Recently, we reported that over half the subsistence hunters from Fort Albany First Nation were aware of avian influenza virus (AIV) being a potential health hazard1. Over a quarter of the total participants perceived a risk of contracting this virus while harvesting birds1. The perception of this risk is not unfounded as several studies have noted that hunters and some bird handlers, such as wildlife biologists, may be at increased risk of exposure to highly pathogenic avian influenza A (HPAI) viruses2-6. Within hunters, HPAI exposure risk varies greatly by location due to the viral prevalence observed during different hunting seasons in birds, species of bird, the average number of birds harvested per hunting period, and the age and ratio of juveniles to adult birds harvested7,8. The group of hunters in this study is particularly vulnerable to exposure due to their subsistence lifestyle combined with the knowledge that HPAI outbreaks have occurred in migratory waterfowl, which is their primary hunting target9,10.
Although relatively rare in human occurrence, the high fatality rate associated with HPAI infection creates a continued cause for human health concern11. The most recent data (1 May 2015) from the WHO reports 840 cases of H5N1 resulting in 447 deaths since 2003 across the globe12. Furthermore, the high hunter contact rate of birds in this study may act as an additional danger due to these people's remoteness and isolation from adequate medical facilities that can recognize and combat this virus. Campagna and colleagues (2011) recently reported that seropositivity of 10 zoonotic infections from two First Nations communities in Quebec was associated with hunting practices13. Because many remote First Nations communities rely on hunted meats for subsistence, they are at an increased risk of AIV exposure as well. In response to community hunter and Band Council (locally elected government) concerns about HPAI (particularly the H5 and H7 subtypes) in the game birds harvested from the Fort Albany First Nation region of northern Ontario, Canada, we assessed a range of hunted fowl for influenza A virus.
All sampling was conducted under the guidance of a trained local community coordinator, all hunting was performed by local traditional hunters, and the project was approved by the Fort Albany Band Council. Cloacal swabs (Starplex Scientific, Etobicoke, Canada) were taken in the spring and autumn hunting seasons of 2013 and in the spring hunting season of 2014. Samples were frozen at -20oC and shipped directly to the laboratory for analysis. Total nucleic acids were extracted from the cloacal swab samples using the MagMAX-96 viral ribonucleic acid (RNA) Isolation Kit in a MagMAX Express-96 Magnetic Particle Processor (Applied Biosystems Inc., Foster City, California). Extracted nucleic acids were screened for the presence of influenza A virus RNA using a real-time reverse transcription polymerase chain reaction targeting a conserved region of influenza A virus M gene14. For hemagglutinin gene subtyping, influenza A virus RNA was amplified using a reverse transcription polymerase chain reaction (PCR)15 and nucleotide sequences of PCR products were determined at the University of Guelph Laboratory Services sequencing facility. Sequences were assembled using the Lasergene software (DNAStar Inc., https://www.dnastar.com) and compared with hemagglutinin gene sequences in GenBank, the US National Institutes of Health genetic sequence database. In total, 304 cloacal swabs were analyzed; the number of samples by species is presented in Table 1. The results of this project were presented to the community under the direction of the community coordinator and the researchers.
Table 1: Number of cloacal swabs, by wild game bird type, obtained in a remote First Nation community in subArctic Ontario, Canada, 2013 and 2014

A 5.26% (n=16) AIV prevalence was found in the 304 cloacal swab samples analyzed. Of the 16 positive samples, 12 were found in mallard ducks, 3 were found in snow geese (wavies), and 1 positive sample was found in partridge (Fig1). Three mallard ducks were successfully subtyped for H3 and one mallard duck was subtyped for H4. The remaining positive samples were unable to be subtyped either due to being positive for multiple strains of avian influenza A, or due to reduced sensitivities in the conventional PCR process used to generate the template for sequencing.
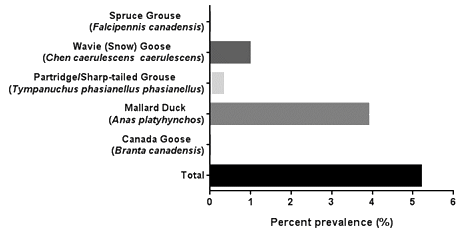
Figure 1: Point prevalence of avian influenza virus in individual bird species sampled and total sample population.
Discussion
To our knowledge, the pilot work presented herein is the first to examine hunter-harvested birds from a subsistence First Nation community. Globally, surveillance and reporting of avian influenza remains weak16,17, and the community-level data presented here is the first step towards mapping risk and exposure by identifying which bird species may be acting as reservoirs or pathogen vectors18. Others have successfully used surveillance data to assess risk from avian influenza19,20 and similar application of this data could provide added security to subsistence hunting communities.
