Introduction
Cigarette smoking is the single most common cause of prevantable morbidity and mortality globally. It is estimated that by 2020, tobacco-related deaths will be the leading cause of deaths in developing countries1,2. While smoking is an important disease risk factor on its own, many adolsecents who smoke also have unhealthy lifestyle clusters such as smoking, alcohol use and illicit drug use3. Smoking may, therefore, be a marker of other harmful adolescents behaviours.
Research into adolescent cigarette smoking has mostly been conducted with current smoking status as the main outcome. However, the history of previous smoking is an important predictor of current or future smoking4. Also, studies into adolescent cigarette smoking have generally reported on the prevalence of smoking without consideration of relevant potential associations with socio-demographic factors. However, the identification of such factors can best inform the design and implementation of public health interventions aimed to reduce adolescent tobacco use. We report on a study aimed to estimate the prevalence of having smoked cigarettes, and its association with relevant socio-demographic characteristics.
Methods
The data were obtained in a cross-sectional school-based survey conducted under the Zambia Global Youth Tobacco Survey (GYTS) in 2002. The GYTS aimed to enroll students aged 13-15 years in order to estimate the prevalence of various forms of tobacco use and relevant socio-demographic variables. School grades likely to have students within the selected age range (13-15 years) were identified. For Zambia, these were students in grades 7 to 9. Identification of study participants was conducted through probability sampling using a multistage sample design. Schools were the primary sampling units, while classes were the secondary sampling units. All the students within the selected classes were eligible to participate regardless of their actual ages, therefore some students particiapted although they were outside the selected age range. The school response rate was 100% while the student response rate was 78.3%. A standardised questionnaire was self-completed anonymously by the students, and this it took between 30 and 40 min. The GYTS core questionnaire collected information on: cigarette smoking (current and ever): knowledge and attitudes of young people towards cigarette smoking; exposure to various media and advertisiments on tobacco; access to cigarettes; tobacco-related school curriculum; exposure to environmental tobacco smoke (ETS); and cessation of cigarette smoking. For the purpose of this study, however, only data related to a history of having smoked cigarettes and associated factors were analyzed. A comprehensive description of GYTS metholodlogy has been reported by Warren et al.5 and the Global Youth Tobacco Surveillance Collaborative Group6.
Study setting
Chongwe district is a rural area and spans for about 100 km from the outskirts of Lusaka, Zambia's capital city. Chongwe's population is approximately 148 000. Agriculture and charcoal production are the main sources of livelihoods, with the Chongwe district being the largest source of charcoal for Lusaka. The district is served by 15 health centers and a mission hospital; some of the health centers are located over 40 km from main roads7.
Data analysis
Data were analysed using SPSS v 14.0 (SPSS Inc; Chicago, IL, USA). It was aimed to assess the prevalence of having smoked cigarettes, stratified by sex. Bivariate logistic regression analyses were conducted to obtain an unadjusted odds ratio between having smoked and explanatory variables of interest (Model 1). Model 2 was multivariate analysis with the explanatory variable of interest as the main exposure controlling for the other variables.
Ethical considerations
Permission to conduct this study was obtained from the Zambian Ministry of Education. Parents and guardians were informed of the study. Parents who did not want their children to participate were asked to inform their children not to particiapte. All students were informed that they were free not to participate or not to answer any questions on the questionnaire. The questionnaire was completed in class time. Students who did not want to particiapte remained in class but could submit an uncompleted questionnaire, because excluding them from the class was potentially stigmatizing. To preserve indidividual confidentiality, the questionnaires were self-completed anonymously by the student. School teachers were not in the classrooms at the time of questionnaire completion.
Results
Distribution of having smoked cigarettes by socio-economic variables
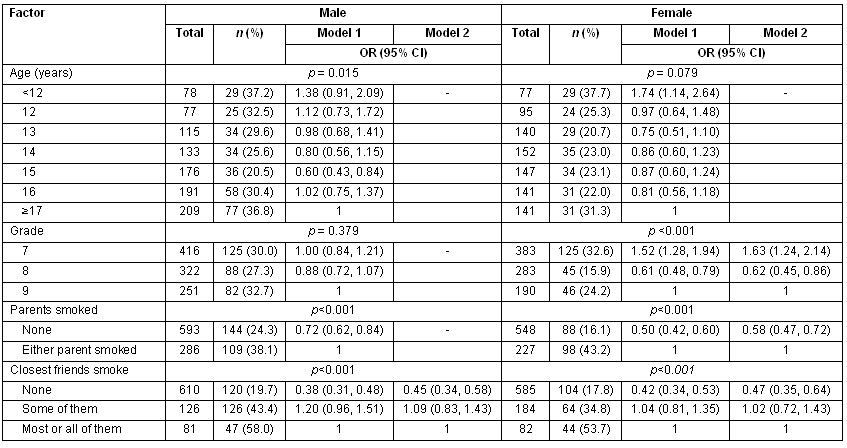
The prevalence of a history of having smoked cigarettes - even a single puff - was assessed. The results are presented (Table 1). Overall, the proportion of adolescents who reported having smoked was 27.0%. (26.8-27.2). Generally all age groups had a prevelance exceeding 20%. However, the extremes of the age group had the highest history of having smoked. A similar dsitribution was observed for class grades where the lowest and highest grades had higher prevalence than the middle grade. Adolescents whose parents were smokers or whose friends were smokers were themsleves more likely to have smoked.
