Ivermectin (Mectizan) is an effective and well-tolerated microfilaricidal drug capable of interrupting the development of microfilariae in the uterus of the adult worm. Ivermectin has emerged as the drug of choice for large-scale treatment of onchocerciasis1. Ivermectin is given to persons 5 years and older, or those weighing more than 15 kg. However, in communities where acceptance and compliance with annual treatment is high, transmission of the disease has been reported to be disrupted and children under five years have been monitored. Repeated doses of ivermectin is reported to lower the incidence of microfilardermia in untreated children living in treated communities2,3 while a single dose will markedly reduce skin microfilarial loads for up to 12 months with a transient fall in microfilarial level4. In addition, ivermectin has a positive effect in reducing transmission2,3,5 while the clinical benefits of ivermectin treatment are widely documented1,6,7.
Since the registration of ivermectin in 1987, the control of onchocerciasis has been revolutionized as effective distribution strategies evolved from vertical distribution using Community-Based Distributors (CBD) to the current Community-Directed Treatment with Ivermectin (CDTI), the major strategy of the African Programme for Onchocerciasis Control (APOC). The success of the CDTI program depends on how effectively the Community-Directed Distributors (CDD) are able to manage the distribution of ivermectin annually, as well as monitor and report the community response to ivermectin so as to properly direct control efforts. Nigeria remains one of the most affected countries in the world with over 6 million people infected, 13 million at risk of having the disease, over 100 000 blind and an estimated 1 million presenting with various forms of skin abnormalities. Onchocerciasis is endemic in 31 of the 36 States of the country including the Federal Capital Territory of Abuja. Repeated annual treatment with ivermectin commenced approximately 10 years ago in most of the endemic communities after undertaking a rapid assessment of communities to be treated using Rapid Epidemiological Mapping of Onchocerciasis (REMO). Although mass ivermectin treatment is continuing in onchocerciasis-endemic communities, reports on the effectiveness of treatment are still scanty despite the existence of a spectrum in the pattern of disease presentation and variation in treatment outcome among geographic regions and countries. Variation in treatment outcome may occur between studies that are closely monitored for ivermectin efficacy in a scientific setting and those set in communities with no operational mechanism for monitoring annual ivermectin distribution (eg the use of out-put indicators such as average acceptance rate [AAR], the average percentage of individuals in a given community who received annual treatment with ivermectin during the treatment period; and community compliance rate [CCR], the percentage of individuals in a given community who completed uninterrupted annual treatment with ivermectin during the treatment period8).
This article complements earlier reports8,9 with a focus, however, on the effect of monitoring annual community-based ivermectin distribution on the Community Microfilarial Load (CMFL) of 10 onchocerciasis-endemic communities in Lade District, Kwara State, Nigeria.
Setting
The topography of Lade District consists of an undulating landscape of hills and valleys with several bridges over through-running culverts. The communities are mainly pockets of settlements about seasonal streams and small rivers that link to a major river, the River Niger. The vegetation is a combination of forest and savanna (forest-savanna mosaic), the Guinea-type vegetation. The thick vegetation that surrounds the seasonal streams provides shade for the Simulium vector, as well as breeding sites.
The population is largely members of the Nupe tribe who are predominantly Muslim. The studied population is stable, mostly farmers who cultivate cereals and rice. The male population is mostly engaged in farming while the women assist on the farms during the harvesting period when more hands are needed. The sex ratio (male : female) of the general population is approximately 1:2 However, the male population of farmers and construction workers is more affected by the primarily rural onchocerciasis than is the female population. The age group studied ranged from 20-68 years. Generally, individuals between 20-55 years old are actively involved in agriculture in this region.
Study area/population
This study covered 10 onchocerciasis-endemic villages in Lade District, Patigi Local Government Area (formerly Edu Local Government Area) of Kwara State, Nigeria. Lade District is located off the Jebba-Kaduna expressway, approximately 167 km from Ilorin, the administrative Capital of Kwara State (Latitude 08°, 45', 33": Longitude 05° 37', 05"). Forty-four communities make up Lade District and they are located at either side of a major road with minor roads and footpaths linking the various communities (Figure 1). Ten communities (Raniworo, Dzwajiwo, Mari, Lile, Gbodongi, Bongi, Chitta, Ndanako, Lata and Magya) were randomly selected for this study from the 30 communities receiving treatment (Figure 1).
