Introduction
Indigenous populations frequently experience poorer health status and higher burden of disease, and these gradients in health outcomes have persisted over time and are consistent across nations1,2. Indigenous populations generally face higher rates of infectious, chronic and mental illnesses, in addition to higher mortality rates and shorter life expectancy compared to non-Indigenous populations globally3,4. Explanations for these health disparities are varied and complex, and reflect more generalized social gradients in health associated with socioeconomic inequity, as well as histories of colonization, family disruption and ethnic marginalization1,5. Despite persistent disparities in health for Indigenous peoples, research examining Indigenous health remains inequitably represented within the health literature5. Indeed, health research for Indigenous populations in lower income countries is scarce, particularly in Africa, home to approximately 14.2 million self-identified Indigenous people5. (Herein the African Commission on Human and Peoples’ Rights6 understanding of Indigenous peoples is followed, which focuses on ‘self-identification’, which requires ‘peoples [to] identify themselves as Indigenous, and as distinctly different from other groups within the state’ and recognizes the unique experiences of ‘marginalization’ and ‘discrimination’.) As such, there remains a limited evidence base for Indigenous public health policy, procedures and program development, particularly in Africa4,5.
Acute gastrointestinal illness (AGI) remains a concern for many Indigenous communities7-10. Vomiting and diarrheal diseases are responsible for a significant proportion of global morbidity and mortality, with particularly high burdens among poor and rural populations in Africa11,12. Studies examining the burden of AGI typically focus on children as a high-risk and vulnerable population13,14; childhood diarrhea remains one of the top five leading causes of death for children aged less than 5 years in Africa15, with a substantial proportion of disease concentrated in sub-Saharan Africa14. Furthermore, AGI is often associated with other infections, malnutrition or nutrient deficiencies, causing a substantial burden from co-morbidities in low-income regions16,17. While the burden of AGI is high in Africa, it is even higher among marginalized subpopulations4; recent research in Uganda, for example, identified higher AGI prevalence among Indigenous Batwa relative to global estimates8. The aim of the present research was to characterize the lived experience of AGI for an Indigenous population in sub-Saharan Africa, the Batwa, in Kanungu District, Uganda. The objectives were to (1) estimate and describe self-reported symptoms and severity of AGI, (2) characterize the multiple consequences of AGI for individuals and households and (3) describe the behavioral motivations and barriers impacting AGI preventative behaviors.
Methods
Study partners
The Batwa of central sub-Saharan Africa were traditionally forest-dwelling hunter-gatherers, with livelihoods primarily based on access to, and use of, forest resources. In the 1990s, the Batwa in south-western Uganda were dispossessed of their lands due to the creation of national conservation areas18. The Batwa were required to adapt to a new sedentary agrarian lifestyle, and they continue to report poor quantity and quality of land and resources, resulting from continued poverty, discrimination and marginalization18. The Batwa experience high rates of child and maternal mortality, exceeding Ugandan averages19,20 and previous research reported a higher prevalence of infectious diseases within some communities, compared to non-Indigenous populations4. A research partnership was formed in 2011 with the 10 formal Batwa settlements in Kanungu District, south-western Uganda. In 2012, the Batwa in Kanungu District self-identified diarrheal and vomiting illnesses as a key health priority21 and a recent study reported a high incidence of self-reported AGI, particularly among children aged less than 3 years8.
Conceptual approach
The Health Belief Model (HBM) conceptualizes behaviors governing positive health actions as products of perceived susceptibility and severity of illness, perceived barriers and benefits of taking preventative action, and exposure to education, media and social cues. The HBM and a community-based participatory framework were utilized to understand Batwa experiences, choices, understandings and opportunities related to AGI outcomes and prevention22,23. This framework is premised on principles of equitable participation, collaboration, transdisciplinarity, social equity, capacity building and knowledge translation24. This research was conducted as part of the Indigenous Health and Adaptation to Climate Change (IHACC) project. In collaboration with community partners the authors have developed ethics protocols to guide research projects (https://www.ihacc.ca/ethics1). Importantly, the authors view the ethics protocol as an ongoing negotiation between themselves, the communities and partner organizations.
Study design
A mixed methods study design using quantitative and qualitative data was utilized to enable both ‘breadth and depth of understanding and corroboration’ in examining AGI among the Batwa in Kanungu District25. Mixed methods approaches are relevant for community-based research as they can increase credibility, improve the utility of the findings and present a diversity of views26. In this project, all data collected were analyzed concurrently for a more robust understanding of the lived experience of AGI, capturing both population level trends as well as individual and community stories.
