Introduction
Growing centralization of cancer services either increases patient travel, or prevents or delays access to specialist services. Concentrating clinical expertise, rationalising access to specialist equipment and enhancing support services most likely does not benefit patients who fail to access treatment. Impact of travel on patients' experience is often underplayed. A literature review1 has highlighted that travel was perceived as a barrier to cancer treatment. Rural patients' travel costs are an important overall component of the costs of care2 but additional travel costs from centralisation are passed on to the patient3. In Scotland, services have been organised into 'managed clinical networks' (MCNs) with Health Boards aligning with a single tertiary oncology/radiotherapy (RT) referral centre, herein described as designated cancer centre (DRT). In consequence, population subgroups have been allocated to treatment centres that are not necessarily the closest to their home address. Finally, with fewer cancer centres and larger distances between them, the possibility of choice, a central tenet of the UK government's health policy is limited by travel considerations. Although the configuration of MCNs may be driven by efficiency savings, it has been argued that costs of health care can vary widely in different areas, with resource allocation not always reflecting clinical need4,5.
The aim of this study was to determine if there are differences in incidence rates and accessibility of care over 3 years with respect to travel times from a patient's home This study was reviewed and received approval from the Dumfries and Galloway Local Ethics Committee.
Methods
Study population and setting
This study covered the population of mainland Scotland, accounting for 5 million people, over a three-year period from January 2000 to December 2002. There were 76 406 cancer registrations and 175 071 hospital discharges for cancer patients. Information services of NHS National Services Scotland (ISD Scotland), sanctioned by their data privacy committee, undertook the analysis of all cancer data (excluding non-melanoma skin cancer).
Derivation of measurements
The incidence (cancer registration) rate for 2000-2002, as well as hospital discharge rates and mean bed days over 2001-2003 were examined. This one-year shift in the hospital discharge data set was necessary to allow time for completion of the cancer patient cohort under analysis. This is standard practice for such population cohort studies in the field of cancer.
Incidence data were based on the Scottish cancer registration system and extracted in July 2005. Age and sex specific population estimates for Scotland were made available to ISD by arrangement with the general register office for Scotland. Hospital discharge information was based on data extracted from the SMR1 (Scottish Morbidity Data) in-patient day case data set, considering only primary diagnosis (in this case cancer). Using the geographical information system 'Pro-territory' and 2001 census data, travel times (for a one-way journey) were calculated from all Scotland's major hospitals to the population centre of each 'census output area' (the smallest geographical unit for which census data are available). The population centres were then allocated to a travel band.
An analysis was then undertaken of patient travel by road, within four travel time bands, to three main destinations: (i) nearest major hospital; (ii) nearest RT centre hospital; and (iii) designated DRT hospital (Appendix I). Due to overlapping geographies 0.5% of all areas were not assigned a travel band. In these areas minimum travel times were manually assigned based on selecting the nearest output area and adding 5 min to travel time. A list of major hospitals and RTs is presented (Appendix I), while Figure 1 is a map with population densities and the RTs. A DRT is the hospital from which the relevant Health Board routinely commissions services. This designation is derived from historical or contractual considerations and may not be the closest RT geographically for individual patients.
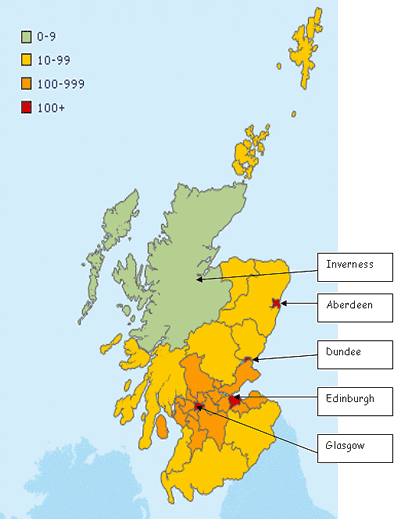
Figure 1: Map of Scotland showing population densities per square kilometre and location of oncology/radiotherapy treatment centres.
Outcomes
The main outcome was the potential (in population terms) and the actual (for the cancer patients studied) minimum travel time to the nearest healthcare facility in each category - nearest major hospital, nearest RT, and DRT respectively - for each census output area. Particular hospital category admission rates and bed days according to allocated travel time were also analysed.
Data analysis
The population and cancer patient numbers by travel time and hospital category are presented (Table 1). Percentage differences between hospital category allocation and potential or actual minimum travel time are highlighted.