Table 1: Demographic factors associated with having tried cigarette smoking among school going adolescents in Chongwe districts, Zambia
Attitudes towards smoking
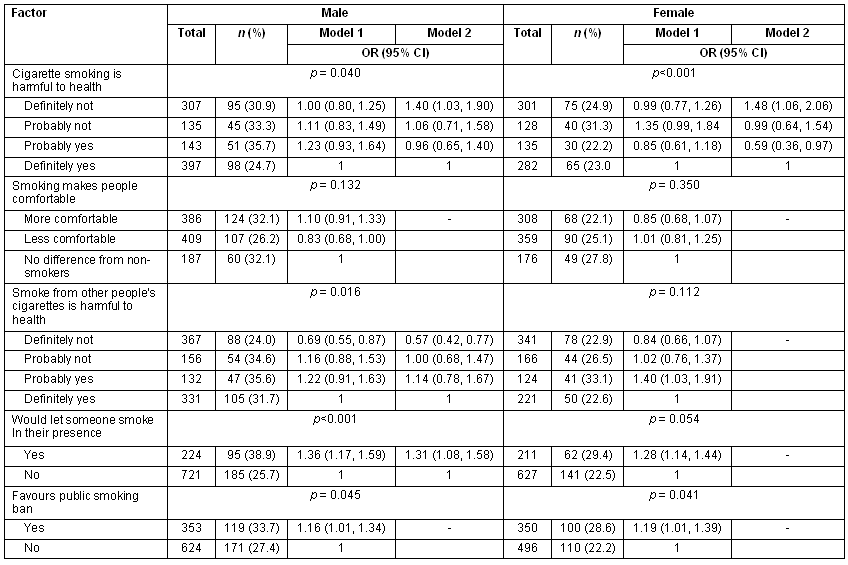
It was also aimed to assess attitudes towards smoking. In general, study participants had negative attitudes towards tobacco or smoking. There were, however, exceptions: for instance, the finding that favouring baning smoking in public and the perception that environmental exposure was harmful were associated with having smoked. The other results are presented (Table 2).
Table 2: Attitudes about smoking, and factors associated with having tried cigarette smoking among school going adolescents in Chongwe districts, Zambia
Tobacco advertisements and smoking
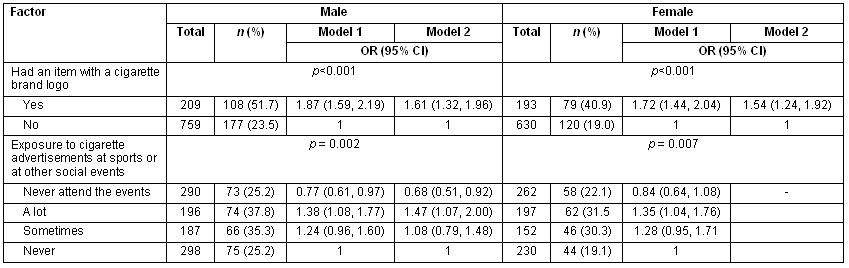
It was also aimed to determine if having smoked was associated with exposure to different forms of tobacco advertisements, owning a cigarette-logo branded item, and exposure to pro-tobacco advertisements at sports or social events. Exposure to both forms of advertisements were associated with having been a smoker. Detailed results are presented (Table 3).
Table 3: Associations between advertisements and having tried cigarette smoking among school-going adolescents in Chongwe districts, Zambia
Discussion
Our study found that the prevalence of having smoked cigarettes among in-school adolescents in Chongwe district, Zambia was 27%. This prevalence is lower than the 31% reported by Mpabulungi and Muula for Arua, rural Uganda8, and the reason for this is unknown. One possible explanation is that Arua's economy largely depends on tobacco farming, while Chongwe's does not, and so there may be a greater tolerance of tobacco use in Arua. Other unknown country-level factors may account for the differences between the two districts (eg in the Greek GYTS, Kyrlesi et al.9 reported a 32.2% prevalence of having smoked among 13-15 year-old adolescents). While the present study revealed no gender differences, another study of Zambian adults identified a higher prevalence among males10.
