Osteoporosis is a chronic disorder characterized by loss of bone mass and deterioration of bone micro-architecture1. The result is in an increased skeletal fragility and increased susceptibility to fracture1. Osteoporotic fractures are associated with pain, decreased quality of life, disability, hospital and nursing home admissions and premature death. Also, they are an economic burden on the healthcare system1,2. Thus, the diagnosis of osteoporosis is important to promote treatment.
Osteoporosis is common in ageing populations and age is a dominating risk factor1,3,4. Life expectancy is rising rapidly in Greenland and a focus on osteoporosis could be relevant as the disease is often overlooked even in societies characterized by frequent fragility fractures and high life expectancy5. This may be speculated to be even more relevant in societies with limited awareness of a disease that may be emerging.
The westernized lifestyle is gaining ground in Greenland and hence there is a decreasing intake of traditional food items, which also associates with a decrease in the intake of vitamin D6,7. As vitamin D is important for skeletal health this is likely to add to an increased risk of osteoporosis in the ageing population in Greenland.
Risk factors for osteoporosis are common in populations in Greenland8 and it may be speculated that the rising number of ageing citizens, as well as the changing diet and limited sun exposure during winter9, further increase the occurrence of osteoporosis among populations in Greenland and throughout the Arctic. This calls for a strategy to diagnose osteoporosis in the Arctic.
Osteoporosis may be diagnosed in two ways: by low bone mineral density (BMD) or by the occurrence of a fragility fracture. BMD is measured by dual energy X-ray absorptiometry (DXA). However, DXA is not available in Greenland. The occurrence of a fracture is assessed by radiographs. The most common fragility fracture is vertebral10. Vertebral fractures are visible on lateral chest radiographs, which may be used to diagnose osteoporosis11-13 that merits treatment1,2. The present case from a small hospital on the west coast of Greenland illustrates a procedure for diagnosing osteoporosis outside the referral hospital in Greenland.
A 51-year-old Greenlandic woman attended the outpatient clinic at the local hospital in Sisimiut, Greenland, due to lower back pain. The back pain had persisted for months. No trauma could be recalled and the patient had not carried any heavy weight. On the physical examination the patient had normal gait, no neurological deficits and normal muscle strength. Laboratory results showed a low vitamin D level (P-25-OH-vitamin D 28 nM, reference >50 nM) but normal total S-calcium level (2.40 mM, reference 2.2-2.7 mM), S-calcium++ (1.26 mM, reference 1.17-1.34 mM), S-phosphate (1.2 mM, reference 0.8-1.5 mM), S-parathyroid hormone 1-84 (2.6 pM, reference 0.3-14.6 pM), S-thyrotropin (0.67 miu/L, reference 0.4-3.8 miu/L), erythrocyte sedimentation rate (7 mm, reference <30 mm), S-creatinine (73 µM, reference 44-114 µM) and hemoglobin (9.0 mM, reference 7.0-9.4 mM). A radiograph of the lumbar spine showed severe spondyloarthrosis and slight scoliosis but all vertebrae were of normal height and without fractures. A chest radiograph was taken (Fig1), which demonstrated compression fractures of the seventh and ninth thoracic vertebrae with a reduction of anterior height of the vertebral bodies of 50% and 40% respectively. The patient's risk factors for osteoporosis are listed in Table 1 along with a line-up of the risk factors included in the QFracture algorithm and in the FRAX fracture risk calculator. Dual energy X-ray absorptiometry for measurement of bone density is not available in Greenland.
The patient was thus diagnosed with osteoporosis based on the two vertebral compression fractures without trauma. The patient had an insufficient vitamin D level. Treatment with vitamin D was recommended followed by an oral anti-resorptive drug once weekly.