Quantitative data collection and analysis
In January 2013, January 2014 and April 2014, cross-sectional burden of illness (BOI) surveys were administered in all 10 of the formal Batwa settlements in Kanungu District, Uganda. The surveys attempted to capture a census of all the Batwa who were present in the settlements at the time of each survey. The questionnaires were carried out by trained research assistants from Kanungu District in the local language (Rukiga). Every individual was encouraged to complete an individual questionnaire and parents/guardians acted as proxy respondents for minors aged less than 12 years. Household level questionnaires were completed by an identified household head aged more than 18 years. Informed oral consent was obtained for each individual and for each new questionnaire (for minors aged less than 18 years, consent was also obtained from the parent/guardian first). The questionnaire design was based on previous international studies on AGI as well as previous qualitative pilot work among the communities for contextually appropriate questions8,27,28.
A case of AGI was defined as three or more loose stools and/or any vomiting in a 24-hour period in the 14 days prior to survey administration following pilot research and consultation with local partners regarding an appropriate, feasible and reliable period for recall. Individuals were excluded if they reported their symptoms were due to any diagnosed chronic condition, alcohol/drug/medication use or pregnancy29. Individual-level questionnaires gathered information on demographics and self-reported AGI symptoms and severity, while household-level questionnaires gathered information on environmental exposures, and water and sanitation infrastructure. Data were collected by hand on paper surveys, then entered manually into a Microsoft Access database.
In January 2013, January 2014 and April 2014, a total of 583 (response rate 99%), 569 (response rate 96%) and 540 (response rate 95%) individuals participated in the survey, respectively. Controlling for repeated measures on individuals, descriptive statistics on the severity of AGI symptoms for cases were calculated over the study period and stratified by age and gender. Measures of statistical difference using Mann–Whitney Wilcoxon for non-parametric data and two-sample t-tests for parametric data were conducted to examine differences in AGI severity by subgroup. The data were analyzed using Stata v13 (Stata Corp.; http://www.stata.com).
Qualitative data collection and analysis
Exploratory qualitative data were collected via key informant interviews and focus group discussions. Participants were invited to participate and informed consent was obtained by either an oral script or a written document as appropriate. Open-ended, flexible, semi-structured interview guides were pre-tested for content and context by local researchers and collaborators and addressed topics on the experience of AGI, the consequences of illness, and the barriers and benefits in taking preventative measures. Participants were encouraged to skip or refuse to answer any questions or topics they did not want to discuss, and were able to end the interview or leave the focus group at any time without penalty. All interviews were audio-recorded with permission, translated by research assistants when necessary30, and transcribed verbatim. Translations were reviewed and checked by Fortunate Twebaze, a Ugandan research team member. Furthermore, the researchers carried out daily debriefing sessions to discuss key findings and to identify and refine new questions. All recordings were stored on secured and encrypted laptops and deleted after transcriptions were completed as per agreements with the communities and participants.
Key informant interviews: Key informants (n=11), at the local, regional and national levels, were purposively recruited based on their knowledge and expertise in Batwa health in Kanungu District, and included individuals from public and private health facilities, government and local and national NGOs working with the Batwa. The interviews were conducted in English by Patterson, Clark and Twebaze, at a place of convenience for the interviewee (eg local restaurant or place of work). Average duration of key informant interviews was 27 (range 20–38) minutes with 272 total interview minutes.
Batwa focus group discussions: Ten Batwa focus group discussions (n=63 participants) were completed in 5 of the 10 formal Batwa settlements in Kanungu District. These five communities were purposively chosen based on their geographic dispersion, and varied micro-environments within Kanungu District. Following a meeting with the village chairperson and introduction to the research, all participants aged more than 18 years were invited to form two gender-stratified groups; these groups ranged in size from 3 to 11 participants based on community size and participant interest. All Batwa group discussions were fully facilitated in Rukiga (the local language) by Twebaze. Average duration of Batwa group discussions was 34 (range 24–42) minutes, with 342 total interview minutes.
