutes of Health Research Institute of Aging 2013-18 strategic plan. (Online) 2013. Available http://www.cihr-irsc.gc.ca/e/documents/IA_Strategic_Plan_En_v5jul13.pdf (Accessed 6 December 2013).
3. British Columbia Ministry of Health. The provincial end-of-life care action plan for British Columbia: priorities and actions for health system and service redesign. (Online) 2013. Available: http://www.health.gov.bc.ca/library/publications/year/2013/end-of-life-care-action-plan.pdf (Accessed 6 December 2013).
4. Parliamentary Committee on Palliative and Compassionate Care. Not to be forgotten: care of vulnerable Canadians. (Online) 2011. Available: http://pcpcc-cpspsc.com/wp-content/uploads/2011/11/ReportEN.pdf (Accessed 6 December 2013).
5. Canadian Institute for Health Information. Health care use at the end of life in Western Canada. (Online) 2007. Available: https://secure.cihi.ca/free_products/end_of_life_report_aug07_e.pdf (Accessed 6 December 2013).
6. Cohen R, Sawatzky R. Quality of life assessment in palliative care. In: E Bruera, I Higginson, CF von Gunten, T Morita (Eds). Textbook of palliative medicine and supportive care, 2nd edn. Boca Raton, FL: CRC Press, 2014.
7. Carstairs S. Raising the bar: a roadmap for the future of palliative care in Canada. Ottawa, ON: Senate of Canada, 2010. Available: http://www.chpca.net/media/7859/Raising_the_Bar_June_2010.pdf (Accessed 12 September 2014).
8. Canadian Hospice Palliative Care Association. Integrating a palliative approach into the management of chronic, life-threatening diseases: who, how and when? (Online) 2012. Available: http://www.hpcintegration.ca/media/36315/TWF-integrating-palliative-approach-report-Eng_final3.pdf (Accessed 12 September 2014).
9. Dandy K, Bollman RD. Seniors in rural Canada. Rural and Small Town Canada Analysis Bulletin, Catalogue no. 21-006-X. Ottawa, ON: Statistics Canada. (Online) 2008. Available: http://www.statcan.gc.ca/pub/21-006-x/21-006-x2007008-eng.pdf (Accessed 12 September 2014).
10. Canadian Institute for Health Information. How healthy are rural Canadians: an assessment of their health status and health determinants. (Online) 2006. Available: https://secure.cihi.ca/free_products/rural_canadians_2006_report_e.pdf (Accessed 6 December 2013).
11. Laurent S. Rural Canada: access to health care. Report no. PRB02-45E. Ottawa, ON: Economics Division, Government of Canada. (Online) 2002. Available: http://publications.gc.ca/collections/Collection-R/LoPBdP/PRB-e/PRB0245-e.pdf (Accessed 12 September 2014).
12. Ministerial Advisory Council on Rural Health. Rural health in rural hands: strategic directions for rural, remote, Northern and Aboriginal communities. (Online) 2002. Available: http://www.srpc.ca/PDF/rural_hands.pdf (Accessed 6 December 2013).
13. Schuurman N, Crooks VA, Amram O. A protocol for determining differences in consistency and depth of palliative care service provision across community sites. Health & Social Care in the Community 2010; 18(5): 537-548.
14. Duggleby WD, Penz KL, Goodridge DM, Wilson DM, Leipert BD, Berry PH, et al. The transition experience of rural older persons with advanced cancer and their families: a grounded theory study. BMC Palliative Care 9: 5. (Online) 2010. Available: http://www.biomedcentral.com (Accessed 17 April 2014).
15. Wilson DM, Thomas R, Kovacs Burns KK, Hewitt JA, Osei-Waree J, Robertson S. Canadian rural-urban differences in end-of-life care setting transitions. Global Journal of Health Science 2012; 4(5): 1-13.
16. Canadian Institute for Health Information. End-of-life hospital care for cancer patients: executive summary. (Online) 2013. Available: https://secure.cihi.ca/free_products/Cancer_Report_EN_web_April2013.pdf (Accessed 6 December 2013).
17. Wilson DM, Joachim C, Deliens L, Hewitt JA, Houttekier D. The preferred place of last days: results of a representative population-based public survey. Journal of Palliative Medicine 2013; 16(5): 502-508.
18. Robinson CA, Pesut B, Bottorff JL. Supporting rural family palliative caregivers. Journal of Family Nursing 2012; 18(4): 467-490.
19. Brazil K, Kaasalainen S, Williams A, Dumont S. A comparison of support needs between rural and urban family caregivers providing palliative care. The American Journal of Hospice & Palliative Care 2014; 31(1): 13-19.