Table 1: Risk factors for osteoporosis in the patient and whether they are included in two commonly used osteoporosis risk assessment tools, FRAX and QFracture
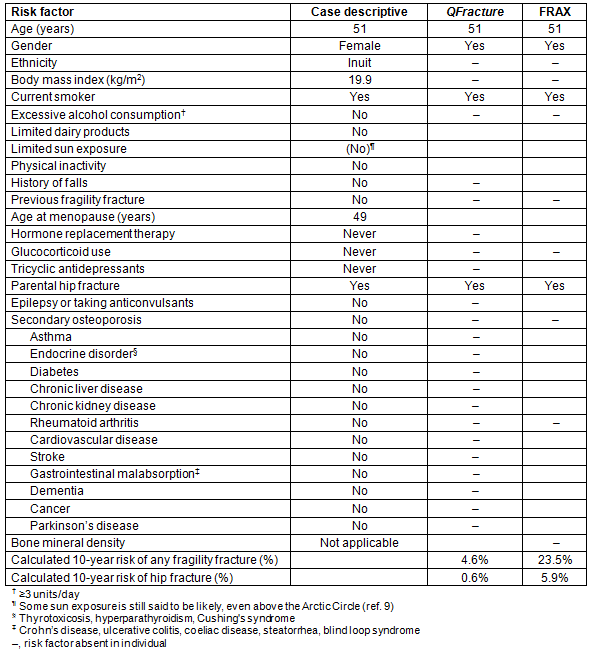
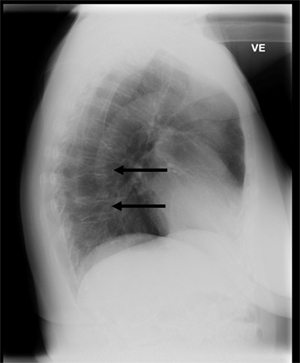
Figure 1: Lateral chest radiograph taken at Sisimiut Hospital on the west coast of Greenland in a patient with back pain but no trauma. It illustrates two vertebral fractures that are diagnostic of osteoporosis in the patient. Risk factors for osteoporosis included smoking, parental hip fracture and female gender. Vitamin D level was in the range of insufficiency (28 nmol/L) but sedimentation rate was normal, and serum calcium, alkaline phosphatase and parathyroid hormone were within the reference ranges.
The patient presented here illustrates a case of vertebral fractures without trauma and hence osteoporotic fractures. Vertebral fractures associate with a rise in the occurrence of future fractures, acute and chronic pain, deformity, disability, decreased quality of life and a markedly increased risk of premature death1,2. It is thus a serious condition that mandates aggressive treatment1,2. However, detection and diagnosis depend on a dedicated diagnostic protocol and focus among healthcare providers. The case presented here illustrates a simple diagnostic tool for osteoporosis that is feasible in remote towns in Greenland and may be used elsewhere in rural and remote areas.
Osteoporosis may be common in Greenland. Sørensen et al11 found that vertebral fractures were very frequent from an evaluation of all lateral chest radiographs performed over a period of 3 months at Greenland's referral hospital in the capital city, Nuuk. This is in keeping with frequent occurrence of radiographically diagnosed fractures at osteoporotic sites without trauma reported in a questionnaire survey in Greenland8. These reports support the emergence of osteoporosis in Greenland. In addition, low treatment frequency for osteoporosis was concluded from the evaluation of dispensing records from the National Pharmacy in Greenland11. Hence, Sørensen et al demonstrated the usefulness of the readily available lateral chest radiograph to diagnose vertebral fractures that merit treatment, and illustrated a striking contrast between a low treatment frequency for osteoporosis in Greenland and the high frequency of fractures at a single osteoporotic site11. This supports the need for a focus on diagnosis of osteoporosis and on the treatment of osteoporosis among the ageing population in Greenland.
The identification of patients at high risk of fragility fractures has become a major topic globally in order to support primary prevention (ie prevent fractures), as effective and cheap therapies are available1,2. A number of tools have been developed to predict the risk of fragility fractures. Most of these rely on the assessment of risk factors with or without measurement of BMD (Table 1). Measurement of BMD by DXA may increase the sensitivity of these tools. However, around one in three people with risk factors for osteoporosis and a low BMD suggesting osteoporosis by DXA do not have an osteoporotic fracture at old age5, and one in five with fragility fractures have non-osteoporotic BMD values5. Thus, the added value of DXA is debated. The risk of fragility fractures may be assessed from clinical risk factors for osteoporosis and BMD.