Qualitative analysis: Following a phenomenological approach23, thematic content analysis31 was conducted to explore the lived experience of AGI for Batwa. Reflective memos were created throughout the data collection, recording and analysis process by authors Patterson, Clark and Twebaze32. Codes to identify themes in the responses were created by authors Patterson, Harper and Twebaze through both theory-driven (deductive) and data-driven (inductive) techniques33. In a process of triangulation, the qualitative key informant and focus group interview data were compared with quantitative survey responses in an effort to find convergence and similarities, as well as divergence in themes across multiple sources of data34. During the analysis, data were compared within and across respondent, gender and community groups31.
Ethics approval
Research Ethics Boards at McGill University and the University of Guelph granted ethics approval for the project (REB File #461–0414).
Results
Description of illness
Batwa focus group participants described that they get ‘affected all the time’ by AGI. In the BOI surveys, 31 survey participants were defined as a case of AGI in January 2013, 29 in January 2014 and 12 in April 2014. Those self-reporting AGI primarily experienced diarrheal symptoms (79%; 95% confidence interval (CI) 68–87%), while half experienced vomiting (50%; 95%CI 38–61%). Focus group responses reflected BOI survey statistics: focus group participants perceived episodes of AGI to last a ‘long time’, ‘even the length of a week’, with ‘little relief’ from symptoms. Similarly, for all Batwa AGI cases aggregated from the BOI surveys (2013–2014), episodes of vomiting lasted on average 3.0 (95%CI 2.1–3.9) days with 2.6 (95%CI 2.1–3.1) average maximum vomiting episodes in a 24-hour period. Episodes of diarrhea were longer in duration than vomiting, lasting on average 3.6 (95%CI 2.8–4.3) days with 4.3 (CI 3.9–4.8) loose stools on the worst day (Table 1). The most common reported symptoms from BOI survey cases were stomach ache, nausea, chills and fever, and focus group participants detailed that blood was sometimes in loose stools. Similar to adults, children aged less than 5 years experienced an average duration of 3.6 (95%CI 2.3–4.9) days of diarrheal symptoms, and an average of 4.7 (95%CI 3.7–5.7) loose stools on the worst day (Table 1). Focus group participants expressed concern over the possible health impacts of AGI, particularly for young children; one male Batwa explained ‘sometimes if the children are not treated earlier, they get malnourished and the illness gets so bad’, and key informants agreed that ‘persistent’ AGI symptoms were a concern for potential underdevelopment. From the BOI surveys, women and men did not significantly differ in duration of diarrheal symptoms or maximum loose stools (p>0.05); however, women on average experienced significantly longer duration of vomiting episodes than men, with 3.8 (95%CI 1.9–5.8) days in duration compared to 2.4 (95%CI 1.7–3.2) days in duration (p=0.02) (Table 1). Dehydration was another concern expressed by many focus group participants and key informants, and this concern included fears of death. One Batwa Elder in a focus group shared: ‘it [AGI] is so common because I have experienced it and it almost killed me’; other common words to describe the illness from interviewees included ‘dangerous’, ‘killer disease’ and ‘deadly’. Key informants in the Bwindi area shared this concern, expressing that vomiting and diarrheal symptoms are some of the most ‘common’ reasons to seek hospitalization.
Table 1: Duration and magnitude of primary symptoms aggregated from acute gastrointestinal illness cases in 10 Batwa communities (January 2013, n=31; January 2014, n =29; and April 2014, n=12; total n=72)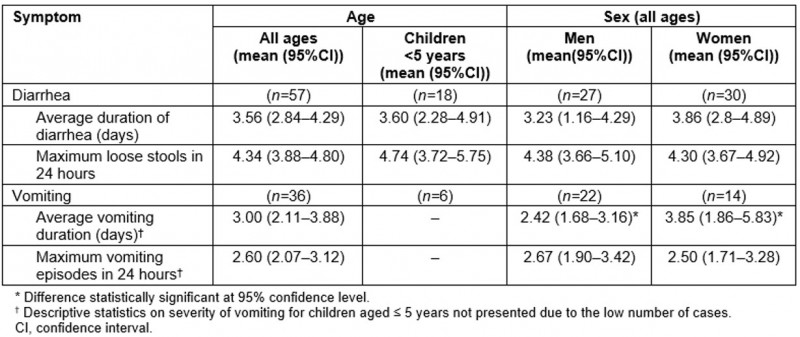
Consequences of illness
Episodes of AGI interrupted Batwa daily lives: 61% (95%CI 54–68) of cases in the BOI surveys reported missing their usual daily activities. Episodes of AGI were described by key informants and focus group participants as ‘unsettling’, ‘disruptive’, ‘weakening’ and ‘affecting’, to the individual as well as to the household. Alongside individual symptom-based health impacts, key informants and focus group participants identified economic, emotional and nutritional consequences for the household. For instance, one female focus group participant explained that when she has AGI she ‘becomes so weak and lose[s] energy’ and ‘thus lack[s] enough money to keep running the family’. Additionally, if a female adult had AGI, she may be incapable of performing household chores such as washing, cleaning, fetching water or cooking – activities traditionally performed by women in these communities – with consequences for health and safety of other household members. A Batwa woman in a focus group explained:
[Households] always face difficulties when women fall sick with the illness because [women] are the ones who run all the activities at home, look for food, cook, fetch water, which men cannot do.