Our study identified that having a parent who smoked was associated with the adolescent having smoked. Ornelas et al.11, in their US study on parental influence on adolescent physical activity, found that parent-child communication and family cohesiveness were important influencing factors. Although that study found that parental monitoring was not a good predictor of adolescent physical activity, Arredondo et al.12 reported that parental monitoring was a good predictor of adolescent healthy eating and physical actovity. Factors involved in parents influencing adolescents' smoking may include permissiveness, the availability of cigarettes in the home and role modeling, and this should be investigated further.
Thomas et al.'s systematic review of family-based tobacco-control programs aimed at preventing adolescent smoking13, suggested that well-conducted programs were effective. School-based programs may also be effective14.
In our study, adolescents who perceived tobacco smoke to be harmful to their health were less likely to have smoked than those who were not concerned about the harmful effects of tobacco. This was also reported by Rodriguez et al15. This suggests an opportunity to change adolescents' beliefs about smoking, by using the media to reduce smoking behavior.
It has been established that tobacco promotion directed at adolescents enocourages the uptake and maintenance of smoking16,17. Gritz et al. reported that exposure to pro-tobacco advertisements was a strong predictor of being a smoker among US adolescents18. Our study supports this trend among adolescents in Chongwe.
Many previous studies have reported the association between adolescent smoking and having a friend who smokes. Bricker et al. found that smoking among adolescents' friends could influence both the initiation of smoking and maintenance of the habit19. Our results suggested that adolescents who had smoked were likely to have had friends who were smokers. Whether friends influenced the initiation of smoking, or whether adolescent smokers are more likely to choose other smokers as friends requires further study.
In our study, 15 year olds had the lowest prevalence of smoking (20.5%), compared with younger and older adolescents. This was an unexpected finding and the reasons should be explored further.
Limitations to the present study include that participants may have under-reported their smoking status, as was demonstrated in a Tanzanian adult-smoking study20. And, in our study, as the sample was drawn from schools, the findings may not be applicable to out of school adolescents.
Conclusions
Many adolescents in rural Chongwe, Zambia, have tried cigarette smoking, and associated factors among in-school adolescents in Chongwe have been identified. The identification of predictors of smoking should guide the design and implementation of programs aimed to prevent the initiation and maintenance of tobacco use among adolescents.
Acknowledgements
We are grateful to Mr Richard Zulu who coordinated the data collection. The data used in this study were obtained from the Global Youth Tobacco Survey (GYTS).
References
1. Mathers CD, Loncar D. Projections of global mortality budern from 2002 t0 2030. PLoS Medicine 2006; 3: e442. Available: http://medicine.plosjournals.org/archive/1549-1676/3/11/pdf/10.1371_journal.pmed.0030442-L.pdf (Accessed 7 January 2007).
2. Lopez AD, Mathers CD, Ezzati M, Jamison DT, Murray CJ. Global and regional burden of disease and risk factor, 2001: systematic analyses of population health data. Lancet 2006; 367: 1747-1757.
3. Faeh D, Viswanathan B, Chiolero A, Warren W, Bovet P. Clustering of smoking, alcohol drinking and cannabis use in adolescents in a rapidly developing country. BMC Public Health 2006; 6: 169.
4. Silva MA, Rivera IR, Carvalho AC, Guerra Ade H Jr.The prevalence of and variables associated with smoking in children and adolescents. Journal of Pediatrics (Rio de Janiero) 2006; 82: 365-370.
5. Warren CW, Riley L, Asma S, Eriksen MP, Green L, Blanton C et al. Tobacco use by youth: a surveillance report from the Global Youth Tobacco Survey Project. Bulletin of the World Health Organisation 2000; 78: 868-876.
6. Global Youth Surveillance Collaborative Group. Tobacco use among youth: a cross country comparison. Tobacco Control 2002; 11: 252-270.
7. Furth R. Zambia Performance-based incentives pilot study. Final report. United States Agency for International Development and the Government of Zambia. (Online) 2006. Avaiilable: http://www.qaproject.org/news/PDFs/ZambiaPerformancePilotStudyInitiatives.pdf (Accessed 20 February 2007).
8. Mpabulungi L, Muula AS. Tobacco use among high school students in a remote district of Arua, Uganda. Rural Remote Health 6: 609. (Online) 2006. Available: www.rrh.org.au (Accessed 22 August 2007).
9. Kyrlesi A, Soteriades ES, Warren CW, Kremastinou J, Papastergiou P, Jones NR et al. Tobacco use among students aged 13-15 yaers in Greece: the GYTS project. BMC Public Health 2007; 7: 3.