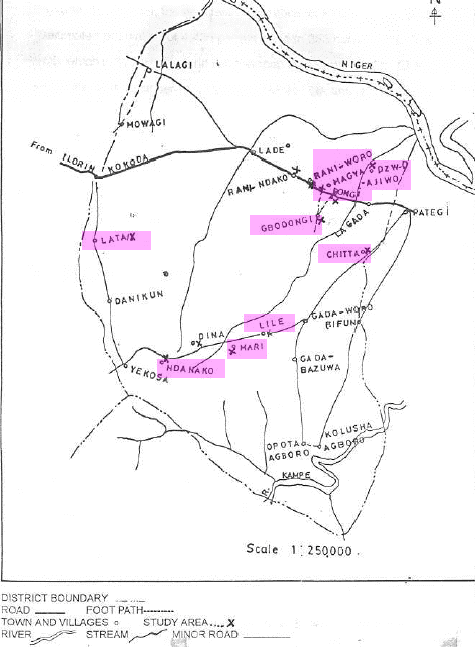
Figure 1: The study area of 10 onchocerciasis-endemic villages in Lade District, Patigi Local Government Area (formerly Edu Local Government Area) of Kwara State, Nigeria. The 10 villages (pink highlight) are Raniworo, Dzwajiwo, Mari, Lile, Gbodongi, Bongi, Chitta, Ndanako, Lata and Magya.
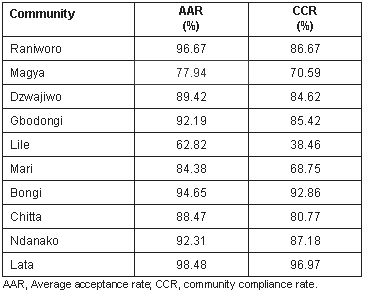
The studied population was a cohort of 330 men from whom parasitologic pre-treatment data were collected before mass ivermectin distribution commenced in 1992. Results of the preliminary evaluation have been reported9. The average (or mean) acceptance rate (AAR) is the average percentage of individuals in a given community who received annual treatment with ivermectin during the treatment period. Mean ivermectin acceptance found by another study of the same communities8 in Lade District in the period 1992-1995 is shown (Table 1). The village Lata was reported to have the highest CCR (the percentage of individuals in a given community who completed uninterrupted annual treatment with ivermectin during the treatment period) at 96.97%, while the lowest was in Lile (38.46%) where 30.77% of the individuals treated with ivermectin presented with adverse reactions at the first round of ivermectin treatment.
Table 1: Mean ivermectin acceptance of the study communities in the period 1992-1995, as found by Oyibo and Fagbenro-Beyioku8
All the communities reported in this study were receiving ivermectin through the CDTI program. The participants were evaluated just before the fifth round of annual ivermectin treatment in August-September 1996, and twelve months after the fifth round in August-September 1997. The August-September 1997 evaluation was conducted just before the sixth round of annual ivermectin treatment. Parasitologic evaluations in the study areas were not immediately possible (1993-1995) due to the unstable political situation in the country that did not provide a conducive setting for fieldwork.
Community mobilization
Following the poor acceptance and compliance with annual ivermectin treatment in some of the communities, the people were properly mobilized by explaining the expected adverse reactions, benefits of the drug, and reassured about the safety of the drug. This was done just before the fifth round of annual ivermectin distribution, the first data collection point of the present study.
Ivermectin administration
In the study areas, an international non-governmental organization, AFRICARE, was the agency involved in annual ivermectin distribution. This agency distributed ivermectin in collaboration with the Kwara State Ministry of Health. Before the commencement of ivermectin distribution, advocacy visits were paid to the Village Head (VH) of each community, who in turn summoned the elders and the people for a briefing about the exercise. The community was thereafter fully mobilized with health education information concerning the disease and its control with ivermectin.