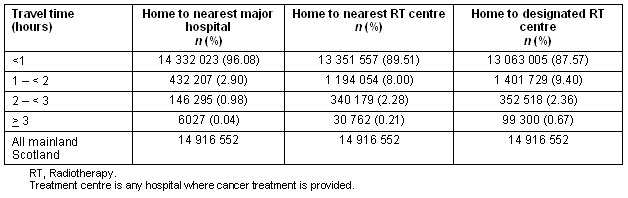
Table 1: Population bases stratified by minimum travel time to hospital; 2000-2002 (person years)
Hospital discharge data from mainland Scotland were selected using 2001 output areas converted to postcode using the postcode address file supplied to ISD by the Royal Mail. These discharge data were then allocated travel bands before generating population at risk to perform standard rate calculations. Finally, hospital discharge information was used for the bed day calculations by selecting a patient diagnosed with cancer within the time period, then extracting the information for the whole patient continuous inpatient stay (including any episodes of care within the same hospital stay pre- and post-cancer diagnosis). Thereafter, the bed day calculation was based on time from admission to the time of discharge over the whole continuous in-patient stay, irrespective of the time period considered.
The rate calculations used Scotland's mainland population by travel band as a base. Rates were standardised to the European Standard population and expressed per 100 000 person-years at risk.
Results
Minimum travel distance to hospital facility category
As Table 1 shows, patients travelling one hour or more to a major hospital account for approximately 4% of the study population. The percentage of the mainland population living between 2 and 3 hours away from major hospital is relatively small (<1%), while the percentage of those living more than 3 hours away is even smaller (0.04%).
In contrast, 10.49%, a two and a half times greater proportion of the population, would have to travel more than one hour for specialist oncology hospital care in order to be treated in their nearest RT facility. However, based on cancer centre designation, the potential minimum travel time could affect a population three times greater (12.43%).
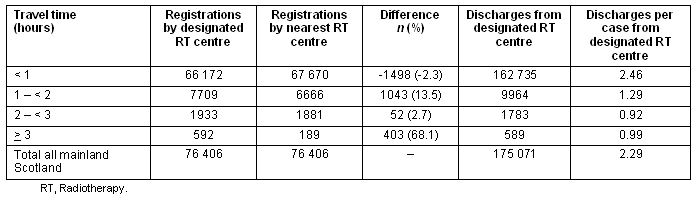
Such cancer centre designation makes little difference for potential patients living between 2 and 3 hours away (2.28 vs 2.36%). However, the less than 1% of the population living more than 3 hours away from their DRT centre are three times more likely to be referred to a geographically inappropriate cancer centre. For this population of 22 846, the DRT centre as defined by the Health Board is not their nearest RT. In practice this accounts for approximately 200 patients annually (592 over 3 years), as is shown (Table 2).
Table 2 also demonstrates the total numbers of individuals diagnosed with cancer who have been admitted to their DRT hospital in the time period 2001-2003. A considerable proportion (13.52%) of patients living between 1 and 2 hours away from their DRT centre could be treated nearer home. While this figure falls to 2.69% for patients living between 2 and 3 hours away, for approximately two-thirds of patients living more than 3 hours away from a cancer centre, the DRT centre is not their nearest.
Table 2: Total cancer registrations (2000-2002) in mainland Scotland (except non-melanoma skin cancer cases) and hospital discharges (2001-2003) according to minimum travel time from treatment centre
Likelihood of receiving specialist care
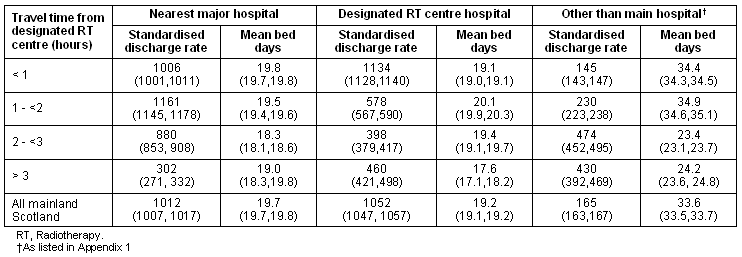
Patients living within one hour of a DRT centre are twice as likely to be admitted for inpatient specialist cancer care (average 2.5 admissions per annum over this time period) compared with all other groups (range 0.92 to 1.29).
Table 3 shows the duration of admission for registered cancer patients, grouped according to travel time from the DRT centre. The standardised discharge rate (admission rate) to the nearest major hospital for patients living between 2 and 3 hours and more than 3 hours away from the DRT centre revealed a significant marked and progressive reduction. However, the mean bed day figure was significantly less only for the '2-3 hour away' group. Patients living more than one hour away from a DRT centre had significantly reduced standardised discharge rate from a cancer centre. However, overall bed usage, as reflected in bed days, was only significantly lower for patients living at the greatest distance, with reduction in mean bed use of approximately 2.5 days overall. This reduction in discharge and bed usage in regard to the nearest major hospital and cancer centre was not reflected in any increase in mean bed days for other major and non-major hospital admissions (Table 3)
Table 3: Age and sex-standardised discharge rates and mean bed days per patient for all cancers from 2001 to 2003 (95% confidence intervals in brackets)
Discussion
Published data for cancer care in Scotland are rather conflicting or confusing. Comparisons of rural and urban populations have shown higher standardised cancer incidence and mortality rates for most cancers in urban populations. Although that suggests a lower overall risk for rural populations in Scotland, increasing distance from a cancer centre was associated with poorer survival6. This increase was attributed to later diagnosis and a lesser likelihood of diagnosis before death7. Our study followed a survey showing the burden of travel on rural cancer patients8. Patients receiving less specialised care are less likely to receive chemotherapy or radiotherapy9-12. An English study has suggested that delays in diagnosis are related to distance from primary care centre, rather than from hospital care13. Thus, for rural patients, later diagnosis and increased mortality is also associated with a lower hospital admission rate to oncology units.
