Introduction
Maternal cigarette use during pregnancy is associated with adverse perinatal outcomes, including increased risk of miscarriage1, ectopic pregnancy2, placental abruption3, intrauterine growth retardation4,5 prematurity6,7, and low birth weight (LBW)1-3,8,9. The offspring of mothers who smoked during pregnancy have higher rates of infant mortality10 and morbidity, including congenital anomalies11,12, respiratory disorders13,14, and behavioural difficulties15,16. Increased awareness and understanding of the risks of smoking during pregnancy and widespread public health efforts to promote smoking cessation have resulted in greatly decreased rates of maternal smoking in Canada17. In certain populations, however, the rate of maternal smoking remain high. Factors that increase the risk of maternal smoking include young maternal age, poverty, Aboriginal background, and low maternal education levels17,18.
High rates of smoking in Aboriginal populations are a public health issue internationally. Statistics from Australia, New Zealand, and the USA all reveal high smoking rates among individuals of Aboriginal descent19-22. Statistics Canada report that the combined Inuit populations in Canada have a smoking rate of 57%, which is significantly higher than both the Canadian average and the combined Aboriginal smoking rate23. While Canadian women overall report a rate of maternal smoking in pregnancy between 10% and 16%17,18,24, the rate in Nunavut (where the population is approximately 85% Inuit)23 is between 60% and 85%18,25,26. Considering that other risk factors for maternal smoking (low education level, low income, and young maternal age) are all prevalent in the territory23, the findings of high maternal smoking rates in Nunavut are not unexpected. However, the actual number of women smoking, as well as the lack of any significant decrease in recent decades, are worrisome. Despite considerable national media attention and numerous local strategies aimed to curb smoking rates, the territory's maternal smoking rate has not changed since the early 1990s25.
Perinatal outcomes in Nunavut are a major public health issue. The territory has the highest rates of LBW (<2500 g) and the highest rate of premature births of all Canadian provinces and territories27,28. Rates of small for gestational age (sGA) births and LBW (<2500 g) are important markers of perinatal health, and smoking has been clearly shown to be the single greatest modifiable risk factor for both5,29,30. While overall evidence supports a relationship between maternal smoking and increased risk of prematurity7,9,31, several other studies, including a previous chart review from the Baffin region between 1998-200026 have shown no clear relationship between premature births and maternal smoking4,32.
In order to explore the relationship between smoking, preterm birth (PTB), LBW, and sGA births in this population, a maternal-infant chart review was performed of all Inuit births occurring from 1 January 2003 to 31 December 2005 in the Qikiqtaaluk (Baffin) region of Nunavut. The primary goal was to determine the effect on perinatal outcomes of maternal smoking in general, as well as the effect of the amount smoked. Maternal age, alcohol and cigarette use were also considered for each category of smokers.
Methods
A retrospective cross-sectional maternal/perinatal chart review of infants born to at least one Inuit parent in the Qikiqtaaluk region of Nunavut, as recorded at Baffin Regional Hospital (now called Qikiqtani General Hospital) was performed for the period between 1 January 2000 and 31 December 2005, with a focus on birth outcome. The vast majority of births for women living in the Qikiqtaaluk Region of Nunavut occur at the Qikiqtani General Hospital. Although high risk births occur at the Children's Hospital of Eastern Ontario in Ottawa, most birth records are returned to communities and medical records at the Qikiqtani General Hospital, allowing a high level of ascertainment for this region. Ethics approval for maternal infant chart review was obtained from the University of British Columbia and Nunavut Research Institute.
A subset only of those born between 1 January 2003 and 31 December 2006 were utilized because smoking status and number of cigarettes smoked were less available on prenatal records for the years 2000, 2001 and 2002. Between January 2003 and 1 January 2006, 1022 births were recorded meeting baseline entry criteria of singleton pregnancy with delivery after 24 weeks gestational age. Of those births, smoking status was recorded at the first prenatal visit on 90% (n=918) of prenatal charts, and of those smoking (918), 80% of charts (592) also included quantitative data of the number of cigarettes smoked per day.