If a child or adolescent experiences AGI, focus group participants reported that the affected individual would miss school, which could result in ‘poor performance’ in their education. Moreover, many Batwa women interviewees agreed that when their children had AGI, ‘it caused them much stress’ and resulted in a disruption of daily activities.
AGI risk and prevention
Environmental exposures: The baseline January 2013 household-level survey (n=131 households) indicated that 61% (95%CI 52–69) of Batwa households did not have access to hand washing facilities; moreover 74% (95%CI 67–82) lacked soap. A key informant said that most Batwa do not have access to ‘safe water’ and only 34% (95%CI 26–42) of Batwa households reported boiling their drinking water. Direct or indirect exposure to animals and animal feces was identified as contaminating the Batwa’s food and water sources by focus group participants; one male focus group participant said ‘there is always contamination of the running water from animals and fecal materials’ and a common observation was that fecal materials contaminated foods such as ‘mangoes and then children end up picking the mangoes and eating them’. The BOI indicated that 22% (95%CI 15–29) of households own at least one animal, and that in 12% (95%CI 8–19) of households animals sleep inside the main house. Garbage and leftover or partially eaten food were identified by key informants as high-risk exposures found around the private compounds and public spaces, particularly for young children who ‘at that age are moving around and picking everything up and eating it’.
AGI prevention behaviors: The Health Belief Model (HBM) conceptual framework was used to critically evaluate and explain the behaviors of the Batwa in regards to preventative actions in reducing the risk of AGI. Key informants and focus group participants articulated health-seeking behavior for AGI in accordance with the three primary constructs of the HBM (Fig1): a perceived threat of illness comprising perceptions of severity and susceptibility, perceived barriers and benefits from taking action, and cues to action (ie impactful life events, exposure to educational programs or media, and/or social norms)35.
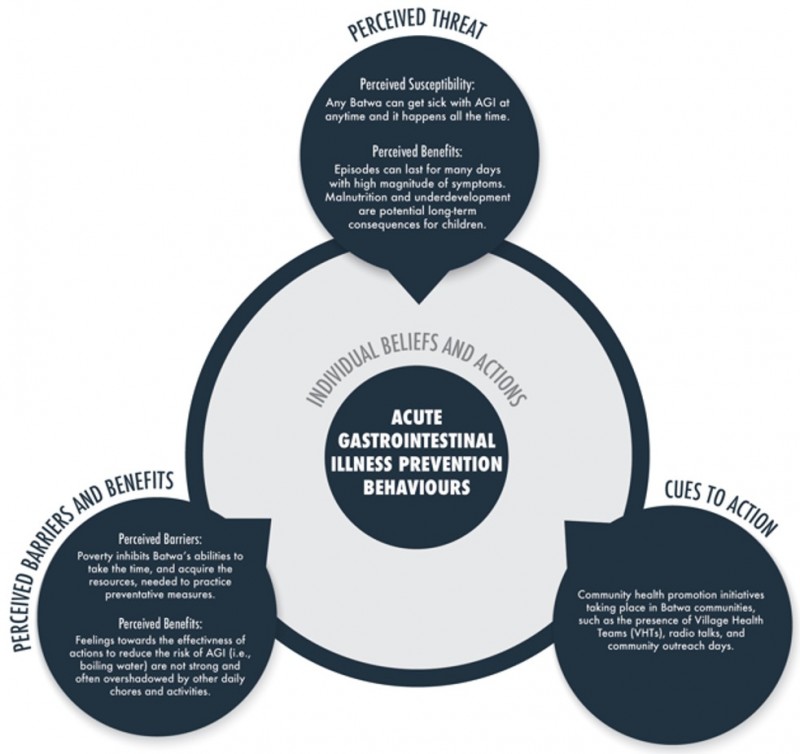 Figure 1: Overview of acute gastrointestinal illness prevention behaviors discussed by key informants and focus group participants.
