Introduction
It is estimated 1.67 million women worldwide were diagnosed with breast cancer in 20121. In Australia, breast cancer is the most common cancer diagnosed amongst women (with the exception of keratinocyte cancers) and the second leading cause of cancer death2.
Inequalities in cancer outcomes continue to exist across the world with these inequalities observed at socioeconomic, racial and geographical levels3. In Australia, there are persistent survival disadvantages for those who live in rural areas compared to their urban counterparts. For example, 5-year relative survival for rural women diagnosed with breast cancer is about 2–5% lower in absolute terms compared with that of urban women2,4-8. While more recent analyses have shown survival has improved over time across all population groups, rural and disadvantaged women continue to experience poorer survival (after adjusting for age and disease stage at diagnosis)9.
Further, variations in the clinical management of breast cancer exist amongst rural and disadvantaged women. Rural and disadvantaged women with early breast cancer are more likely to undergo mastectomy than breast-conserving surgery (BCS), with these differentials remaining after adjustment for clinical factors10-12. Similar variations have been observed in other countries13-16. While rates of mastectomy for early breast cancer have reduced over time, rural women continue to undergo mastectomy more frequently than their urban counterparts17. A number of studies have also shown lower rates of sentinel node biopsy for rural women, with authors suggesting the results may reflect difficulties in obtaining radiopharmaceuticals required for this procedure as well as a lack of trained specialists18-20. While more recent population-based registry analysis showed an increase in the use of sentinel node biopsy overall, there remained no evidence of a reduction in the geographical disparity over time21.
Current evidenced-based guidelines for the management of early breast cancer state that, where clinically appropriate, women should be provided with information about mastectomy and BCS to assist them in making an informed choice22,23. While both procedures have equivalent survival, BCS is associated with reduced morbidity and better quality of life24. However, choice of mastectomy among women who are eligible for BCS appears dependent on a number of factors. Studies have shown these include clinical factors (stage, tumour size, grade, previous history of breast cancer or breast disease)25, as well as non-clinical factors such as age, socioeconomic status and geographical location26-28. Distance to closest radiation facility has also been identified as a barrier for rural women29-31.
While historically clinician and patient discussions regarding treatment options were uncommon, the shared decision-making model is increasingly being used, particularly where more than one treatment is available32. Factors that influence treatment decisions can be complex and individualistic. A key factor influencing treatment decisions is the perception of a need to do everything possible to reduce risk of the disease recurring and to afford the best chance of long-term survival. Covelli and colleagues33 found an overriding factor influencing treatment decisions was fear of recurrence, and a previous experience of cancer involving a family member or close friend. Access to health care has been proposed as an important factor in the treatment decision process. For example, while adjuvant radiotherapy is often used in combination with BCS, distance to the closest radiation service along with access to radiation oncologists have been reported as being a barrier for rural women in particular29-31.
The aim of this present study was to examine factors that influence treatment decisions of women with breast cancer, with a particular focus on identifying whether there are differences between rural (including regional, remote and very remote) and metropolitan women.
Methods
This study used a mixed-methods approach involving qualitative interviews followed by an online survey utilising Breast Cancer Network Australia’s (BCNA) membership database. BCNA is the peak national organisation for Australians affected by breast cancer, with over 120 000 members Australia-wide.
Qualitative interviews
Semi-structured interviews were conducted with nine women living in rural locations throughout Australia in September 2016 recruited via BCNA’s Review & Survey Group. A semi-structured interview guide was used to ensure relevant topics were discussed including treatment decision-making and factors that may have influenced this, information received/obtained at that time, satisfaction with treatment decisions and information and support received, along with adherence to treatment regimens.
Interviews were audio-recorded (with consent), transcribed and analysed using NVivo v11.3.1 (QSR International; https://www.qsrinternational.com/nvivo/nvivo-products). The thematic analysis approach was based on an interpretive phenomenological framework that aims to capture the lived experience of the participant and adopts a shared understanding on the part of the researcher and the participant. Emergent themes were identified by grouping similar concepts. Data saturation was achieved during analysis and new codes were not generated with later participants.