The births were divided into five categories of maternal cigarette use: (i) non-smokers; (ii) 5 or less cigarettes per day; (iii) 6-10 cigarettes per day; (iv) more than 10 cigarettes per day; and (v) smoking documented on the chart, but no recorded number of cigarettes available. The cases in each category were analyzed for rates of PTB (defined as less than 37 weeks gestational age), LBW (<2500 g) and sGA births. Classification of sGA was based on a Canadian standard for mean birth weights33, with birth weights below the 10th percentile of weight for gestational age meeting the criteria for sGA.
In addition, for each category average birth weights of those delivered at term (>37 and <42 weeks gestation) were compared. Preterm births were excluded from this analysis to minimize the potential confounding effect of reduced birth weight secondary to shorter gestations.
The proportion of women reporting use of alcohol and street drugs (marijuana, cocaine, other) during pregnancy within each category was documented. Furthermore, PTB, LBW, and sGA were calculated for 4 maternal age groups: (i) 15-20; (ii) 21-24; (iii) 25-29; and (iv) 30-39 years. Because the risk for LBW, PTB, and sGA in the general population are highest with both extremes of maternal age (less than 20 years and over 35), the 25-29 year age group was used as the referent group for comparison.
Statistical analysis was carried out using Fisher's exact test, Student's t-test odds ratios with 95% confidence intervals (CI), ANOVA and Tukey's HSD when indicated, with the statistics program Prism GraphPad (GraphPad Software; http://www.graphpad.com/prism/Prism.htm) and Tukey's HSD (Origin 8.0; http://www.sciencesoftware.com).
Results
A total of 1022 births were recorded between 1 January 2003 and 31 December 2005 at the Baffin Regional Hospital, and 918 births (90%) met the inclusion criteria for this analysis (at least one Inuit parent, singleton birth after 24 weeks gestation, and smoking status documented on the prenatal record).
Smoking rates and amount smoked
Of these, 81% of mothers (n=743) reported smoking to their healthcare providers, usually documented at the first prenatal visit in the first trimester. The other 19% of mothers (n=175) self-reported as non-smokers. Of the smokers with documented numbers of cigarettes per day (592), 36% (n=215) smoked 5 or less cigarettes daily, 33% (N=196) smoked 6-10 cigarettes daily, and 30% (n=181) smoked more than 10 cigarettes daily.
Small for gestational age births
For the study population, 8.3% were sGA, consistent with the Canadian population as a whole27 (Fig1). When the study population was categorized according to amount smoked 4% of infants born to non-smokers and 9.3% born to all smokers were sGA (OR 2.5; 95% CI 1.1-5.4). Fifteen percent of births to women smoking greater than 10 cigarettes daily were sGA, (OR 3.7; 95% CI 1.6-8.8 when compared with non-smokers).
Birth weight
Average birth weights at term decreased from 3681 g for non-smokers to 3310 for those smoking more than 10 cigarettes per day (one way ANOVA, p<0.05) (Fig2). There was a statistically significant difference between all smokers compared with non-smokers (243 g weight difference), and a difference between birth weights of each individual smoking group compared with non-smokers (one-way ANOVA, p< 0.05). Although a consistent gradient of lower birth weight was observed for each of the smoking categories, the differences reached significance between categories only for those smoking 1-5 cigarettes daily and those smoking >10 cigarettes daily (one-way ANOVA, p< 0.05).
Low birth weight infants
The overall rate of LBW infants (born <2500 g) was 7.4% in the study population, which is higher than the 2005 Canadian average of 6.0%28 (Fig3). For Baffin non-smokers, 2.3% had infants with LBW, 1-5 cigarettes daily 3.3%, 6-10 cigarettes 5.6% and 15.4% for infants of mothers who smoked more than 10 cigarettes daily. The over-all risk to have an infant with LBW for all smokers compared with non-smokers was nearly 4 times (OR 3.8, 95% CI 1.4-10.5) and even higher for those smoking more than 10 cigarettes per day, compared with non-smokers (OR 6.7, 95% CI 2.3-19.6).
Preterm birth
In the over-all study population 10.7% of infants were born preterm, well above the Canadian average of 8.1%27 (Fig4). Although there was no statistical difference between premature birth rates for non-smokers (8%) compared with all smokers (11.3%) (OR 1.4, 95% CI 0.8-2.5), for those smoking at least more than 10 cigarettes per day the risk was clearly significant (OR 2.1, 95% CI 1.1-4.2).