HPAI remains a concern globally, and those who hunt and handle birds may be at additional risk. However, no HPAI subtypes were identified in any of the samples collected by community members of Fort Albany First Nation; only low pathogenic AIV was found. The H3 and H4 low pathogenic AIV subtypes are most commonly found among dabbling ducks8, and this was represented in this study. Ferro et al. found that dabbling ducks and snow geese had lower prevalences of AIV (10.9% and 0%, respectively) in hunter-harvested waterfowl from the Texas coast21 when compared to our study (28.6% for mallard duck only and 6.3%, respectively). AIV prevalence within dabbling ducks varies, and others have reported a 24.3% prevalence in mallard ducks, which is similar to our value22. Stallknecht et al. found that prevalence of AIV varied by season23, and others have noted that age, species, location, and number of birds harvested all cause variation in AIV prevalence7,8,24. While it is possible that we did not capture true prevalence of all birds from the Fort Albany community, the majority of the samples were collected during the two main hunting seasons (spring and autumn) and this represents the highest potential hazard period for hunters. Additionally, research suggests that northern breeding grounds in colder environments may act as reservoirs for some types of AIV, even though they might seem unfavorable25.
Community-level surveillance of AIV in remote subsistence hunting communities is an important aspect of identifying risks and working to control a potential outbreak that may otherwise lead to adverse social and economic effects26. While general Canadian pandemic plans27 and recommendations for First Nations communities28 have been developed, data such as that presented here can help address monitoring gaps and improve hunter education. This is an important first step to assessing risk as Aboriginal (First Nations, Inuit, and Metis} people have had the highest rate of H1N1 worldwide29 and a HPAI outbreak in a remote community would have significant adverse consequences. Although only low pathogenic AIV was found in the 304 cloacal swab samples tested herein, ongoing monitoring of birds from areas with potentially high exposure groups such as subsistence hunters is recommended.
Acknowledgements
We wish to thank the hunters of Fort Albany First Nation who participated in this research project. Funding was provided by Health Canada's Regional First Nations Environmental Contaminants Program and a Ryerson University Faculty of Community Services Seed Grant.
References
1. Charania NA, Martin ID, Liberda EN, Meldrum R, Tsuji LJ. Bird harvesting practices and knowledge, risk perceptions, and attitudes regarding avian influenza among Canadian First Nations subsistence hunters: implications for influenza pandemic plans. BMC Public Health 2014; 14: 1113. https://doi.org/10.1186/1471-2458-14-1113
2. Reed C, Bruden D, Byrd KK, Veguilla V, Bruce M, Hurlburt D, et al. Characterizing wild bird contact and seropositivity to highly pathogenic avian influenza A (H5N1) virus in Alaskan residents. Influenza and Other Respiratory Viruses 2014; 8(5): 516-523. https://doi.org/10.1111/irv.12253
3. Shafir SC, Fuller T, Smith TB, Rimoin AW. A national study of individuals who handle migratory birds for evidence of avian and swine-origin influenza virus infections. Journal of Clinical Virology 2012; 54(4): 364-367. https://doi.org/10.1016/j.jcv.2012.05.001
4. Gray GC, Ferguson DD, Lowther PE, Heil GL, Friary JA. A national study of US bird banders for evidence of avian influenza virus infections. Journal of Clinical Virology 2011; 51(2): 132-135. https://doi.org/10.1016/j.jcv.2011.03.011
5. Gray GC, McCarthy T, Capuano AW, Setterquist SF, Alavanja MC, Lynch CF. Evidence for avian influenza A infections among Iowa's agricultural workers. Influenza and Other Respiratory Viruses 2008; 2(2): 61-69. https://doi.org/10.1111/j.1750-2659.2008.00041.x
6. Gill JS, Webby R, Gilchrist MJ, Gray GC. Avian influenza among waterfowl hunters and wildlife professionals. Emerging Infectious Diseases 2006; 12(8): 1284-1286. https://doi.org/10.3201/eid1708.060492
7. Dorea FC, Cole DJ, Stallknecht DE. Quantitative exposure assessment of waterfowl hunters to avian influenza viruses. Epidemiology and Infection 2013; 141(5): 1039-1049. https://doi.org/10.1017/S0950268812001720
8. Kistler WM, Stallknecht DE, DeLiberto TJ, Van Why K, Yabsley MJ. Subtype-specific influenza A virus antibodies in Canada geese (Branta canadensis). Veterinary Microbiology 2015; 177(3-4): 296-301. https://doi.org/10.1016/j.vetmic.2015.03.021
9. Chen H, Smith GJ, Zhang SY, Qin K, Wang J, Li KS, et al. Avian flu: H5N1 virus outbreak in migratory waterfowl. Nature 2005; 436(7048): 191-192. https://doi.org/10.1038/nature03974
10. Normile D. Avian influenza. Evidence points to migratory birds in H5N1 spread. Science 2006; 311(5765): 1225. https://doi.org/10.1126/science.311.5765.1225
11. Van Kerkhove MD. Brief literature review for the WHO global influenza research agenda - highly pathogenic avian influenza H5N1 risk in humans. Influenza and Other respiratory Viruses 2013; 7(Suppl 2): 26-33. https://doi.org/10.1111/irv.12077
12. WHO, Global Influenza Programme. Cumulative number of confirmed human cases for avian influenza A(H5N1) reported to WHO, 2003-2015. (Internet) 2015. Available: http://www.who.int/influenza/human_animal_interface/EN_GIP_20150501CumulativeNumberH5N1cases.pdf?ua=12015 (Accessed 1 June 2015).