Online survey
The online survey was developed in consultation with BCNA. Survey questions were additionally shaped by the themes that emerged from the qualitative interviews. The survey was pilot-tested among a small sample of BCNA consumer representatives. The survey was specifically aimed at women diagnosed with early stage breast cancer more than 12 months but less than 5 years ago who had completed treatment (with the exception of ongoing hormone therapy), and those diagnosed with metastatic breast cancer more than 12 months but less than 3 years ago.
The survey included sociodemographics including age, postcode at residential location, Indigenous status, education and annual household income. Participants were asked to indicate the type of breast cancer they had been diagnosed with: ductal carcinoma in-situ (DCIS), lobular carcinoma in-situ (LCIS), early stage breast cancer, metastatic breast cancer or ‘don’t know what type of breast cancer’. Residential postcode was coded to major city, inner regional, outer regional, remote and very remote using the Australian Bureau of Statistics Australian Statistical Geographical Standards Remoteness Structure 2011.
Involvement in treatment decision was assessed by asking participants whether they were involved in deciding what treatment(s) they would have at time of their initial diagnosis. Participants were further asked who was involved in making treatment decisions. Women not involved in the treatment discussions were additionally asked the reasons why (Figure 1 provides response categories.) Using the Control Preference Scale from Degner and Sloan34, researchers asked participants how they would have preferred the treatment decision to be made with possible responses being:
- I was satisfied with how the decision was made.
- I would have preferred that the decision was made by my doctor(s) with little input from me.
- I would have preferred that my doctor(s) made the decision, but seriously considered my opinion.
- I would have preferred that my doctor and I made the decision together.
- I would have preferred to make the decision after seriously considering my doctor’s opinion.
- I would have preferred to have made the decision with little input from my doctor(s) with little input from my doctor(s).
Participants were additionally asked to identify to what degree various factors influenced their treatment decisions using a Likert-type scale (‘not at all’, ‘a little, ‘moderately’, ‘quite a bit’ or ‘a lot’ with the factors adapted from the work of Degner et al35.
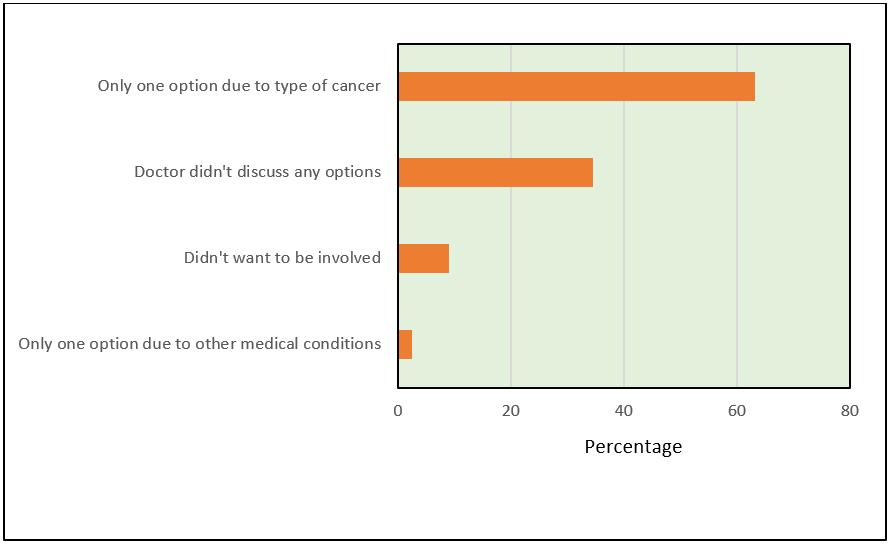 Figure 1: Most common reasons why women were not involved in treatment decision.
Figure 1: Most common reasons why women were not involved in treatment decision.
Online survey participants
The BCNA database contained 14 762 members who met the inclusion criteria (ie individuals with an email address who were diagnosed with early stage breast cancer between December 2011 and December 2017 or those diagnosed with metastatic breast cancer between December 2013 and December 2015). As there were significantly more members living in metropolitan compared to rural locations, the researchers elected to contact a random sample of 4000 metropolitan members and all 2311 rural members. The random sample of metropolitan members was representative of the whole group. Recruitment was supplemented through contact with BCNA member group facilitators (n=351), BCNA’s Facebook and their online network. Potential participants were contacted by email and provided with a brief summary of the aims of the study along with a website address they could access to complete the survey after first giving online consent. There were no significant differences across sociodemographics according to recruitment method.