Alcohol and drug use
In the chart review, 13.1% of women reported alcohol use during pregnancy, and 15.8% of women reported the use of street drugs, most often marijuana (Fig5). Overall, 26.2% of women reported consuming either alcohol, illicit drugs, or both during pregnancy.
Among non-smokers, 12.6% reported alcohol use during pregnancy, and 9% reported other substance use. Over-all 16.7% of non-smokers were using at least one substance (alcohol or drugs), as reported during the first trimester.
Alcohol use was reported among 13.3% of smokers, and 18.3% reported other substance use during pregnancy. For the smokers, over-all 28% of mothers reported the use of alcohol, drugs, or both. When divided according to smoking category, 40% of women in the highest smoking group reporting using alcohol, other substances or both which was notably higher than the non-smoking group (p<.0001; OR 3.3 95% CI: 1.9 to 5.4).
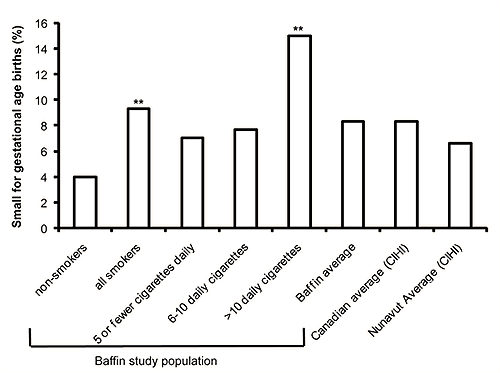
Figure 1: Small for gestational age births. Percentage of births categorized as sGA in the Baffin Region (2003-2005), by smoking amount. Asterisk above bar denotes statistical significance compared with non-smokers. Non-smokers compared with all smokers (OR 2.5, 95% CI 1.1-5.4) and >10 cigarettes per day (OR 3.7, 95% CI 1.6-8.8). The overall sGA rate in the Baffin Region was found to be 8.3%, consistent with reported Canadian average, but higher than average reported for all of Nunavut27.
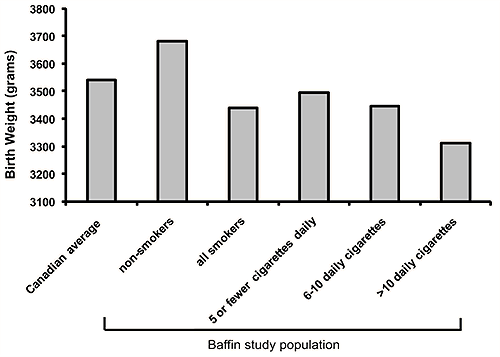
Figure 2: Average term birth weights (>37 weeks and <42 weeks) by smoking amount. For non-smokers the average term birth weight was 3681 g, for all smokers it was 3438 g and the gradient consistently decreased with increased number of cigarettes smoked (one way ANOVA p<0.05 for comparison with all smoking groups compared with non-smokers).
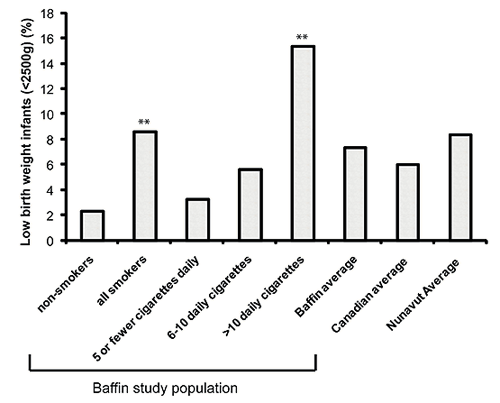
Figure 3: Low birth weight (LBW; <2500 g) by smoking amount. Overall 7.4% of the study population was LBW. Non-smokers had the lowest rate at 2.3%. Asterisk above bar denotes statistical significance compared with non-smokers. For all smokers, 8.6% of infants were LBW (OR 3.8, 95% CI 1.4-10.5 compared with non-smokers). The gradient of LBW increased with the number of cigarettes smoked per day, most significantly (15.4%) in infants of those smoking more than 10 daily cigarettes/day (OR 6.7; 95% CI 2.3-19.6 compared with non-smokers). This was notably higher than reported for all of Canada and Nunavut28.