10. Pampel FC. Patterns of tobacco use in the early epidemic stages: Malawi and Zambia. American Journal of Health 2005; 95: 1009-1015.
11. Orenelas IJ, Perreira KM, Ayala GX. Parental influences on adolescent physical activity: a longitudinal study. International Journal of Behavioral Nutrition and Physical Activity 2007; 4: 3.
12. Arredondo EM, Elder JP, Ayala GX, Campbell N, Baquero B, Duerksen S. Is parenting style related to children's healthy eating and physical activity in Latino families. Health Education Research 2006; 21: 862-871.
13. Thomas RE, Baker P, Lorenzetti D. Family-based programmes for preventing smoking by children and adolescents. Cochrane Database of Systematic Reviews 2007, Issue 1. Art. no.: CD004493. DOI: 10.1002/14651858.CD004493.pub2.
14. Thomas R, Perera R. School-based programmes for preventing smoking. Cochrane Database of Systematic Reviews 2006, Issue 3. Art. no.: CD001293. DOI: 10.1002/14651858.CD001293.pub2.
15. Rodriguez D, Romer D, Audrain-McGovern J. Beliefs about the risks of smoking mediate the relationship between exposure to smoking and smoking. Psychosomatic Medicine 2007; 69:106-103.
18. Lovato C, Linn G, Stead LF, Best A. Impact of tobacco advertising and promotion on increasing adolescent smoking behaviours. Cochrane Database of Systematic Reviews 2003, Issue 3. Art. no.: CD003439. DOI: 10.1002/14651858.CD003439.
17. Sargent JD, Dalton M, Beach M, Bernhardt A, Heatherton T, Stevens M. Effect of cigarette promotions on smoking uptake among adolescents. Preventive Medicine 2000; 30: 320-327.
18. Gritz ER, Prokhorov AV, Hudmon KS, Mullin Jones M, Rosenblum C, Chang CC et al. Predictors of susceptibility of smoking and ever-smoking: longitudinal study of a triethnic sample of adolescents. Nicotine and Tobacco Research 2003; 5: 493-506.
19. Bricker JB, Peterson AV Jr, Andersen MR, Rajan KB, Leroux BG, Sarason IG. Childhood friends who smoke: do they influence adolescents to make smoking transitions? Addiction Behavior 2006; 31: 889-900.
20. Jagoe K, Edwards R, Mugusi F, Whiting D, Unwin N. Tobacco smoking in Tanzania, East Africa: population based smoking prevalence using expired alveolar carbon monoxide as a validation tool. Tobacco Control2002; 11: 210-304.
Correction: Changes were made to the title and text on 3 October 2007, as follow: (1) In the title, "Luangwa" was changed to "Chongwe"; (2) in the abstract introduction, "Luangwa" was changed to "Lusaka", and in the abstract conclusion, "Luangwa" was changed to "Chongwe"; (3) in the legends of Tables 1,2,& 3, "Chongwe and Luangwa" was changed to "Chongwe"; (4) in the Conclusions, "Luangwa" was changed to "Chongwe".
Abstract
Introduction: Cigarette smoking is a leading cause of morbidity and mortality globally. There are limited data on the prevalence of and factors associated with smoking among in-school adolescents in developing countries. Objectives: To estimate prevalence of those who have smoked cigarettes and to identify associated socio-demographic factors among adolescents in Chongwe district, Lusaka Province, Zambia.
Methods: Data used was from the Zambia Global Youth Tobacco Survey, which was conducted using standardized methodology among in-school adolescents in 2002. Data were analyzed to assess if selected socio-demographic variables were associated with having smoked cigarettes. Bivariate and multivariate logistic regression analysis was used to assess the associations.
Results: A history of having smoked cigarettes ranged from 20.5% among 15 year olds to 37.2% among males younger than 12 years old. In females, 20.7% of 13 year olds and 37.7% of those less than 12 years old had smoked. Parental smoking, friends smoking, a lack of perception that smoking was harmful, and exposure to pro-tobacco advertisements were associated with having smoked cigarettes. Adolescents who had smoked cigarettes were more likely to allow others smoke in their presence.
Conclusions: Many adolescents in rural Chongwe, Zambia had tried cigarette smoking. The identification of predictors for smoking should guide the design and implementation of programs aimed to prevent initiation and maintenance of tobacco use among adolescents.
Key words: adolescent health, Chongwe, cigarette smoking, tobacco, Zambia.
You might also be interested in:
2016 - Access to chlamydia testing in remote and rural Scotland
2009 - Reducing the risk of pandemic influenza in Aboriginal communities