20. Brazil K, Kaasalainen S, Williams A, Rodriguez C. Comparing the experiences of rural and urban family caregivers of the terminally ill. Rural and Remote Health 13(1): 2250. (Online) 2013. Available: www.rrh.org.au (Accessed 17 April 2014).
21. Pesut B, Bottorff JL, Robinson CA. Be known, be available, be mutual: a qualitative ethical analysis of social values in rural palliative care. BMC Medical Ethics 12: 19. (Online) 2011. Available: http://www.biomedcentral.com (Accessed 17 April 2014).
22. Pesut B, Beswick F, Robinson CA, Bottorff JL. Philosophizing social justice in rural palliative care: Hayek's moral stone? Nursing Philosophy 2012; 13(1): 46-55.
23. Pesut B, Robinson CA, Bottorff JL. Among neighbours: an ethnographic account of responsibilities in rural palliative care. Palliative & Supportive Care 2014; 12(2): 127-138.
24. Sussman J, Barbera L, Bainbridge D, Howell D, Yang J, Husain A, et al. Health system characteristics of quality care delivery: a comparative case study examination of palliative care for cancer patients in four regions in Ontario, Canada. Palliative Medicine 2012; 26(4): 322-335.
25. Artnak KE, McGraw RM, Stanley VF. Health care accessibility for chronic illness management and end-of-life care: a view from rural America. Journal of Law, Medicine & Ethics 2011; 39(2): 140-155.
26. Duggleby WD, Penz K, Leipert BD, Wilson DM, Goodridge D, Williams A. 'I am part of the community but...' the changing context of rural living for persons with advanced cancer and their families. Rural and Remote Health 11(3): 1733. (Online) 2011. Available: www.rrh.org.au (Accessed 17 April 2014).
27. Kitchen P, Williams A, Chowhan J. Sense of community belonging and health in Canada: a regional analysis. Social Indicators Research 2012; 107(1): 103-126.
28. Williams AM, Kulig JC. Health and place in rural Canada. In: JC Kulig, AM Willams (Eds). Health in rural Canada. Vancouver, BC: UBC Press, 2012; 1-22.
29. Kelley ML. Developing rural communities' capacity for palliative care: a conceptual model. Journal of Palliative Care 2007; 23(3): 143-153.
30. Kelley ML, Williams A, DeMiglio L, Mettam H. Developing rural palliative care: validating a conceptual model. Rural and Remote Health 11(2): 1717. (Online) 2011. Available: www.rrh.org.au (Accessed 17 April 2014).
31. McIntosh MJ, Mettam H, Kelley ML, Williams A. Evaluating the knowledge utilization of a toolkit to enhance the capacity of rural communities to deliver palliative care. The Journal of Rural and Community Development 2012; 7(4): 92-108.
32. Kelley ML, DeMiglio L, Williams A, Eby J, McIntosh M. Community capacity development in palliative care: an illustrative case study in rural Northwestern Ontario. Journal of Rural and Community Development 2012; 7(4): 76-91.
33. Castleden H, Crooks VA, Schuurman N, Hanlon N. 'It's not necessarily the distance on the map...': using place as an analytic tool to elucidate geographic issues central to rural palliative care. Health & Place 2010; 16(2): 284-290.
34. Cinnamon J, Schuurman N, Crooks VA. A method to determine spatial access to specialized palliative care services using GIS. BMC Health Services Research 8: 140. (Online) 2008. Available: http://www.biomedcentral.com (Accessed 17 April 2014).
35. Crooks VA, Castleden H, Schuurman N, Hanlon N. Visioning for secondary palliative care service hubs in rural communities: a qualitative case study from British Columbia's Interior. BMC Palliative Care 8: 15. (Online) 2009. Available: http://www.biomedcentral.com (Accessed 17 April 2014).
36. du Plessis V, Beshiri R, Bollman RD, Clemenson HA. Definitions of 'rural'. Agriculture and Rural Working Paper Series, Catalogue no. 21-601-MIE. Ottawa, ON: Statistics Canada, 2002.
37. Markey S, Halseth G, Manson D. Capacity, scale and place: pragmatic lessons for doing community-based research in the rural setting. The Canadian Geographer 2010; 54(2): 158-176.
38. Stoecker R. Research methods for community change. Los Angeles, CA: Sage, 2013.
39. Moss AH, Lunney JR, Culp S, Auber M, Kurian S, Rogers J, et al. Prognostic significance of the 'surprise' question in cancer patients. Journal of Palliative Medicine 2010; 13(7): 837-840.
40. McGregor D, Porterfield P. Identification of patients who may benefit from palliative care [unpublished Powerpoint presentation]. Vancouver Coastal Health, 2009.
41. Fillion L, Cook S, Veillette AM, Aubin Ml, de Serres M, Rainville F, et al. Professional navigation framework: elaboration and validation in a Canadian context. Oncology Nursing Forum 2012; 39(1): E58-E69.