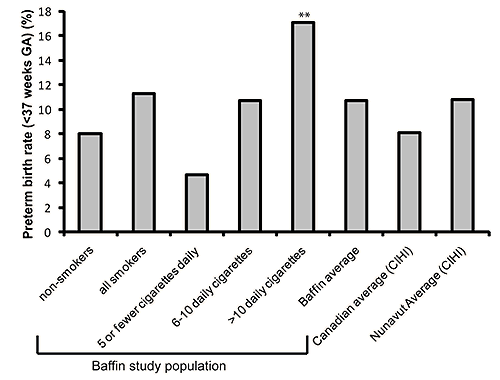
Figure 4: Preterm birth rates by smoking amount. Over-all 10.7% of the present study population were born preterm, which was increased compared with the Canadian average, but was similar to the Nunavut reported average. Eight percent of infants born to non-smokers were preterm, compared with 11.3% for all smokers, which was not statistically significant. Although the rates increased according to cigarettes smoked, statistical significance was only evident for those smoking more than 10 cigarettes/day (indicated by asterisk above bar), compared with non-smokers (OR 2.1; 95% CI 1.1-4.2).
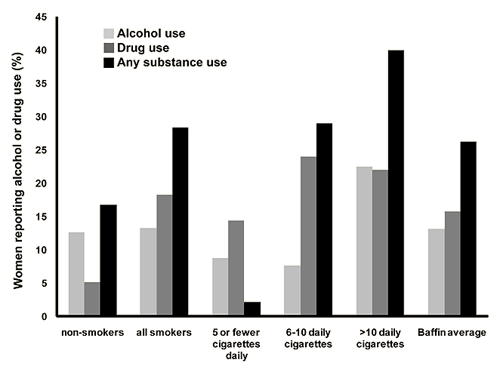
Figure 5: Maternal alcohol and drug use by smoking amount for study population. Percentage of study population reporting alcohol and street drug use according to category of amount of cigarettes smoked.
Maternal age and number of cigarettes per day
Over 75% of births were to women under the age of 30 years, and approximately 25% to women under the age of 20. Average maternal age in the chart review was 24.3 years (Table 1).
The average age of non-smoking women was significantly older than the average age of smokers (25.6 vs 23.9 years old; one-way ANOVA, p <0.05). Women aged 35-39 years had the lowest rates of reported smoking (64%); while women aged 20-24 were most likely to smoke (88% reporting smoking).
While the older age groups were found to have lower overall smoking rates, older mothers who smoked used more cigarettes daily compared with those who were younger. Among smokers aged 15-19 years, 35.4% reported smoking five or less cigarettes daily, while only 11.7% of smokers aged 35-39 years were in the lightest smoking group.
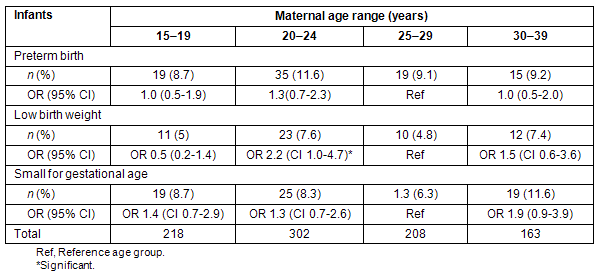
For each age group, PTB, LBW infants, and sGA births were assessed. In comparison with the referent group, there was no statistical difference for all outcomes between the between the age groups; however, the 30-34 year age group exhibited the highest rates for PTB, LBW, and sGA although the small sample size in this group limits generalizations.