Figure 1: Overview of acute gastrointestinal illness prevention behaviors discussed by key informants and focus group participants.
Perceived threat of illness Focus group participants expressed a perceived severity of illness by articulating that if one gets AGI they ‘become badly off’, ‘become so weak’, and ‘if it gets worse you can die’. Acute episodes could also last a long time, even ‘the length of the week’, and lead to more serious long-term health problems such as ‘malnourishment’ and ‘underdevelopment’. Batwa also perceived that everyone was susceptible to AGI, ‘because it happens all of the time’ in the communities. Many focus group participants believed that they were susceptible to AGI symptoms, yet they did not report many well-known AGI environmental risk factors as being the cause of their AGI symptoms, such as non-boiled water. However, eating ‘too much’, ‘too varied’, or ‘raw’ and ‘undercooked foods’ were often perceived as a causal link to AGI symptoms. Key informants speculated that the Batwa’s lack of perceived threat to many AGI environmental risk factors could be from the Batwa’s recent transition to sedentary communities from the forest environments, where their sanitation and hygiene behavioral norms were previously adapted to semi-nomadic, hunter-gatherer lifestyles. A key informant explained:
In the forest Batwa would take water not boiled, but in the forest contamination levels are very low compared to what they are now, and it is hard to perceive that there is a difference between water in the forest and water in the stream.
Perceived barriers and benefits For the Batwa, perceived barriers and benefits of preventing AGI were critical to the current behavioral challenges in reducing AGI magnitude and severity in the communities. Particularly in the realm of safe water consumption practices, boiling water was perceived by many focus group participants to be ineffective in improving water quality; community members explained that boiled water ‘does not taste good’, ‘makes us feel sick’ and ‘does not quench our thirst’, resulting in a lack of a perceived benefit from this precautionary measure. Focus group participants stressed that knowledge of AGI etiology and prevention are overshadowed by the lack of – and limited access to – adequate resources and infrastructure to reduce the risk of AGI; participants noted that ‘looking for food that we know bring up better health [is not possible] because we are poor we can’t afford that – children thus have to eat whatever food they find even if it causes diarrhea and vomiting’. Focus group participants said that monetary constraints made daily hand washing unlikely: ‘Soap is expensive; a piece of soap is something we cannot afford’. Furthermore, some focus group participants explained that due to long work days and general impoverishment their lifestyles did not permit them the time or energy to clean: ‘I would like to keep good hygiene by cleaning around my house but I can’t find the time because I wake up and I think about going to work for money’.
Cues to action Key informants described a number of health initiatives in all 10 Batwa communities that aimed to ‘reduce sanitation and hygiene related illness through education of families, and community members’. Key informants said the local hospital and health facilities have outreach programs and conduct weekly ‘health education on the radio’. Furthermore, each community has a village health promoter, whose job is to educate other community members on disease prevention. Key informants reported that health promotion programs were a key strategy used to combat environmental and behavioral risks associated with diarrheal and vomiting illnesses. Community health workers often spoke of myths and people’s perceptions interchangeably: ‘We try to address the social factors around diarrheal disease, addressing myths, people’s perceptions, what they think’. However, there was no consensus on the success of educational promotion alone to alter health-seeking behaviors. Focus group participants agreed that sanitation in some settlements had ‘greatly improved’ in recent years, and key informants attributed this to ‘continuous follow-up’; however, there was also a general consensus among key informants that there was still ‘a lot of work to be done’. In particular, the focus group discussions indicated that educational health promotion did not sufficiently translate into prevention of AGI: ‘Even when we have health education and they tell us about the preventive measures to some diseases, it ends with the education and we do not put it into practice’.
Discussion
This study characterized the lived experience of AGI for Indigenous Batwa in south-western Uganda, which included the clinical, financial and psychological consequences of AGI for the individual as well as the affected household. In addition, this research reported on perceptions of common precautionary measures to mitigate AGI risk analyzed through a HBM framework. This research can inform contextually and culturally appropriate interventions for the Batwa locally, as well as Indigenous and vulnerable groups in low-resource contexts facing similar challenges of infectious diarrheal disease.