Each community had a trained CBD for the purpose of distributing ivermectin. Ivermectin (150 mg/kg body weight) was administered house-to-house by the CBD who observed the manufacturer's exclusion criteria and administered the drug to each eligible subject after weighing. Subjects weighing less than 15 kg, children under 5 years old, severely ill persons, pregnant/lactating women and the aged were excluded from treatment. Those who weighed 25-44 kg received 1 x 6 mg tablet; 45-64 kg, 1.5 x 6 mg tablets; and those weighing greater than 65 kg, 2 x 6 mg tablets of ivermectin. The drug was swallowed in the presence of the CBD at each distribution point.
Methods
Parasitologic examination
As mentioned previously, the study population was a cohort of 330 men from whom pparasitological pre-treatment data were collected before mass ivermectin distribition commenced in 1992.
Skin snips were obtained using standard parasitologic procedure. Two bloodless skin snips from participants' left and right iliac crests were obtained with the aid of a 2.0 mm Holth type Corneo-Scleral Punch (Storz Instrument, GMBH, Heidelberg, Germany) after swabbing the area with 70% alcohol. The skin snips were incubated for 24 h in an 0.5 mL microfuge tube containing 0.2 mL normal saline, before microscopic examination and enumeration of microfilariae. Aseptic sterilization procedure was applied after obtaining the skin snips from each subject: the Corneo-Scleral Punch was cleaned with cotton wool soaked in 70% alcohol, immersed in 75% ethanol for 30 s, 2% sodium hypochlorite solution for 1 min, 2% glutaraldehyde for 45 s and then cleaned again with cotton wool soaked in 70% alcohol. Although cumbersome, this method ensured proper sterilization of the instrument to avoid the transfer of infectious agents from one subject to another.
The CMFL for each community, the most sensitive parasitologic indicator, was computed using the log (x + 1) transformation described by Remme et al.10. The change in filarral load was analysed using t-tests and Fisher's exact tests with significance defined at the 95% confidence level.
Results
Decrease in CMFL was recorded in a number of communities at the first post-treatment evaluation in July-August 1996 and three were significant (p = <0.05) (Table 2). There was a significant increase (p = <0.05) in CMFL in two communities.
Table 2: 1992 pre-treatment and 1996 and 1997 post-treatment community microfilarial load levels in Lade District, Kwara State, Nigeria
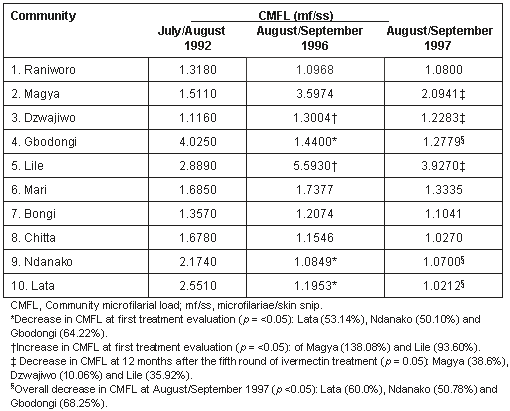
At the August-September 1997 evaluation, 12 months after the fifth round of ivermectin treatment, there was an insignificant increase (p = 0.05) in the CMFL levels in approximately 30% of the communities when compared with the July-August 1992 pretreatment CMFL levels (Table 2). When increases in CMFL at the August-September 1997 evaluation were compared with the increase recorded at the first post-treatment evaluation in August-September 1996, the 138.1% increase in Magya was reduced to 38.63% (72.05% reduction); Dzuwajiwo, 16.52% to 10.06% (39.10% reduction); and Lile, 93.6% to 35.92% (61.62% reduction). Worthy of note is the overall reduction in Mari from 20.86% below the 1992 pre-treatment level (Mari recorded a CMFL increase of 3.13% in August-September 1996). There were variable reductions in the CMFL of the remaining communities and three were significant (p = <0.05; Table 2).