Age is a dominating risk factor for osteoporosis in other populations1,3,4. Greenland is a developing society, and life expectancy and the number of older people is rising steeply. It may be hypothesized that this will increase the occurrence of fragility fractures in Greenland. This is supported by the finding that age associates with an increased occurrence of vertebral fractures in Greenland. This is illustrated in Figure 2, which is based on data from the study by Sørensen et al11 and further supported by the data by Jakobsen et al8. Hence, the increase in life expectancy in the developing society warrants a focus on osteoporosis.
Low levels of vitamin D are associated with an increased risk of fragility fractures14. The traditional Inuit diet is of marine origin, rich in vitamin D, and the intake is associated with levels of vitamin D in populations in Greenland6,7. A diet based on westernized food items is advancing in Greenland and this may be a harbinger of low vitamin D levels in the population6,7. In addition, limited sun exposure with the westernized lifestyle in the Arctic might add to the risk of vitamin D insufficiency9. The reduced intake of vitamin D from the diet and from sun exposure may be hypothesized to further increase the risk of osteoporosis in populations in Greenland.
Ethnicity may influence the risk of osteoporosis. The risk is influenced by ethnic origins such as Indian, Pakistani, Bangladeshi, other Asian, black African, black Caribbean and Chinese. However, it remains unsettled whether Inuit ethnicity is of importance for the risk of osteoporosis. There is some indication that the accumulation of risk factors, known to influence the risk of fractures in other populations, increases the risk of fracture in populations in Nuuk in Greenland8. However, these data are from a questionnaire survey and they need confirmation. Furthermore, the genetic admixture may complicate the discrimination of genetically distinct ethnic groups. Yet, the importance of ethnicity is upheld in osteoporosis risk evaluation by, for example, the QFracture algorithm15 and should be considered in Greenland.
Smoking is an established risk factor for osteoporosis4,15,16. Smoking has a high prevalence among populations in Greenland17 and it adds to the risk of osteoporosis. Additional Arctic risk factors included are the increased risk of falling during the Arctic winter and the frequent occurrence of type 2 diabetes in Greenland18 as diabetes markedly increases bone fragility19. These frequent risk factors add to the need for a focus on osteoporosis in Greenland.
A gap was described in the occurrence and management of osteoporosis between urban and rural areas in Canada20, with lower health resource utilization rates in small towns and rural areas that was associated with the frequency of fragility fractures20. This needs to be addressed, and our case supports focus on management of osteoporosis using existing electronic facilities. Such facilities could be linked to and supported by an electronic consult program such as has been shown recently to improve osteoporosis evaluation and treatment frequency in rural and remote areas21. This should not stand alone, and educational programs22 as well as preventive measures that are likely to reduce the occurrence of fragility fractures23 may support recognition of symptoms and focus on management of emerging chronic diseases such as osteoporosis.
The case presented here was from the west coast of Greenland. The patient presented at the local hospital with back pain. Clinical risk factors for osteoporosis included current smoking and a parental history of an osteoporotic fracture. On lateral chest radiograph she had two severe vertebral fractures. No relevant trauma could be recalled and she was diagnosed with osteoporosis based on two fragility fractures. This demonstrates the usefulness of a lateral chest radiograph for diagnosing osteoporosis in a remote town. The use of available lateral chest radiograph is a simple tool for diagnosing vertebral fractures and hence osteoporosis that should be treated1,2.
Treatment should be initiated in all patients with an osteoporotic fracture as a secondary prophylactic measure because these fractures are harbingers of future fracture24, pain, disability and premature death1,2. The detection of fragility fractures from radiographs is highly sensitive given a history that rules out trauma. The sensitivity for any osteoporotic fracture is limited by the area visible, but the lower thoracic spine is a main site for vertebral fractures11,24, which supports the suggested focus on chest radiographs.