42. Case MAB. Oncology nurse navigator: ensuring safe passage. Clinical Journal of Oncology Nursing 2011; 15(1): 33-40.
43. Anderson F, Downing GM, Hill J, Casorso L, Lerch N. Palliative Performance Scale (PPS): a new tool. Journal of Palliative Care 1996; 12(1): 5-11.
44. Ho F, Lau F, Downing MG, Lesperance M. A reliability and validity study of the Palliative Performance Scale. BMC Palliative Care 7: 10. (Online) 2008. Available: http://www.biomedcentral.com (Accessed 17 April 2014).
45. Cohen SR, Mount BM, Strobel MG, Bui F. The McGill Quality of Life Questionnaire: a measure of quality of life appropriate for people with advanced disease. A preliminary study of validity and acceptability. Palliative Medicine 1995; 9(3): 207-219.
46. Cohen SR, Mount BM, Bruera E, Provost M, Rowe J, Tong K. Validity of the McGill Quality of Life Questionnaire in the palliative care setting: a multi-centre Canadian study demonstrating the importance of the existential domain. Palliative Medicine 1997; 11(1): 3-20.
47. Dumont S, Turgeon J, Allard P, Gagnon P, Charbonneau C, Vezina L. Caring for a loved one with advanced cancer: determinants of psychological distress in family caregivers. Journal of Palliative Medicine 2006; 9(4): 912-921.
48. Hileman JW, Lackey NR, Hassanein RS: Identifying the needs of home caregivers of patients with cancer. Oncology Nursing Forum 1992; 19(5): 771-777.
49. Faschingbauer TR, Zisook S, DeVaul R. The Texas Revised Inventory of Grief. In: S Zisook (Eds). Biopsychosocial aspects of bereavement. Washington, DC: American Psychiatric Press, 1987; 109-124.
50. Drennan J. Cognitive interviewing: verbal data in the design and pretesting of questionnaires. Journal of Advanced Nursing 2003; 42(1): 57-63.
51. Hawe P, Noort M, Gifford SM, Lloyd B. Working invisibly: health workers talk about capacity-building in health promotion. Health Promotion International 1998; 13(4): 285-296.
52. Hayes H, Parchman ML, Howard R. A logic model framework for evaluation and planning in a primary care practice-based research network (PBRN). Journal of the American Board of Family Medicine 2011; 24(5): 576-582.
53. Knowlton LW, Phillips CC. The logic model guidebook: better strategies for great results. Los Angeles, CA: Sage, 2013.
54. Pesut B, Hooper B, Sawatzky R, Robinson CA, Bottorff JL, Dalhuisen M. Program assessment framework for a rural palliative supportive service. Palliative Care: Research and Treatment 2013; 7: 7-17.
55. Bruera E, Kuehn N, Miller MJ, Selmser P, Macmillan K. The Edmonton Symptom Assessment System (ESAS): a simple method for the assessment of palliative care patients. Journal of Palliative Care 1991; 7: 6-9.
56. Watanabe SM, Nekolaichuk C, Beaumont C, Johnson L, Myers J, Strasser F. A multi-centre comparison of two numerical versions of the Edmonton Symptom Assessment System in palliative care patients. Journal of Pain and Symptom Management 2011; 41: 456-468.
57. Ferris FD, Balfour HM, Bowen K, Farley J, Hardwick M, Lamontagne C, et al. A model to guide hospice palliative care. Ottawa, ON: Canadian Hospice Palliative Care Association, 2002.
58. Black N. Patient reported outcome measures could help transform healthcare. British Medical Journal 346: f167. (Online) 2013. Available: http://www.bmj.com (Accessed 17 April 2014).
59. Antunes B, Harding R, Higginson IJ. Implementing patient-reported outcome measures in palliative care clinical practice: a systematic review of facilitators and barriers. Palliative Medicine 2014; 28(2): 158-175.
60. Greenhalgh J. The applications of PROs in clinical practice: what are they, do they work, and why? Quality of Life Research: An International Journal of Quality of Life Aspects of Treatment, Care and Rehabilitation 2009; 18(1): 115-123.
61. Bausewein C, Simon ST, Benalia H, Downing J, Mwangi-Powell F, Daveson BA, et al. Implementing patient reported outcome measures (PROMs) in palliative care - users' cry for help. Health and Quality of Life Outcomes 2011; 9(1): 27-37.
62. John Hopkins University Evidence-based Practice Center. Improving health care and palliative care for advanced and serious illness. Closing the quality gap: revisiting the state of the science. Report no.: 12(13)-E014-EF. Rockville, MD: Agency for Healthcare Research and Quality, 2012.