Discussion
Repeated treatment with ivermectin after 5 years and 11 months (just before the sixth round of annual administration) in Lade District had some effect on the CMFL in the studied communities. These effects however varied from one community to another. Because CMFL is the most sensitive indicator of measuring the effect of onchocerciasis control in the community10, reductions in CMFL essentially indicate a positive effect of ivermectin treatment as the number of Onchocerca volvulus microfilariae, capable of contributing to the transmission of the disease in the community, are reduced2,3,5. In these communities, CBD-distributed ivermectin is given annually house-to-house after receipt of the drug from the distributing agency (in collaboration with the officials of the Ministry of Health).
At the first evaluation in August-September 1996 following 4 years of cumulative annual ivermectin distribution, there were reductions in the CMFL levels in all except four communities, three of which were significant (Table 2). Similar reductions in infectivity have been reported in previous studies2,3,6,7,11-13. However, the results obtained at this period of evaluation (August-September 1996) exposed lapses that could occur in community-based ivermectin control efforts when compared with results obtained in scientifically monitored or controlled studies where all-round significant reductions have been reported in Togo14, and in Ivory Coast15. These lapses were identified in a study carried out immediately after the parasitologic examination of 19968.
In the previous study by the present authors, it was observed that one of the communities with increased CMFL, Lile, had a low AAR average of 62.82% for the first 4 years of repeated ivermectin treatment, compared with the other nine communities' AAR which ranged from 77.94%-98.48% (Table 1)8. In addition, three communities with high AAR (Lata, Ndanako and Gbodongi) recorded significant reductions in CMFL; although Bongi and Raniworo had high AAR and CCR, significant reduction in CMFL was not observed. In the previous study, the reported CCR with annual ivermectin treatment (defined as all individuals that took complete doses of ivermectin in the 4 year period8) showed Lile to have the lowest CCR 46% compared with the other communities' CCR which ranged from 68.75% (Mari) to 96.97% (Lata). Based on our results, a relationship among AAR, CCR and CMFL in communities receiving ivermectin is possible. In communities with high AAR, CMFL was low; while communities with low CCR, CMFL was high. Because a relationship was not validated in this study, we are proposing a larger future study to enable a definite inference. A validated relationship in a multi-country study would indicate that the out-put indicators AAR and CCR could be used to rapidly identify the direction of control efforts with ivermectin. One important issue to be addressed is the cut-off points at which AAR and CCR could affect the CMFL.
The highest rejection of ivermectin treatment was in Lile (19.87%)8. Ivermectin was, however, well tolerated in the other communities in Lade District. Absenteeism from treatment was 17.65% in Magya and 14.10% Lile8. Lile, which had a high ivermectin acceptance rate at the first year of ivermectin distribution (94.87%) dropped to 41.03% at the fourth year of treatment due to adverse effects of ivermectin (38.46%) and this was responsible for the poor compliance with annual ivermectin treatment8. Poor post-treatment follow up and poor mobilization with ivermectin-treatment information were the reasons for the poor response to annual ivermectin acceptance.
In the present study, we were able to achieve reductions in the CMFL levels in the communities after effective mobilization by explaining the expected adverse reactions, and reassuring community members of the safety of the drug so as to avoid future rejection. This was done just before the fifth round of annual ivermectin distribution (1996), where there was greater than 90% ivermectin acceptance in each community. The post-treatment parasitologic evaluation 12 months after treatment (1997) showed significant reduction in the CMFL level in three communities Magya (Table 2). The CMFL reductions in three other communities were remarkable but did not fall below the 1992 pre-treatment level, except in Mari where an overall reduction was observed (Table 2).
These data confirm the efficacy of ivermectin in the control of onchocerciasis in the reduction of community microfilarial load and in the variability of treatment outcome 2,3,6,7,9,11,12. Nevertheless, in community-based annual ivermectin distribution, poor mobilization and non-monitoring of community treatment using simple indices such as CCR and AAR8, could work against onchocerciasis control. The situation in Lile where CMFL levels increased with subsequent ivermectin rejections following adverse reactions is worthy of mentioning here. Other studies16,17 reported encouraging participation in annual ivermectin distribution even after severe adverse reactions16,17. This was achieved by the use of health education presentations and the implementation of other health programs. In these studies, however, participant communities were closely monitored after each treatment round.