Analyses
Descriptive analyses were used to examine frequency distributions across survey questions. The statistical significance of bivariate comparisons between various survey questions and residential location, age group and education was estimated using χ2 test. Analyses additionally included separate logistic regression models to examine factors independently associated with treatment decision-making. For the purposes of this analysis participants living in major city locations were classified as ‘metropolitan’ and those living in inner or outer regional, or remote and very remote locations were collapsed into the broader category of ‘rural’. However, when analysing factors associated with treatment decision making, we included three categories (metropolitan, inner regional and outer regional/remote/very remote). All online survey analyses were conducted using Stata v14.2 (Stata Corp, https://www.stata.com).
Ethics approval
Ethics approval for the study was obtained from the University of the Sunshine Coast (approval #A16/168).
Results
Description of participants
Nine rural women agreed to take part in the qualitative interviews (24% response rate). Mean age was 55 years with half having dependent children living at home and all participants, with the exception of one, with a current partner/husband. All interview participants had received surgery, seven radiotherapy and six chemotherapy. Most were diagnosed with early stage breast cancer and two with metastatic breast cancer.
In all, 6662 women were emailed regarding the online survey, with 1046 (15.7%) initially consenting to participate. Of those, 815 (77.9%) completed all survey items. Table 1 provides an overview of survey participants. Mean age was 57 years (range 29–83 years) and 1.4% identified as Aboriginal or Torres Strait Islander (hereafter referred to respectfully as ‘Indigenous’). Approximately 45% lived in a metropolitan location. Of those living in rural areas, just under one-third (31.9%) were from an inner regional area, 20.1% from outer regional areas and 3.2% (n=26) were from remote or very remote locations. Just over one-third were educated at an undergraduate or postgraduate level (39.0%) and 32.8% had a diploma or certificate. Education at an under- or postgraduate level was more common amongst metropolitan women (p=0.002). About one-third (31.8%) of all women reported an annual household income of less than $65,000 per annum, with this being more frequently observed for rural women (p<0.001). In relation to breast cancer diagnosis, the majority of women reported being diagnosed with early stage breast cancer (69.9%), about 1 in 5 reported a diagnosis of DCIS or LCIS and 5.8% had metastatic breast cancer with these proportions being similar amongst rural and metropolitan women. Nearly all women reported having some form of surgery (98.8%), with just under three-quarters reporting they had radiotherapy; 61.6% had chemotherapy and 73.7% had or were currently receiving hormone therapy. There were no significant differences observed according to residential location. Overall, just under 1 in 5 women (19.5%) indicated they had undergone breast reconstruction, with this being significantly more common amongst metropolitan compared to rural women (p=0.009).
Table 1: Description of 815 participants completing the online survey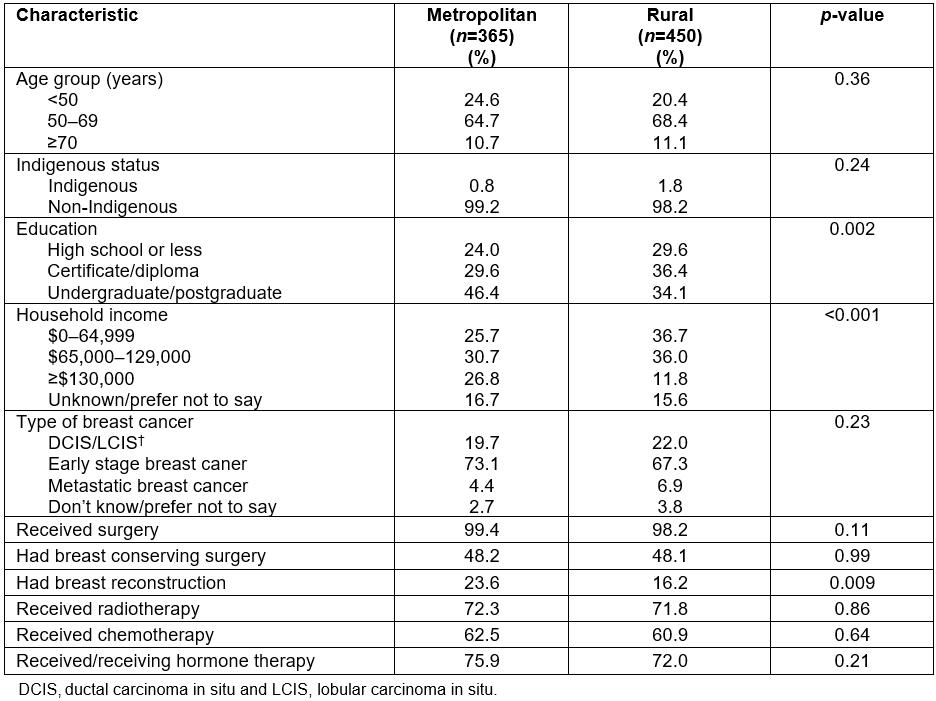
Involvement in decision treatment decision-making process
In qualitative interviews, most women concurred with the recommendations of their treating doctor(s) and did not see this experience as ‘making a decision’. However, two participants reported disagreeing with the recommended treatment options presented, and ‘making their own decision’ based on their career experience/knowledge and research.