The duration of AGI symptoms reported by Batwa using a 14-day recall period surpasses comparable global estimates in low- and middle-income countries using a similar case definition and a 28/30-day recall period27,36-40 (Fig2). Compared to other Indigenous populations in developed countries with similar incidence of AGI, the Batwa’s estimated duration of symptoms was almost double that of Indigenous Inuit in the Canadian North10(Fig2). This differential experience of AGI reflects substantial morbidity from diarrheal and vomiting illnesses in sub-Saharan Africa and for marginalized populations41,42. The average duration of diarrheal symptoms for Batwa children aged less than 5 years was slightly lower than estimates from a review of children in low- and middle-income countries43. However, similar to other studies examining self-reported illness, by their nature, these estimates may result in over- or under-estimates of AGI severity due to potential recall biases. Furthermore, this study did not test for specific pathogens and warrants further research in these communities to understand what pathogens might be causing the longer duration of AGI symptoms.
Trends of high susceptibility and high severity of AGI for Batwa children are consistent with other global studies on the burden of diarrheal and vomiting illness in developing regions14,41,44. Batwa children may be at a heightened risk of more frequent and severe AGI symptoms due to persistent reported food insecurity in these communities21. Acute gastrointestinal illness in itself can also be a risk factor that exacerbates malnourishment45,46, and this infectious-chronic illness cycle was articulated by focus group participants as a major concern for children in long-term illness risk reduction. Thus, the high severity of symptoms identified for Batwa children emphasized the need for longer term monitoring and nutritional rehabilitation of all children with diarrheal disease.
Key informant and focus group participants emphasized that negative consequences of AGI extended beyond individual symptoms and included indirect consequences for the household. Negative economic consequences of AGI for Batwa were consistent with several other studies in low-resource contexts that highlighted severe income shocks to households were associated with acute infectious diseases47-49. Key informants reported, and other reports confirm, that the majority of Batwa in these communities had minimal monetary or asset-based savings and live on a day-to-day income basis18,19. Similar to other research conducted in low-income settings, AGI cases for Batwa were reported as major sources of income shocks, putting household nutritional and other health needs at risk48-50. These results highlight that the burden of a single case of AGI may include a multitude of indirect consequences that can affect several individuals in a household.
In sub-Saharan Africa, and for the Batwa in this study specifically, when a household experiences AGI, time spent on caregiving is often disproportionately experienced by women49,51. Not only do women as caregivers face psychological and physical stress when household members are sick, but the results suggest – and other studies confirm – that women’s health status, livelihood capabilities and education have a significant impact on the health of the household50,52,53. The results indicate that the financial and nutritional consequences of episodes of AGI for Batwa families were greatest when women were either ill with AGI or had to attend to an ill child. Women’s impact on household health is confirmed by past studies that found maternal food production and parasitic infection were associated with the nutritional status of children50.
The HBM was identified through data-driven inductive techniques54 as a good fit for understanding the Batwa’s behaviors surrounding AGI prevention. However, in this assessment of AGI prevention behaviors, only measures promoted locally were assessed (cooking meat, person-to-person contact and so on were not examined). Within the Batwa communities, cues to action in the form of health education outreach were reported as widely used strategies to address myths and misconceptions to trigger behavioral change. Addressing health misconceptions is challenging, especially in rural areas; utilizing engaging communication strategies such as videos, drama, testimonials can be successful in tackling misinformation55-58. Similar to other studies that found maternal education significantly increased household uptake of preventative measures (eg mosquito nets, oral rehydration solutions59,60), Batwa women’s roles as caregiver and provider for children suggests that radio broadcasts and community health campaigns aimed at increasing good sanitation, hygiene and water-handling practices might be effectively directed towards Batwa women. Health education and communication are commonly based around a knowledge deficit framework61; however, more recent research suggests that perceived barriers related to socioeconomic and resource availability factors may be more important determinants of modifying behaviors towards prevention of diarrhea62. Indeed, in Batwa communities and in other resource-poor settings, daily monetary and livelihood constraints were important factors influencing preventative activities to reduce individual risk of AGI62,63.