Table 1: Preterm births, small for gestational age and low birth weight infants according to maternal age
Discussion
Over 80% of pregnant women included in the chart review reported smoking during pregnancy, which is higher than that found in the majority of reports of Nunavut as a whole18,24, but is consistent with previous data on smoking in women delivering in the Qikiqtaaluk Region25,26. While this smoking rate was especially high compared with the rest of the country, the majority of the women were smoking none or less than 10 cigarettes per day. Only 20% of all pregnant women in the review reported using more than 10 cigarettes per day, and this small proportion of the population who smoked more heavily during pregnancy had the worst perinatal outcomes of all study groups. Smokers who used more than 10 cigarettes daily had six times the rate of LBW infants, a nearly four-fold increased risk of sGA births, and double the rate of premature births compared with non-smokers. Smokers (of any amount) also had inferior perinatal outcomes, with almost a four-fold increased risk of LBW, a two-fold increased risk of sGA and significantly decreased average birth weights compared with the non-smoking group. Among Inuit women who were non-smokers, perinatal outcomes are similar to or even better than outcomes in the rest of Canada, with rates of sGA and LBW infants well below the national average, and rates of prematurity equal to the national average. Although many studies in the general population have suggested an association between PTB and smoking6,32,34, a previous report from the Baffin region did not26. However, by categorizing according to the documented number of cigarettes smoked, the present study demonstrated a clear relationship between smoking and PTB rates, but only for women smoking more than 10 cigarettes daily. In the present study, a comparison of all smokers with non-smokers did not demonstrate a difference, which is consistent with the previous report. Only by comparing reported amounts of smoking was it possible to see the effects of smoking on prematurity. The overall rate of PTB (10.7%) in the region was consistent with previously reported PTB rates in Nunavut (10.8%)27, which is well above the national average of 8.1%27.
Birth weights for term infants in the study population decreased linearly with increasing amounts of cigarettes smoked per day. The dose-response relationship between smoking amounts and birth weights found in the present study population is consistent with previous studies which have found a 12-27 g decrease in birth weight per cigarette smoked8,35,36 or an average decreased birth weight of 200-300 g among smokers in general37,38.
Maternal ages overall were significantly younger than the Canadian average. Although it is expected that younger mothers are more likely to smoke17,18,24, in the present study younger mothers smoked less cigarettes per day, compared with smokers in the older age groups. Over a quarter of the women in the present study reported using alcohol, street drugs, or both during pregnancy. Smokers reported higher rates of substance use compared with non-smokers, and the highest rates of substance use were among women smoking more than 10 cigarettes daily (40% reported alcohol use, other substance use or both). Overall at 13%, the reported rate of alcohol use in the study population was similar to the overall Canadian average of 10.5 to 12.5% for the same time period18,24; However, reported use of street drugs was higher. While 15.8% of women in the present study reported street drug use during pregnancy, only 1% of all Canadian women recently admitted to street drug use during pregnancy18. The accuracy of the information in the present study and other studies may be influenced by women's reluctance to reveal drug use to healthcare providers, because it is generally considered socially unacceptable, as well as being illegal.
A major limitation in this analysis is the lack of data on other related socioeconomic factors which are known to affect PTB rates as well as birth weight, including marital status, family supports, economic factors, and nutritional status29-31,39. Considering that households in Nunavut spend on average 23% of the household budget on food (compared with the national average of 11%)40, it may be that expense of cigarettes could result in less nutritious food being available which could also influence pregnancy outcomes. Furthermore, co-existing depression was not documented, which could predispose to increased smoking, and other substance use in general.
A further limitation of the present study is that study data were derived only on first trimester recording of smoking. There is now evidence that stopping smoking early in pregnancy greatly reduces the detrimental effects of cigarette exposure to the fetus29,41. It was not possible to assess whether those in the lowest smoking groups might have ceased smoking during the pregnancy. thus contributing to their good outcomes. Current public health efforts underway in Nunavut that propose recording smoking and other substance use in every trimester will provide those data.
Despite these limitations, these data suggests that the number of cigarettes smoked at the first prenatal visit may at least flag a high risk outcome. Although it is not suggested that public health strategies focus only on smoking in the high risk group, it is worth considering directing efforts to this group as a priority. The average healthcare costs of the delivery of full-term infant in the neonatal period in Canada is $1,060, while premature infants cost the system, on average, $9,23327. The expense of a premature delivery is likely to be substantially higher for those living in the remote communities of Baffin Island, where an airlift to a tertiary care centre increases costs substantially. Any public health focus that aims to limit the number of pre-term births should have a considerable cost-saving effect.
Although strategies to promote permanent and complete smoking cessation for all smokers is ideal, these data may also suggest a role for targeted risk-reduction strategies. Most importantly, Inuit women in the Baffin region who are not smoking, and those who are smoking less than 5 cigarettes per day have perinatal outcomes equal to or better than Canadian averages, in sharp contrast to an increased risk for adverse outcomes of 2-6 times for those smoking greater than 10 cigarettes per day. Raising the awareness of the importance of positive birth outcomes when not smoking, or at least reducing smoking to less than 5 cigarettes per day, would be an important public health message. At the same time, further research and direct preventative public health strategies can be directed to the highest risk group, with an over-all aim to reduce adverse birth outcomes.