In the online survey the majority of women (83.2%) indicated they had at least some involvement in deciding what treatment(s) they would have (Table 2). At a bivariate level, while no significant differences were observed according to residential location, fewer women educated at certificate/diploma level or high school or lower level indicated they were involved in the decision-making process (p=0.007). In multivariable logistic regression analysis, compared to women educated at an under- or postgraduate level, those whose education was high school or less, and those at a level of diploma/certificate, were both about 50% less likely to indicate they were involved in the treatment decision process (odds ratio (OR)=0.51, 95% confidence interval (CI)=0.30–0.86 and OR=0.48, 95%CI=0.29–0.78, respectively, p=0.008). When the analysis was restricted to those diagnosed with early breast cancer, the strength of the association between not being involved in the treatment decision-making process and lower levels of education was even more pronounced. Further, compared to women diagnosed with metastatic breast cancer, those with DCIS/LCIS or early breast cancer were significantly more likely to report being involved in the treatment decision-making process (OR=2.29, 95%CI=1.32–5.10 and OR=3.32, 95%CI=1.51–7.31, respectively, p=0.02) (Table 2).
Table 2: Factors associated with treatment decision-making amongst a sample of women with breast cancer in Australia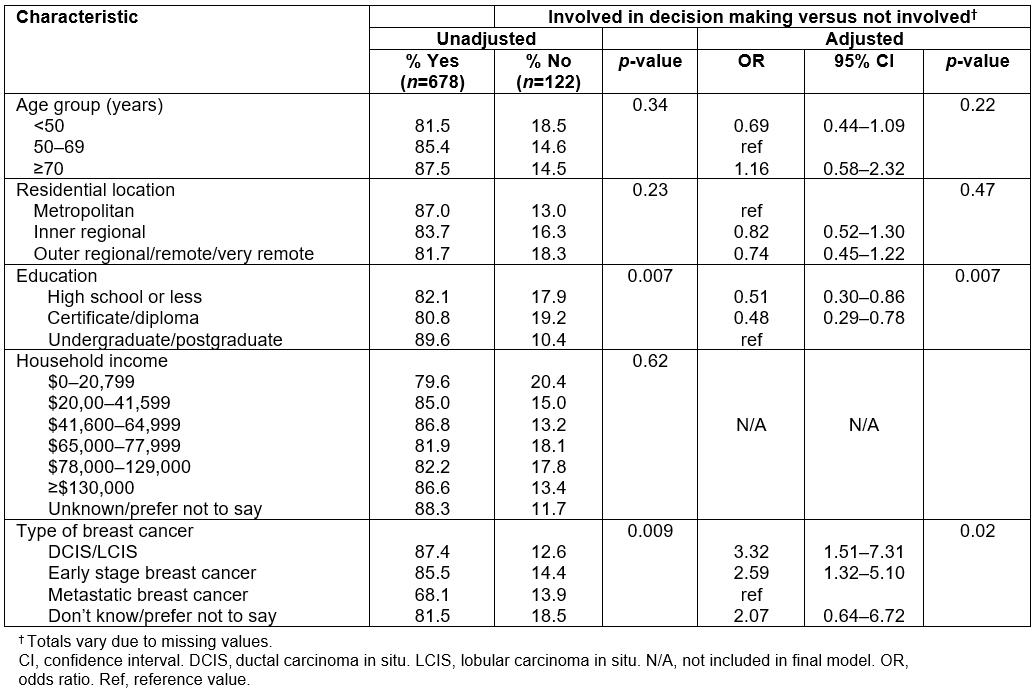
Who was involved in making treatment decisions?