13. Campagna S, Levesque B, Anassour-Laouan-Sidi E, Cote S, Serhir B, Ward BJ, et al. Seroprevalence of 10 zoonotic infections in 2 Canadian Cree communities. Diagnostic Microbiology and Infectious Disease 2011; 70(2): 191-199. https://doi.org/10.1016/j.diagmicrobio.2011.01.009
14. Weingartl HM, Berhane Y, Hisanaga T, Neufeld J, Kehler H, Emburry-Hyatt C, et al. Genetic and pathobiologic characterization of pandemic H1N1 2009 influenza viruses from a naturally infected swine herd. Journal of Virology 2010; 84(5): 2245-2256. https://doi.org/10.1128/JVI.02118-09
15. Phipps LP, Essen SC, Brown IH. Genetic subtyping of influenza A viruses using RT-PCR with a single set of primers based on conserved sequences within the HA2 coding region. Journal of Virological Methods 2004; 122(1): 119-122. https://doi.org/10.1016/j.jviromet.2004.08.008
16. Huston P. The advance of H5N1/La progression du H5N1. Canadian Journal of Public Health 2005; Nov-Dec: 405.
17. Butler D. 'Refusal to share' leaves agency struggling to monitor bird flu. Nature 2005; 435(7039): 131. https://doi.org/10.1038/435131a
18. Caron A, de Garine-Wichatitsky M, Chiweshe N, Gaidet N, Cumming GS. Estimating dynamic risk factors for pathogen transmission using community-level bird census data at the wildlife/domestic interface. Ecology and Society 2010; 15(3).
19. Cumming GS, Hockey PAR, Bruinzeel LW, Du Plessis MA. Wild bird movements and avian influenza risk mapping in Southern Africa. Ecology and Society 2008; 13(2). https://doi.org/10.5751/ES-02536-130226
20. Peterson AT, Williams RAJ. Risk mapping of highly pathogenic avian influenza distribution and spread. Ecology and Society 2008; 13(2). https://doi.org/10.5751/ES-02532-130215
21. Ferro PJ, Khan O, Peterson MJ, Batchuluun D, Reddy SM, Lupiani B. Avian influenza virus surveillance in hunter-harvested waterfowl, Texas Coast, September 2009 - January 2010. Avian Diseases 2012; 56(4s1): 1006-1009.
22. Wilcox BR, Knutsen GA, Berdeen J, Goekjian V, Poulson R, Goyal S, et al. Influenza-A viruses in ducks in Northwestern Minnesota: fine scale spatial and temporal variation in prevalence and subtype diversity. PLoS ONE 2011; 6(9): e24010. https://doi.org/10.1371/journal.pone.0024010
23. Stallknecht DE, Shane SM, Zwank PJ, Senne DA, Kearney MT. Avian influenza viruses from migratory and resident ducks of coastal Louisiana. Avian Diseases 1990; 34(2): 398-405. https://doi.org/10.2307/1591427
24. Olsen B, Munster VJ, Wallensten A, Waldenstrom J, Osterhaus AD, Fouchier RA. Global patterns of influenza A virus in wild birds. Science 2006; 312(5772): 384-388. https://doi.org/10.1126/science.1122438
25. Henaux V, Samuel MD, Dusek RJ, Fleskes JP, Ip HS. Presence of avian influenza viruses in waterfowl and wetlands during summer 2010 in California: are resident birds a potential reservoir? PloS ONE 2012; 7(2): e31471. https://doi.org/10.1371/journal.pone.0031471
26. Fukuda K, WHO, Global Influenza Programme. Pandemic influenza preparedness and response, a WHO guidance document. (Internet) 2009. Available: http://public.eblib.com/choice/publicfullrecord.aspx?p=434246 (Accessed 24 June 2015).
27. Public Health Agency of Canada. The Canadian Pandemic Influenza Plan for the Health Sector pandemic influenza - Pandémie d'influenza. (Internet) 2006. Available: http://www.deslibris.ca/ID/205568 (Accessed 24 June 2015).
28. Charania NA, Tsuji LJS. Recommended mitigation measures for an influenza pandemic in remote and isolated First Nations communities of Ontario, Canada: a community-based participatory research approach. International Indigenous Policy Journal 2014; 5(3): 1-22. https://doi.org/10.18584/iipj.2014.5.3.2
29. Kermode-Scott B. Canada has world's highest rate of confirmed cases of A/H1N1, with Aboriginal people hardest hit. British Medical Journal 2009; 339(jul06 2): b2746-b.