Acknowledgements
The authors acknowledge the staff of medical records of the former Baffin Regional Hospital (now Qikiqtani General Hospital), as well as Audrey Saxten and Dr Sandy Macdonald for their assistance in enabling the original chart review. The authors gratefully acknowledge Dr Sirisha Asuri of the UBC Community Genetics Research program, for her assistance in manuscript preparation and Dr Hamish Mehaffey, University of California San Francisco for his assistance with statistical analysis. Nunavut Tunngavik Corporation (www.tunngavik.com) has reviewed the results. This work was funded through a CIHR New Aboriginal Health Researcher grant to LA. LA is a Michael Smith for Health Research Scholar award recipient.
References
1. Armstrong BG, McDonald AD, Sloan M. Cigarette, alcohol, and coffee consumption and spontaneous abortion. American Journal of Public Health1992; 82(1): 85-87.
2. Castles A, Adams EK, Melvin CL, Kelsch C, Boulton ML. Effects of smoking during pregnancy. Five meta-analyses. American Journal of Preventive Medicine 1999; 16(3): 08-215.
3. Raymond EG, Mills JL. Placental abruption. Maternal risk factors and associated fetal conditions. Acta Obstetricia et Gynecologica Scandinavica1993; 72(8): 633-639.
4. Horta BL, Victora CG, Menezes AM, Halpern R, Barros FC. Low birthweight, preterm births and intrauterine growth retardation in relation to maternal smoking. Paediatric and Perinatal Epidemiology 1997; 11(2): 140-151.
5. Vielwerth SE, Jensen RB, Larsen T, Greisen G. The impact of maternal smoking on fetal and infant growth. Early Human Development 2007; 83(8): 491-495.
6. Shiono PH, Klebanoff MA, Rhoads GG. Smoking and drinking during pregnancy. Their effects on preterm birth. JAMA 1986; 255(1): 82-84.
7. Jaddoe VW, Troe EJ, Hofman A, Mackenbach JP, Moll HA, Steegers EA et al. Active and passive maternal smoking during pregnancy and the risks of low birthweight and preterm birth: the Generation R Study. Paediatric and Perinatal Epidemiology 2008; 22(2): 162-171.
8. Bernstein IM, Mongeon JA, Badger GJ, Solomon L, Heil SH, Higgins ST. Maternal smoking and its association with birth weight. Obstetrics and Gynecology 2005; 106(5-Pt1): 986-991.
9. Higgins S. Smoking in pregnancy. Current Opinion in Obstetrics and Gynecology 2002; 14(2): 145-151.
10. Wisborg K, Kesmodel U, Henriksen TB, Olsen SF, Secher NJ. A prospective study of smoking during pregnancy and SIDS. Archives of Disease in Childhood 2000; 83(3): 203-206.
11. Malik S, Cleves MA, Honein MA, Romitti PA, Botto LD, Yang S et al. Maternal smoking and congenital heart defects. Pediatrics 2008; 121(4): e810-816.
12. Wyszynski DF, Duffy DL, Beaty TH. Maternal cigarette smoking and oral clefts: a meta-analysis. Cleft Palate-Craniofacial Journal 1997; 34(3): 206-210.
13. Weitzman M, Gortmaker S, Walker DK, Sobol A. Maternal smoking and childhood asthma. Pediatrics 1990; 85(4): 505-511.
14. Taylor B, Wadsworth J. Maternal smoking during pregnancy and lower respiratory tract illness in early life. Archives of Disease in Childhood 1987; 62(8): 786-791.
15. Weitzman M, Gortmaker S, Sobol A. Maternal smoking and behavior problems of children. Pediatrics1992; 90(3): 342-349.
16. Brook DW, Zhang C, Rosenberg G, Brook JS. Maternal cigarette smoking during pregnancy and child aggressive behavior. American Journal on Addictions 2006; 15(6): 450-456.
17. Millar WJ, Hill G. Pregnancy and smoking. Health Reports 2004; 15(4): 53-56.
18. Public Health Agency of Canada. What mothers say: The Canadian Maternity Experiences Survey. (Online) 2009. Available: http://www.phac-aspc.gc.ca/rhs-ssg/pdf/survey-eng.pdf (Accessed 1 December 2009).