Table 3 provides the frequency of responses when asking participating women who was involved in making the treatment decision (includes only those women who indicated some involvement in the decision-making process (n=678)). The most common response was that both the woman and her doctor(s) made the decision together (40.0%) with a further 25.0% of women indicating they made the decision after seriously considering their doctor’s opinion. The researchers found no significant differences in the frequency of responses according to residential location (p=0.13). This was similar when only women with DCIS/LCIS or early breast cancer were included. However, when researchers examined the frequency of responses for women with either early breast cancer or DCIS/LCIS who did not have BCS (n=315), a significantly higher proportion of rural compared to metropolitan women was found, indicating they decided on their treatment(s) after seriously considering their doctor’s opinion (37.3% and 21.5%, respectively) (p=0.01). No such pattern was found for women who had BCS (Table 3).
Table 3: Who was involved in making treatment decision(s) amongst metropolitan and rural women diagnosed with breast cancer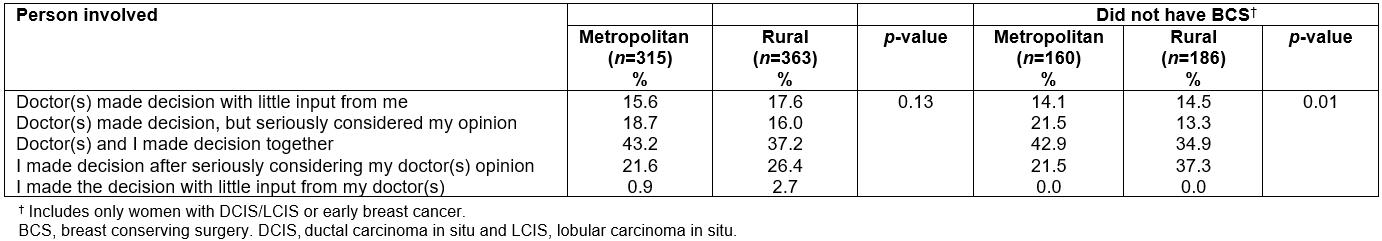
Why some women were not involved in treatment decisions
Approximately 15% of women indicated they were not involved in deciding what treatments they would have. The most common responses for why they were not involved was that they did not have options due to their type of breast cancer (63.1%) or that their doctor did not discuss any options with them (34.4%) (Fig1). Responses were similar for rural and metropolitan women and across age groups.
Factors influencing treatment decisions and options
Table 4 shows the component themes identified in the qualitative interviews. Many participants discussed wanting to do all treatment available in order to give themselves the best chance of surviving their breast cancer and reducing any risk of the disease recurring in the future. Some participants indicated that their experiences with cancer, either within the family or as a health professional, influenced their decision-making. Participants also identified the need to have treatment locally to avoid time away from family. This was particularly a concern for participants who were the main caregiver. One participant, rather than have treatment locally, elected to have treatment away from her community due to concerns about her own privacy.
Similarly, in the online survey, the vast majority of women indicated their treatment decision was strongly influenced by ‘minimising the chance the cancer will come back’ and ‘the need to do everything possible to get better’, with no significant differences in the frequency of responses according to residential location (Table 5). However, a significantly higher proportion of rural (42.8%), compared to metropolitan women (28.1%), indicated their treatment decision was influenced ‘quite a bit/a lot’ by the ‘location of treatment services and facilities’ (p<0.001). Similarly, nearly one in three rural, compared to one in five metropolitan women, said their decision was strongly influenced by the ‘financial costs of treatment’ (p=0.04). Having ‘reconstructive surgery as an option’ appeared to be of more importance to metropolitan women with 44.1% of them indicating this option strongly influenced their treatment decision compared to 27.7% of rural women (p<0.001). Overall, the researchers found about one in five women indicated their treatment decision was strongly influenced by the wishes of family/friends, with these results being similar for both metropolitan and rural women (Table 5).