Main findings
Almost 4% of the Scottish population lived more than one hour away from a general hospital; 10.5% of the population would have to travel more than one hour (2 hours return journey) to reach their nearest cancer centre. When referral followed cancer centre designation guidelines, this proportion increased to 12.5%. Thus 2% of the population who lived more than a two-hour round-trip journey from a cancer centre endured some unnecessary travel.
This article shows that travel time affects the type and frequency of hospital admission. Described as 'distance decay', the effect on admission rate is twofold after travelling for one hour. More than 13.5% of cancer patients living between 1 and 2 hours away from their designated radiotherapy centre could be treated at a cancer centre nearer home. The reason for these choices was not clear. The proportion falls for patients living between 2 and 3 hours away. More remote areas of Scotland (more than 3 hours travel) were disproportionately affected with a three to one likelihood of referral to a cancer centre that was, in geographical terms, inappropriate. Two out of 3 patients living more than 3 hours from a cancer centre had to travel further to a designated centre, rather than to the nearest. For those living in the heavily populated central belt, there may be a very small difference in travel time when choosing between available cancer centres. Such choice is a less likely explanation for those travelling more than 3 hours; for example in the South-west, travelling to Glasgow (the nearest DRT) takes 2 hours less than travelling to the Edinburgh DRT. For this group, the differential appears to reflect boundaries that suit organisations rather than the patient.
Travel time affects hospital care patterns. All patient groups more than one hour away from a cancer centre had reduced standardised discharge rates. The patients who had to travel more than 3 hours to their designated radiotherapy centre had a significantly lower overall bed usage of that facility. This reduction in admission rate also occurred in respect of the nearest major hospital access of this group of patients, and was not reflected in any increase in admissions to community (non-major) hospitals.
Comparison with existing data: Although reduced access to radiotherapy (25-75 miles/40-120 km) and chemotherapy is described11,12, we are not aware of comparable data on travel time for cancer centre care. This article shows distance decay applies to patients with cancer, as it does to those with ischaemic heart disease (20km)14, bronchopneumonia and chronic bronchitis15.
A recent study has suggested that differences in mortality are mostly affected by distance from a GP13. Other studies have suggested that delays in primary care contribute to the increased mortality7. This article shows that there are differences in the nature and duration of secondary and tertiary care offered that could potentially influence outcome. This may, however, simply be a marker for those patients who are also (because of living in a sparsely populated area) distanced from primary care. Such marked differences in the uptake of hospital services, both in type of hospital and admission rates, could compound or simply reflect differences arising from access to primary care. Pitchforth9 describes differences in chemotherapy uptake between general and oncology hospitals that could well be relevant to the different type of care offered to rural patients within both secondary and tertiary care. Some differential in access to specialist care is predictable, but a two or threefold effect is alarming and, in light of reduced survival for rural patients, requires further investigation. Those who argue for centralisation as the way to improve services should be concerned at the extent of the difference in service delivery revealed. A recent Cochrane Systematic Review 'supported the hypothesis that specialist outreach can improve access, outcomes and service use, especially when delivered as part of a multifaceted intervention'16.
Study limitations
This was a national study over a period of several years using robust and verifiable data. Cancer registration data in Scotland have a high level of accuracy compared with other registers, with serious discrepancies estimated to be under 3%17. A main weakness of this study was that it did not examine possible reasons for these findings, nor differences in site specific cancers. Travelling times quoted were for single journeys by private transport. Public transport or ambulance transfers can occasionally double journey time to 12 hours for a return journey. Local variation in road type, traffic volume and weather are also likely to affect travel times disproportionately.
Implications
Within the UK as a whole, the findings that the relatively short travel time of over an hour had an effect on access to specialist care constitutes a large potential risk. The cost of resolving access problems for those travelling furthest should not prove prohibitive, given the small numbers involved. For the 1% or so who had to travel more than 3 hours, there would need to be little re-alignment of specialist resources within tertiary care centres, if they all preferred to be treated nearer home. The inverse care law, whereby the most needy get the least resource18, appears to apply to the most remote patients who do not get care in the nearest RT and who received less inpatient hospital treatment overall.