19. Environics Research Group Limited. 2004 Baseline Study Among First Nations (On-Reserve) and Inuit. Toronto, ON: ERG, 2004.
20. Thomas DP. Smoking prevalence trends in Indigenous Australians, 1994-2004: a typical rather than an exceptional epidemic. International Journal of Equity in Health 2009; 8: 37.
21. CDC. Cigarette Smoking Among Adults and Trends in Smoking Cessation - United States, 2008. MMWR 2009; 58(44): 1227-1232. Available: http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5844a2.htm (Accessed December 16th, 2009).
22. Panaretto K, Lee H, Mitchell M, Larkins S, Manessis V, Buettner P et al. Risk factors for preterm, low birth weight and small for gestational age birth in urban Aboriginal and Torres Strait Islander women in Townsville. Australian and New Zealand Journal of Public Health 2006; 30(2): 163-170.
23. Tait H. Aboriginal Peoples Survey, 2006: Inuit Health and Social Conditions. Social and Aboriginal Statistics Division. (Online) 2008. Available: http://www.statcan.gc.ca/pub/89-637-x/89-637-x2008001-eng.pdf (Accessed 1 January 2010).
24. Public Health Agency of Canada. Canadian Perinatal Health Report. (Online) 2008. Available: http://www.phac-aspc.gc.ca/publicat/2008/cphr-rspc/pdf/cphr-rspc08-eng.pdf. (Accessed 1 December 2009).
25. Arbour L, Gilpin C, Millor-Roy V, Platt R, Pekeles G, Egeland GM et al. Heart defects and other malformations in the Inuit in Canada: a baseline study. International Journal of Circumpolar Health 2004; 63(3): 251-266.
26. Muggah E, Way D, Muirhead M, Baskerville B. Preterm delivery among Inuit women in the Baffin Region of the Canadian Arctic. International Journal of Circumpolar Health 2004; 63(Suppl2): 242-247.
27. Canadian Institute for Health Information. Too early, too small: a profile of small babies across Canada. (Online) 2009. Available: http://secure.cihi.ca/cihiweb/products/too_early_too_small_en.pdf (Accessed 1 December 2009).
28. Statistics Canada. Health - low birth weight. Live birth, by birth weight (less than 2,500 grams) and sex, Canada, provinces and territories, annual (CANSIM Table 102-4509). (Online) 2008. Available: http://www4.hrsdc.gc.ca/.3ndic.1t.4r@-eng.jsp?iid=4#M_4 (Accessed 1 November 2009).
29. McCowan L, Horgan RP. Risk factors for small for gestational age infants. Best Practice Research in Clinical Obstetrics and Gynaecology 2009; 23(6): 779-793.
30. Tsukamoto H, Fukuoka H, Koyasu M, Nagai Y, Takimoto H. Risk factors for small for gestational age. Pediatrics International 2007; 49(6): 985-990.
31. Chan A, Keane RJ, Robinson JS. The contribution of maternal smoking to preterm birth, small for gestational age and low birthweight among Aboriginal and non-Aboriginal births in South Australia. Medical Journal of Australia 2001; 174(8): 389-393.
32. Peacock JL, Bland JM, Anderson HR. Preterm delivery: effects of socioeconomic factors, psychological stress, smoking, alcohol, and caffeine. BMJ1995; 311(7004): 531-535.
33. Kramer MS, Platt RW, Wen SW, Joseph KS, Allen A, Abrahamowicz M et al. A new and improved population-based Canadian reference for birth weight for gestational age. Pediatrics 2001; 108(2): E35.
34. Cnattingius S, Forman MR, Berendes HW, Graubard BI, Isotalo L. Effect of age, parity, and smoking on pregnancy outcome: a population-based study. American Journal of Obstetrics and Gynecology 1993; 168(1-Pt1): 16-21.
35. Mathai M, Skinner A, Lawton K, Weindling AM. Maternal smoking, urinary cotinine levels and birth-weight. Australian and New Zealand Journal of Obstetrics and Gynaecology 1990; 30(1): 33-36.