Additionally, the researchers found some significant differences in responses for a number of factors across age groups (Table 5). For example, over 80% of women aged less than 70 years, and just under 60% (58.7%) of those aged ≥70 years, indicated their treatment decision was influenced ‘quite a bit/a lot’ by ‘wanting to have every possible treatment to get better’ (p<0.001). Similarly, for older women (≥70 years), having reconstructive surgery as an option did not appear to have influenced treatment decisions to the same degree than for those aged less than 50 years (14.6% and 48.5%, respectively, p<0.001). Further, about one in four women (28.3%) aged less than 50 years indicated their treatment decisions were strongly influenced by ‘the financial costs of treatment’, compared to only about 1 in 10 women aged ≥70+ years (9.1%, p<0.001). Body image appeared of more importance to younger women, with about one-third (31.3%) saying their decision was influenced ‘quite a bit/a lot’ by ‘how their body would look after treatment’, compared to 22.0% of women aged 50–69 years and 11.5% of those aged ≥70 years (p=0.005). About one in two women aged ≥70 years (46.3%) indicated their treatment decision was strongly influenced by ‘wanting to avoid having chemotherapy’ compared to 21.8% of those aged under 50 years and 30.6% for women aged 50–69 years (p=0.004). Just over half (51.5%) of women indicated their treatment decision was not at all influenced by the wishes of their family or friends, with these results being similar across age groups.
Table 4: Component themes influencing treatment decisions identified in qualitative interviews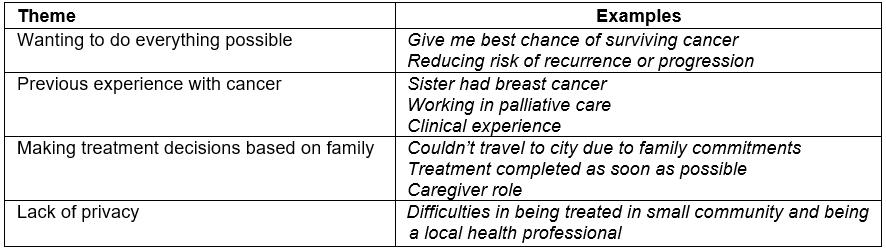
Table 5: Factors influencing treatment decisions amongst metropolitan and rural women with breast cancer in Australia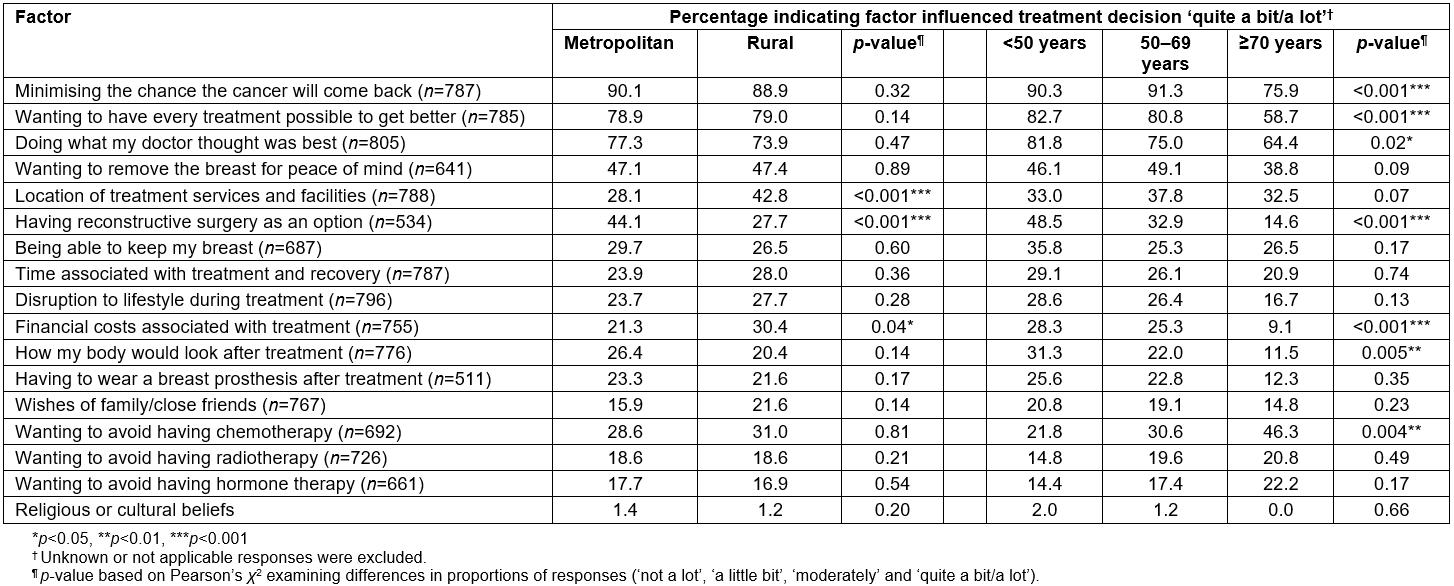
Were women satisfied with their treatment decision?