Conclusion and Recommendation
The authors recommend routine monitoring of biannual ivermectin distribution and treatment, and the institution of an effective bi-directional feedback mechanism as a vital component of a community-directed ivermectin distribution program. This should be closely studied in further investigations.
There needs to be a flexible approach to distribution. Unlike the situation where a CBD did not retain a supply of medication for those who were absent during the period of annual ivermectin distribution, CDD who retain unused medication for a short period can then distribute ivermectin to individuals who were absent during the main ivermectin distribution period.
An efficient reporting system or mechanism enables the managers of ivermectin distribution (CDDs) in CDTI programs to monitor the trend of treatment and report back to the supervising government authority. The use of simple out-put indicators, such as AAR and CCR, in a cross-sectional community study is recommended8.
References
1. WHO. Onchocerciasis and its Control. Report of a WHO expert committee on onchocerciasis control. Technical Report Series no. 852. Geneva: WHO, 1995.
2. Boussinesq M, Chippaux YP, Ernould JC, Quillevere D, Prod'hon J. Effects of repeated treatments with ivermectin on incidence of onchocerciasis in northern Cameroon. American Journal of Tropical Medicine and Hygiene 1995; 53: 63-67.
3. Boussinesq M, Prod'hon J, Chippaux JP. Onchocerca volvulus Striking decrease in transmission in the Vina Valley (Cameroon) after eight annual large-scale ivermectin treatments. Transcriptions of the Royal Society of Tropical Medicine and Hygiene 1997; 91: 82-86.
4. Taylor HR, Greene BM. The status of ivermectin in the control of onchocerciasis. Acta Leiden 1989; 59: 201-206.
5. Boatin BA, Hougard JM, Alley ES, Akpoboua LKB, Yameogo L, Dembele N. The impact of Mectizan treatment on the transmission of onchocerciasis. Annals of Tropical Medicine and Parasitology 1998; 92(Suppl. 1): S47- S60.
6. Whitworth JAG, Morgan D, Maude GH, Luty JF, Taylor DW. A community trial of ivermectin for onchocerciasis in Sierra Leone: clinical and parasitological responses to four doses given at six-monthly interval. Transcriptions of the Royal Society of Tropical Medicine and Hygiene1992; 84: 277-280.
7. Whitworth JAG, Luty JF, Maude GH, Morgan D, Downham MD, Taylor HR. Ivermectin does not reduce the burden of itching in an onchocerciasis endemic community. Transcriptions of the Royal Society of Tropical Medicine and Hygiene 1992; 86: 281-283.
8. Oyibo WA, Fagbenro-Beyioku AF. Community response to repeated annual ivermectin treatment of onchocerciasis in Nigeria. Parasitology Research 2002; 88: 704-707.
9. Oyibo WA, Fagbenro-Beyioku AF. Reduced prevalence of onchocerciasis following mass ivermectin treatment over four years. East African Medical Journal 1997; 74: 326-330.
10. Remme J, Ba O, Dadzie KY, Karam M. A force-of-infection model for onchocerciasis and its implication in the epidemiological control programme in the Volta River Basin Area. Bulletin of the World Health Organisation 1986; 64: 667-681.
11. Collins RC, Gonzales-Perata C, Castro J et al. Ivermectin: reduction in prevalence and infection intensity of Onchocerca volvulus following bi-annual treatments in five Guatemalan communities. American Journal of Tropical Medicine and Hygiene1992; 47: 156-169.
12. Rodriguez-Perez MA, Rodriguez MH, Margeli-Perez HM, Rivas-Alcala AR. Effects of semi- annual treatments of ivermectin on the prevalence and intensity of Onchocerca volvulus skin infection, ocular lesions, and intensity of Simulium ochraceum populations in Southern Mexico. American Journal of Tropical Medicine and Hygiene 1995; 52: 429-434.