The reduction in bed usage (mean bed days) for patients living more than 3 hours away is counter-intuitive, because distance from care should delay rather than expedite discharge. Conversely, if the most 'distant' patients were indeed admitted for what was an appropriate time to affect their assessment and treatment, the implication is that there is considerable unnecessary over-utilisation of resources by patients who travel less.
These figures demonstrate differing patterns of care between rural and urban cancer patients. It is known that there is also a difference in mortality between these patient groups. A number of associations exist between rural cancer patients and increased mortality. Further work needs to be done to determine whether the main effect on patient outcome lies at the community level (distance from a primary care centre) or at the secondary or tertiary care level.
Conclusion
This study provides evidence that the recognized increased cancer mortality in rural patients is indeed compounded by an increased travel burden. The reasons for this reduced access require further research. The reduced cancer centre bed days by patients living more than 3 hours away show inequitable treatment and raise the question of overutilization of services by patients who are required to travel less time. Finally, the finding of very remote patients travelling to tertiary hospitals that were not their nearest should be further investigated.
Acknowledgements
This study was conducted with the support of RARARI funding. The authors thank ISD for their assistance in supplying statistical data for this study.
References
1. Payne S, Jarrett N, Jeffs D. The impact of travel on cancer patients' experiences of treatment: a literature review. European Journal of Cancer Care 2000; 9(4): 197-203.
2. Yabroff K, Warren J, Knopf K, Davis W W, Brown ML. Estimating patient time costs associated with colorectal cancer care. Medical Care 2005; 43(7): 640-648.
3. Mungall I. Trend towards centralisation of hospital services, and its effect on access to care for rural and remote communities in the UK. Rural and Remote Health 5(2): 390. (Online) 2005. Available: www.rrh.org.au (Accessed 1 November 2008).
4. Asthana S, Gibson A. Rationing in response to NHS deficits: rural patients are likely to be affected most. BMJ 2005; 331(7530): 1472.
5. Asthana S, Gibson A, Moon G, Brigham P. Allocating resources for health and social care: the significance of rurality. Health Society of Care Community 2003; 11(6): 486-493.
6. Campbell NC, Elliott AM, Sharp L, Ritchie LD, Cassidy J, Little J. Rural factors and survival from cancer: analysis of Scottish cancer registrations. British Journal of Cancer 2000; 82(11):1863-1866.
7. Campbell NC, Elliott AM, Sharp L. Rural and urban differences in stage at diagnosis of colorectal and lung cancers. British Journal of Cancer 2001; 84(7): 910-914.
8. Baird A G, Donnelly C M, Miscampbell N T, Wemyss HD. Centralisation of cancer services in rural areas has disadvantages. BMJ 2000; 320: 717.
9. Pitchforth E, Russell E, Van der Pol M. Access to specialist cancer care: is it equitable? British Journal of Cancer 2002; 87(11): 1221-1226.
10. Fergusson RJ, Thomson CS, Brewster DH, Brown PH, Milroy R. Lung cancer: the importance of seeing a respiratory physician. European Respiratory Journal 2003; 21(4): 606-610.
11. Schroen AT, Brenin DR, Kelly MD, Knaus WA, Slingluff Jr CL. Impact of patient distance to radiation therapy on mastectomy use in early-stage breast cancer patients. Journal of Clinical Oncology 2005; 23(28): 7074-7080.
12. Punglia RS, Weeks JC, Neville BA, Earle CC. Effect of distance to radiation treatment facility on use of radiation therapy after mastectomy in elderly women. International Journal of Radiation, Oncology, Biology, Physiology 2006; 66(1): 56-63.
13. Jones AP, Haynes R, Sauerzapf V, Crawford SM, Zhao H, Forman D. Travel times to health care and survival from cancers in Northern England. European Journal of Cancer 2007; 44(2): 269-274.
14. Hippisley-Cox J, Pringle M. Inequalities in access to coronary angiography and revascularisation: the association of deprivation and location of primary care services. British Journal of General Practice 2000; 50(455): 449-454.
15. Twigger J P, Jessop E G. Small area variation in hospital admission: random or systematic? Public Health 2000; 114(5): 328-329.
16. Gruen RL, Weeramanthri TS, Knight SSE, Bailie RS. Specialist outreach clinics in primary care and rural hospital settings. Cochrane Database of Systematic Reviews 2004, issue 1. art. no.: CD003798. DOI: 10.1002/14651858.CD003798.pub2
17. Brewster D, Crichton J, Muir C. How accurate are Scottish cancer registration data? British Journal of Cancer1994; 70(5): 954 -959
18. Hart JT. The inverse care law. Lancet 1971; 1(7696): 405-412.
______________________