In the qualitative interviews, most participants reported being satisfied with their treatment decisions, with many stating they felt they had done everything they could to give themselves the best chance. For example, one participant said ‘So the decisions I made were to I suppose, survive and get myself the best options available’. Another participant reported doubts and anxiety about whether they should have had chemotherapy and a few also discussed fears about their cancer returning.
In the online survey the majority of women strongly agreed (58.0%) or agreed (31.9%) that they were satisfied with their treatment decision. Similarly, the majority strongly agreed (54.3%) or agreed (34.0%) that they felt they had made an informed choice; the vast majority (90.0%) agreed/strongly agreed they felt confident the treatment they received would achieve a good result for them. Responses were similar for metropolitan and rural women and across age groups (data not shown).
Discussion
This study examined the treatment decisions, and the factors influencing those decisions, of metropolitan and rural women in Australia treated for breast cancer.
It was found that the majority of women in this study were involved in the treatment decision process and this was similar for rural and metropolitan women. Only about 15% of women indicated no involvement in the process and the vast majority of those women said this was because they had no options due to the type of breast cancer they had. Overall, women did not necessarily see the experience of discussing treatments as ‘making a decision’, with most concurring with the recommendations of their doctor. This for a large part was driven by the need to do everything to live. While, rural and metropolitan women appeared to be equally involved in the decision-making process, the researchers did find significant differences in the degree of involvement according to education level. The finding that women educated at a high school level (compared to those with an undergraduate or postgraduate qualification) were significantly less likely to indicate involvement in treatment decisions is concerning. Similar findings have been reported elsewhere36. The reason for this is to a large extent unknown. Communicating what is often complex information is an important part of the consultation process, and needs at the time of diagnosis have been shown to be greatest37. It has been reported that women with lower levels of education do not necessarily want to be involved in discussing treatment options36, and women with a higher level of education prefer to be more involved and have more control over the decision-making process38. Taking into account health literacy when delivering information about breast cancer and treatment during consultations may help to empower women with lower levels of education and enable them to take part in the decision-making process.
In this study around 40% of women indicated they shared the decision-making with their doctor, with a further one-quarter saying they made the decision after seriously considering the opinion of their doctor, with this being similar amongst rural and metropolitan women. The present study did however find some differences in treatment decision-making according to residential location for women with early breast cancer who did not have BCS. Over one-third of rural women with early breast cancer who did not have BCS made their own treatment decision (after considering their doctor’s opinion) compared to only about one-quarter of metropolitan women. This finding was not replicated for women who had undergone BCS. While the present study could find no significant differences overall in the proportion or metropolitan and rural women who had received BCS, a number of studies report lower rates of BCS amongst rural women10,12. It is possible that rural women who did not have BCS were more likely to have made the treatment decision themselves due to factors such as the need to have follow-up radiotherapy, coupled with travel issues. Katz and colleagues reported more patient involvement in decision making when undergoing mastectomy with patient attitudes being significantly associated with surgical procedure39. As radiotherapy often follows BCS, travel distance to radiation services can be problematic for women living in rural locations26-28. Thus distance to closest radiation centre may mean that where rural women are given treatment options, it plays a more significant role in the decision-making process.
In the present study three factors strongly influenced the treatment decisions for both rural and metropolitan women equally. These were the need to do everything possible to get better, minimising the chances of recurrence and doing what the doctor thought best. These factors are in line with other research. A number of studies have shown the overriding factors influencing treatment decisions have been fear of the cancer returning or progressing, and having all possible treatments to survive the disease33,40. Further work has shown that women cite the recommendation of the surgeon as being an important influence in their treatment decision41.