Radiographs are feasible in the absence of other testing. The radiation dose is markedly higher than that of DXA and it is thus unsuitable as an unrestricted screening tool. However, it is suggested to develop a tool to detect those at the highest risk for a targeted high-risk screening program. Existing tools such as FRAX, QFracture and others were developed to assess subjects for either screening with DXA for treatment or for plain treatment decisions25,26. In the evaluation of these tools, the aim has been a high sensitivity of around 90%. This was a trade-off causing a specificity reduced to around 40-50% for diagnosing osteoporosis by DXA or fracture25,26. The more simple screening tools suffice in this context26, while evaluations are limited for assessment of osteoporosis fracture risk prediction models with a higher specificity. A tool with higher specificity should be evaluated for use in remote populations. This could be further assessed for the benefit of using radiographs for treatment decisions.
Until an osteoporosis-specific tool has been validated for higher specificity, evaluation of lateral chest radiographs is a simple tool that may be applied throughout remote Arctic societies. The need for this is emphasized by the frequent occurrence of risk factors known to increase the risk of osteoporotic fractures among other populations. There is some indication that the same risk factors account for Inuit but this needs confirmation. If confirmed, a diagnostic protocol such as an Inuit version of FRAX or QFracture may guide diagnosis of osteoporosis among Inuit populations. This approach is appealing in rural and remote areas where DXA is unavailable or inaccessible, such as the sparsely populated Arctic.
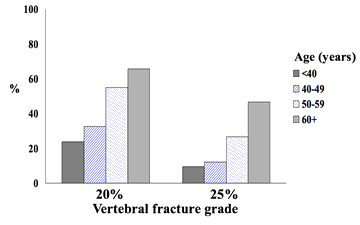
Figure 2: Occurrence of vertebral fractures increased with age (p<0.001). (Data from the study by Sørensen et al11 )
References
1. Papaioannou A, Watts NB, Kendler DL, Yuen CK, Adachi JD, Ferko N. Diagnosis and management of vertebral fractures in elderly adults. American Journal of Medicine 2002; 113(3): 220-228.
2. Ishtiaq S, Fogelman I, Hampson G. Treatment of post-menopausal osteoporosis: beyond bisphosphonates. Journal of Endocrinological Investigations 2015; 38(1): 13-29.
3. Kanis JA, Adami S. Bone loss in the elderly. Osteoporosis International 1994; 4(Suppl 1): 59-65.
4. Andersen S, Laurberg P. Age impact on clinical risk factors does not justify the age related change in referral pattern for osteoporosis assessment - data from the Aalborg University Hospital Record for Osteoporosis Risk Assessment (AURORA). Maturitas 2015; 80(3): 302-307.
5. Andersen S, Laurberg P. Age discrimination in osteoporosis screening - data from the Aalborg University Hospital Record for Osteoporosis Risk Assessment (AURORA). Maturitas 2014; 77(4): 330-335.
6. Andersen S, Laurberg P, Hvingel B, Kleinschmidt K, Heickendorff L, Mosekilde L. Vitamin D status in Greenland is influenced by diet and ethnicity: a population-based survey in an Arctic society in transition. British Journal of Nutrition 2013; 109(5): 928-935.
7. Nielsen NO, Jorgensen ME, Friis H, Melbye M, Soborg B, Jeppesen C, et al. Decrease in vitamin D status in the Greenlandic adult population from 1987-2010. PLoS One 2014; 9(12): e112949.
8. Jakobsen A, Laurberg P, Vestergaard P, Andersen S. Clinical risk factors for osteoporosis are common among elderly people in Nuuk, Greenland. International Journal of Circumpolar Health 2013; 72: 19596.
9. Andersen S, Jakobsen A, Rex HL, Lyngaard F, Kleist IL, Kern P, et al. Vitamin D status in Greenland - dermal and dietary donations. International Journal of Circumpolar Health 2013; 72.
10. Warriner AH, Patkar NM, Curtis JR, Delzell E, Gary L, Kilgore M, et al. Which fractures are most attributable to osteoporosis? Journal of Clinical Epidemiology 2011; 64(1): 46-53.