Previous research points to perceived severity and susceptibility as highly influential constructs related to local vaccine demand. As such, these constructs may have important implications for future distributions of this form of prevention measure that tries to combat vaccine-preventable diarrheal diseases within Batwa communities64. Future public health initiatives and educational advice given by health workers within these Batwa communities should be informed by local practices and contextual conditions to see the greatest gains in AGI reduction.
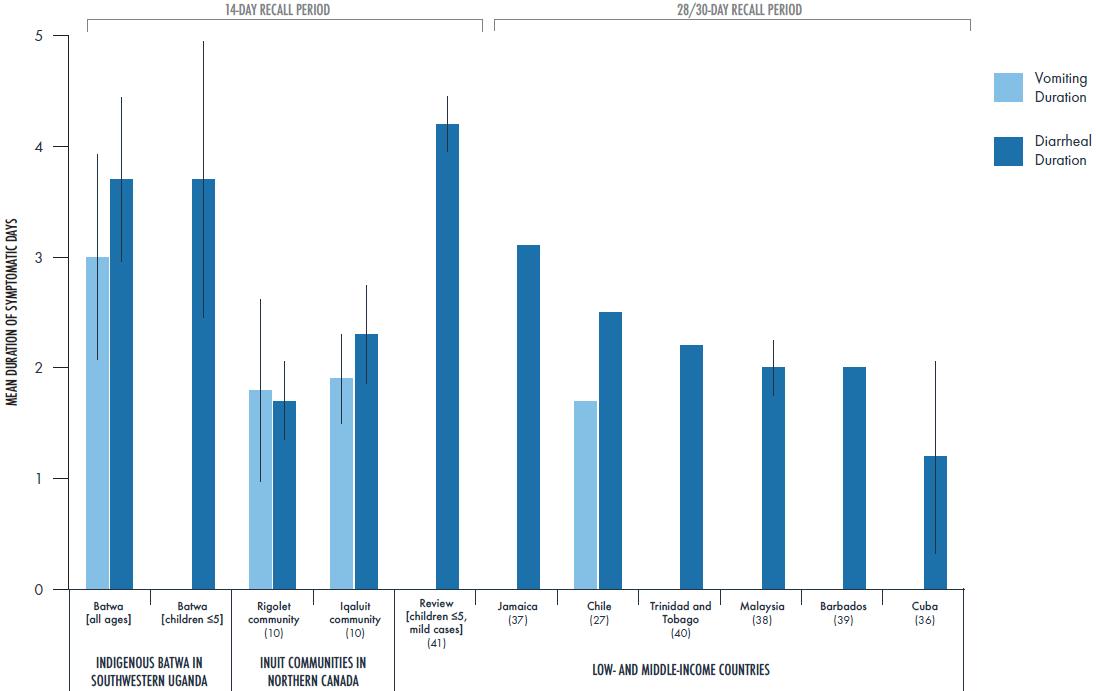 Figure 2: Comparison of duration of acute gastrointestinal symptoms among comparable self-reported studies in Indigenous communities8 that defined a case as three or more loose stools or any vomiting in a 24-hour period29 using a 14-day recall period.
Figure 2: Comparison of duration of acute gastrointestinal symptoms among comparable self-reported studies in Indigenous communities8 that defined a case as three or more loose stools or any vomiting in a 24-hour period29 using a 14-day recall period.
Conclusion
This research highlights the lived experience of AGI for Indigenous Batwa in south-western Uganda using a mixed methods approach to elucidate the severity, impacts and prevention behaviors related to AGI risk. Symptoms of AGI for Batwa were perceived as severe and reported to have negative economic and long-term health consequences for the affected household. These negative consequences were particularly pronounced when women were ill or attended to an ill child. Even in the midst of active efforts to reduce knowledge deficits in AGI transmission dynamics and prevention measures, perceived barriers and a lack of perceived benefits largely underscored behaviors surrounding AGI prevention. This research sheds light on the everyday challenges of AGI management and prevention for Batwa, which can provide important information for local public health officials in continued efforts to implement effective community-based interventions and monitoring.
Acknowledgements
This research is part of the IHACC project. We would like to thank all the Batwa communities and all community members for their invaluable contributions in this project. We could not have done this project without the help of our research assistants: Sabastian Twesigomwe, Evas Ninsiima and Grace Asaasire. We also thank our local partners: Ugandan Ministry of Health, Kanungu District Administration, Bwindi Community Hospital, Batwa Development Program. All of our figures were designed by Dr Alex Sawatsky, for which we thank her very much.