The present study found significant differences in responses for three other factors amongst rural and metropolitan women. Rural women were more likely to indicate the location of treatment services and facilities had a strong influence over their treatment decision. Similar findings have been observed in a number of studies where travel distance, particularly for radiation therapy and other specialist cancer services, plays a significant role in treatment decisions29-31. While the present study did not find any differences in the proportion of metropolitan and rural women indicating their treatment decision was strongly influenced by wanting to avoid having radiotherapy, Australia is a geographically large country, where most health services are concentrated in major cities. Therefore, rural patients are more likely to rate issues with distance and access more highly than those living in metropolitan areas.
A significantly greater number of rural compared to metropolitan women indicated their treatment decisions were strongly influenced by the financial costs associated with treatment. It is likely the concern about costs includes the costs of travelling for treatment, being away from home and the financial burden associated with not working. Similar findings have been reported elsewhere where the need to stay in accommodation away from home and the potential costs of travelling have been reported as playing a significant role in deciding what treatments to undergo28. Further, Australian research has noted potential loss of income and travel costs are associated with patient choice of treatment42.While patient-assisted travel schemes are available for patients in Australia who reside more than 100 km from a treatment centre, these schemes vary from state to state in how they are accessed and managed, and reimbursement amounts. State-based cancer councils in Australia can also assist with transport and accommodation costs and, in some cases, can provide accommodation for patients and their carers travelling a long distance for treatment. While such schemes do not cover costs associated with loss of income, knowledge about the availability of the scheme at the time of treatment discussions may help patients when deciding on a course of treatment.
This study additionally found about double the number of metropolitan compared to rural women indicated their treatment decision was strongly influenced by having reconstructive surgery as an option. In Australia, rates of reconstructive surgery decline with increasing remoteness. A recent Queensland study found about 14% of women living in highly accessible areas had breast reconstruction compared to about 4% of those living in less accessible areas. While the disparity had reduced over time, after adjustment for a number of factors it remained that women in less accessible areas were about 70% less likely to have breast reconstruction17.
The present study additionally found some differences in factors that influence treatment decisions across age groups. By and large, younger women were more likely to place more emphasis on the need to reduce risk of recurrence and doing what the doctor thought best. A significantly higher proportion of younger compared to older (≥70 years) women, indicated their treatment decision was influenced by factors associated with body image. These factors included having reconstructive surgery as an option and how their body would look after treatment. Again, these results are in keeping with other findings that body image plays an important role in treatment decisions of younger women42,43.
Limitations
There are limitations to this study that need to be acknowledged. The main limitation was the use of the BCNA member database as the primary recruitment method. While BCNA represents the peak national organisation for those diagnosed with breast cancer, it is possible the participant sample was biased. The study examined participant interactions with BCNA in the 12 months prior to the survey being undertaken and found nearly three-quarters had at least some interaction with BCNA during that time (mostly though BCNA’s regular newsletters or through their website). Thus, it may be the case that participants were more engaged and have already had access to numerous information resources focusing on breast cancer in general and breast cancer treatments. That said, BCNA’s member network includes over 120 000 members and the sample of metropolitan and rural women who participated was reflective of the whole group. Further, the mean age of participants, the proportion living in metropolitan or rural areas, and levels of education were similar to those observed in a recent population-based study of women diagnosed with breast cancer44. Finally, the researchers elected to collapse participants from inner regional, outer regional, remote and very remote areas into one group (rural). While it could be argued that participants from inner regional areas have better access to treatment services and therefore they may more closely reflect the results of those living in major cities, it was actually found in this study that they were more reflective of those living in outer regional areas.
Conclusions
While the treatment decisions of the majority of women were strongly influenced by the need to do everything possible to get better and reduce the risk that their breast cancer would progress or recur, rural women indicated their decisions were influenced by additional factors. These primarily focused around access to treatment services and the financial costs associated with treatment. Addressing the issues around travel and costs for rural women with breast cancer should be prioritised. Additionally, the finding that women with lower levels of education were significantly less likely to be involved in the decision-making process highlights the need to develop new or tailor existing resources for this group in particular.