11. Sørensen VN, Wojtek P, Pedersen DS, Andersen S. An efficient case finding strategy to diagnose osteoporosis in a developing society with low treatment frequency. Journal of Endocrinological Investigations 2015; 38(8): 841-847.
12. Clemmensen K, Videbaek O. Osteoporosis in unselected patients in an ambulatory clinic for pulmonary medicine. An inexpensive diagnostic possibility not requiring extra time. Ugeskrift for Laeger 1998; 160(34): 4882-4885.
13. Oschatz E, Prosch H, Kohansal R, Valipour A, Mostbeck G. COPD and osteoporosis: detection and grading of vertebral fractures on lateral chest radiography. Journal of Thoracic Imaging 2009; 24(3): 212-215.
14. Maier GS, Seeger JB, Horas K, Roth KE, Kurth AA, Maus U. The prevalence of vitamin D deficiency in patients with vertebral fragility fractures. Bone Joint Journal 2015; 97-B(1): 89-93.
15. Hippisley-Cox J, Coupland C. Derivation and validation of updated QFracture algorithm to predict risk of osteoporotic fracture in primary care in the United Kingdom: prospective open cohort study. British Medical Journal 2012; 344: e3427.
16. Kanis JA, Johansson H, Oden A, McCloskey EV. Assessment of fracture risk. European Journal of Radiology 2009; 71(3): 392-397.
17. Bjerregaard P, Jørgensen ME, Borch-Johnsen K. Cardiovascular risk amongst migrant and non-migrant Greenland Inuit in a gender perspective. Scandinavian Journal of Public Health 2007; 35(4): 380-386.
18. Jørgensen M, Bjerregaard P, Borch-Johnsen K, Backer V, Becker U, Jørgensen T, et al. Diabetes and impaired glucose tolerance among the Inuit population of Greenland. Diabetes Care 2002; 25: 1766-1771.
19. Schwartz AV, Sellmeyer DE, Ensrud KE, Cauley JA, Tabor HK, Schreiner PJ, et al. Older women with diabetes have an increased risk of fracture: a prospective study. Journal of Clinical Endocrinology and Metabolism 2001; 86(1): 32-38.
20. Vanasse A, Courteau J, Cohen AA, Orzanoc MG, Drouin C. Rural-urban disparities in the management and health issues of chronic diseases in Quebec (Canada) in the early 2000s. Rural and Remote Health (Internet) 2010; 10: 1548. Available: www.rrh.org.au (Accessed 13 February 2017).
21. Lee R, Pearson M, Lyles KW, Jenkins PW, Colon-Emeric C. Geographic scope and accessibility of a centralized, electronic consult program for patients with recent fracture. Rural and Remote Health (Internet) 2016; 16: 3440. Available: www.rrh.org.au (Accessed 13 February 2017).
22. Jackson MNG, McCulloch BJ. Heart attack symptoms and decision-making: the case of older rural women. Rural and Remote Health (Internet) 2014; 14: 2560. Available: www.rrh.org.au (Accessed 13 February 2017).
23. Brown DS. Do leaves have to fall in their Autumn? A falls prevention strategy in action in the South East of South Australia. Rural and Remote Health (Internet) 2004; 4: 231. Available: www.rrh.org.au (Accessed 13 February 2017).
24. O'Neill TW, Felsenberg D, Varlow J, Cooper C, Kanis JA, Silman AJ. The prevalence of vertebral deformity in European men and women: the European Vertebral Osteoporosis Study. Journal of Bone and Mineral Research 1996; 11: 1010-1018.
25. Nayak S, Edwards DL, Saleh AA, Greenspan SL. Systematic review and meta-analysis of the performance of clinical risk assessment instruments for screening for osteoporosis or low bone density. Osteoporosis International 2015; 26: 1543-1554.
26. Rubin KH, Abrahamsen B, Friis-Holmberg T, Hjelmborg JVB, Bech M, Hermann AP, et al. Comparison of different screening tools (FRAX, OST, ORAI, OSIRIS, SCORE and age alone) to identify women with increased risk of fracture. A population-based prospective study. Bone 2013; 56: 16-22.