36. England LJ, Kendrick JS, Wilson HG, Merritt RK, Gargiullo PM, Zahniser SC. Effects of smoking reduction during pregnancy on the birth weight of term infants. American Journal of Epidemiology 2001; 154(8): 694-701.
37. Ellard GA, Johnstone FD, Prescott RJ, Ji-Xian W, Jian-Hua M. Smoking during pregnancy: the dose dependence of birthweight deficits. British Journal of Obstetrics and Gynaecology 1996; 103(8): 806-813.
38. Secker-Walker RH, Vacek PM, Flynn BS, Mead PB. Estimated gains in birth weight associated with reductions in smoking during pregnancy. Journal of Reproductive Medicine 1998; 43(11): 967-974.
39. Wenman WM, Joffres MR, Tataryn IV. A prospective cohort study of pregnancy risk factors and birth outcomes in Aboriginal women. Canada Medical Association Journal 2004; 171(6): 585-589.
40. Statistics Canada. The Daily: Household spending. (Online) 2002. Available: http://www.statcan.gc.ca/daily-quotidien/021211/dq021211a-eng.htm (Accessed 1 January 2010).
41. Polakowski LL, Akinbami LJ, Mendola P. Prenatal smoking cessation and the risk of delivering preterm and small-for-gestational-age newborns. Obstetrics and Gynecology 2009; 114(2-Pt1): 318-325.
Abstract
Introduction: In Nunavut, 60-80% of pregnant women report smoking in pregnancy, a rate five times the Canadian average. Nunavut also has the highest rates of preterm birth and low birth weight infants in Canada. The present study assessed whether the number of cigarettes smoked per day, as recorded in the first trimester, influenced birth outcomes.
Methods: Maternal-newborn charts were reviewed for infants born between 1 January 2003 and 31 December 2005 to at least one Inuit parent in the Qikiqtaaluk (Baffin) region of Nunavut. Smoking data, as reported by the mother at an early prenatal visit, were extracted from the prenatal record. Birth outcomes including birth weights (for term births), low birth weight, small for gestational age births and rates of preterm birth, were analysed according to category of reported number of cigarettes smoked (0, 1-5, 6-10, and >10 per day). Maternal age, alcohol and street drug use were also assessed for each category of smokers. Statistical analysis among groups was carried out.
Results: Of 918 births meeting the study criteria, more than 80% of women reported smoking. For 80% of those, the amount smoked per day was available. Non-smokers and women smoking less than 5 cigarettes daily had perinatal outcomes equal to or better than Canadian averages for low birth weight, small for gestational age, and preterm birth. Furthermore, average birth weights at term significantly decreased from 3681 g for non smokers to 3310 g for those smoking more than 10 cigarettes per day. Compared with non-smokers, women in the highest smoking category (>10 cigarettes daily) had a six-fold increase in low birth weight infants (OR 6.7, 95% CI 2.3-19.6), almost a four-fold increase for small for gestational age births (OR 3.7, 95% CI 1.6-8.8) and twice the chance of a premature birth compared with non-smokers (OR 2.14, 95% CI 1.1-4.2). Those in the highest smoking category were also most likely to report alcohol and other substance use.
Conclusions: Inuit women who reported not smoking, or smoking less than 5 cigarettes per day had birth outcomes equal or superior to average Canadian outcomes in each category evaluated. Those women reporting smoking more than 10 cigarettes daily had significantly increased risk for preterm birth, low birth weight and small for gestational age infants. Because those in the highest smoking category also reported the highest rates of alcohol and other substance use, it is likely that a combination of factors assessed in this study influenced the outcomes. Along with public health preventative measures to reduce smoking directed to this high risk group, other contributing factors for adverse birth outcomes need to be assessed more fully to understand the complex interactions that lead to increased smoking, substance use and, therefore, adverse birth outcomes. Furthermore, evidence from this study suggests that smoking more than 10 cigarettes per day, reported at first prenatal visit, may provide a marker for those women at highest risk of poor outcomes, which could provide direction for focused public health efforts.
Key words: Aboriginal, Inuit, low birth weight, Nunavut, pregnancy, prematurity, risk-reduction, small for gestational age, smoking.
You might also be interested in:
2022 - Developing supervision capacity for training rural generalist doctors in small towns in Victoria
2005 - Retaining rural medical practitioners: Time for a new paradigm?