13. Newell ED. Effects of mass treatment with ivermectin, with only partial compliance, on the prevalence and intensity of O. volvulus infection in adults in Burundi. Tropical Medicine and International Health1997; 2: 912-916.
14. Schulz-Key H., Heuschkel C, Banla M. Long-term efficacy of ivermectin on Onchocerca volvulus. Annals of Tropical Medicine and Parasitology 1989; 40: 493-494.
15. Lariviere M., Beauvais B, Aziz M. Etude en Cote-d'Ivore (1985-1987) de l'effectaute et de la torelance de l'ivermectine (mectizan) dans l'onchocerciasis humaine. II. Evaluation, en vue de campagnes de masse, des effects de l'administration annuelle ou semestrielle de doses uniques orales de 100, 150, ou 200mcg/kg. Bulletin de la Societe Pathologie Exotique 1989; 82: 48-57.
16. Baraka OZ, Khier MM, Ahmed MK et al. Community-based distribution of ivermectin in eastern Sudan: acceptability and early post treatment reactions. Transcriptions of the Royal Society of Tropical Medicine and Hygiene (London) 1995; 89: 316-318.
17. Guderian RH, Anselmi RM, Espinel M, Mancerot T, Riverdeneira G, Proano R. Successful control of onchocerciasis with community-based ivermectin distribution in the Rio Santiago focus in Ecuador. Tropical Medicine and International Health 1997; 2: 982-988.
Abstract
Introduction: Ivermectin (Mectizan) is an effective and well-tolerated microfilaricidal drug that has emerged as the drug of choice for large-scale treatment of onchocerciasis. Repeated doses of ivermectin is reported to lower the incidence of microfilardermia, while a single dose will markedly reduce skin microfilarial loads for up to 12 months with a transient fall in microfilarial level.
Aim: The aim of the study was to evaluate the effect of repeated community-based ivermectin treatment on the intensity of onchocerciasis in 10 onchocerciasis-endemic communities of Lade District, Pategi Local Government Area of Kware State, Nigeria.
Methods: The participants, a cohort of 330 men from whom parasitologic pre-treatment data were collected before mass ivermectin distribution commenced in 1992, were evaluated just before the fifth round of annual ivermectin treatment in August-September 1996, and 12 months after the fifth round in August-September 1997. Two bloodless skin snips were obtained from participants' left and right iliac crests and incubated for 24 h in an 0.5 mL microfuge tube containing 0.2 mL normal saline, before microscopic examination and enumeration of microfilariae. The community microfilarial load (CMFL), the most sensitive parasitologic indicator, was computed.
Results: Following four rounds of repeated annual treatments of onchocerciasis with ivermectin in the 10 communities studied, varying degrees of reduction in CMFL were observed in six of the 10 communities. These reductions were significant in three of the six communities at the first evaluation carried out just before the fifth round of annual ivermectin treatment (p = <0.05). There was an increase in the remaining four communities, mainly attributed to poor annual acceptance and compliance with annual ivermectin treatment due to the occurrence of severe adverse reactions and absenteeism. Post-treatment evaluation (12 months after the previous evaluation), just before the sixth round of annual ivermectin treatment was preceded by community mobilization (enlightenment on the usefulness of ivermectin an assurance on its safety), especially in the four affected communities. This brought about reductions in the CMFL in the four communities that were not previously affected. Although the CMFL of these four communities was reduced, the CMFL of three of the communities did not fall below the 1992 pre-treatment level but was below the level recorded 12 months earlier. One of these affected communities had its CMFL reduced below the 1992 pre-treatment CMFL.
Conclusion: This study confirms the efficacy of ivermectin in the reduction of CMFL but exposes the danger of failing to monitor annual ivermectin treatment where community ivermectin-based control efforts are ongoing. The results obtained from this study are not as impressive as results reported from closely monitored scientific evaluations of ivermectin efficacy studies.
Key words: chemotherapy, community treatment, ivermectin, onchocerciasis.
You might also be interested in:
2011 - Earthquake, tsunami, radiation leak, and crisis in rural health in Japan

