Introduction
Dilated cardiomyopathy is a significant health problem in Africa. It has been found to account for up to 48% of hospital admissions with heart failure in South Africa1. Diagnosis and treatment can be challenging as it frequently affects young patients and those without risk factors for cardiac disease. Patients can also be morbidly unwell at the time of presentation. These challenges are further compounded within a rural setting. No data exist to suggest that dilated cardiomyopathy is any more prevalent within rural areas. However, local factors such as high HIV prevalence, prevalent familial forms and exposure to toxins, including venom, may theoretically predispose individuals to developing the disease2. Diagnosis and treatment are often further hindered by limited resources and a small range of available investigations.
The following case study is directed at junior medical staff currently working within rural settings. Consent was obtained from the patient for the publication of this case as a teaching article.
The case of a young South African male who presented as acutely unwell to Mseleni Hospital with dilated cardiomyopathy is described. Mseleni Hospital is a small rural hospital in the north of South Africa. It has approximately 200 beds and caters for male and female medical and surgical patients, obstetrics and paediatrics. It has a catchment population of 90 000. Medical staffing is variable and the hospital frequently has a relatively high concentration of foreign doctors. Although the permanent staff are very experienced, there is no specialist care and the nearest referral centre is over 4 hours drive away. Diagnostic tests are limited, although plain film radiology, routine blood tests and simple ultrasound are all possible. Current guidelines for management and treatment of the disease and the unique challenges it presents to those working in a rural setting are discussed. In doing so, it is hoped prompt diagnosis and efficacious treatment will be encouraged, and overall prognosis improved.
Case
A 17 year old African boy presented to the outpatient department of Mseleni Hospital in South Africa on 17 December 2011 with a 1 month history of shortness of breath. His symptoms began gradually with a prodromal history of headache and malaise. His shortness of breath had worsened and he was now short of breath at rest. Exertion of any kind exacerbated his symptoms, as did lying down, and he had also developed a dull central chest pain. He denied any history of cough and also denied any illicit drug abuse, alcohol intake or exposure to toxins. He had no allergies to his knowledge and was taking no regular medicines. He had no significant past medical or family history.
On admission, he was clearly in respiratory distress. He had no peripheral stigmata of chronic disease. His JVP was elevated and he had a displaced apex beat with a parasternal thrill. On auscultation of the chest, he had a loud P2, third heart sound and a pansystolic murmur heard loudest at the apex. His breath sounds were vesicular with added bibasal crepitations. His abdomen was soft. He had a pulsatile non-tender liver edge palpable 3cm below the costal margin. His calves were soft and non-tender and unusually he had no peripheral oedema.
Bloods were unremarkable. His anti-streptolysin O (ASO) titre was negative and Enzyme-Linked Immunosrobant Assay (ELISA) for both HIV 1 and 2 were negative. A chest x-ray was performed, which showed a prominent pulmonary trunk and a grossly dilated cardiac shadow that prompted a cardiac ultrasound. This showed no pericardial effusion but did reveal a globally dilated and hypokinetic left ventricle.
A diagnosis of dilated cardiomyopathy secondary to a viral infection was made on the basis of the history. He was started on diuretics, enalapril, digoxin and aspirin. He clinically improved and was then started on a low dose of carvediolol before being discharged for follow up with cardiology.
Discussion
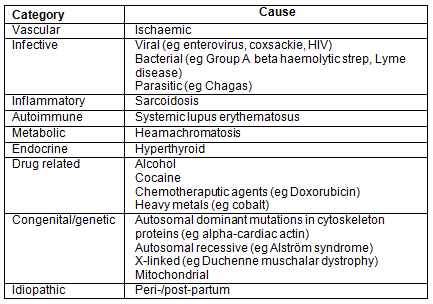
Cardiomyopathies are diseases of the cardiac muscle that lead to a deterioration in its function. The term encompasses a large number of conditions that have both intrinsic and extrinsic causes. In dilated cardiomyopathy, a portion of the ventricle expands leading to impaired systolic function and, as a consequence, further enlargement and remodelling of the heart driven by the release of hormones including catecholamines and angiotensin II3. A dilated ventricle requires more energy for effective contraction, as can be shown by Laplace's Law for the relationship between wall tension, pressure and radius of an expanding sphere4. Although an often overlooked diagnosis, it is relatively common within South Africa where it has been found in up to 10-17% of patients at post-mortem with cardiac-related causes of death, and makes up to 48% of admissions with heart failure1. There are many different causes and it is thought to be a combination of several insults that cause the initial damage (Table 1)5. Contributing factors include toxins and alcohol exposure, viral infections, pregnancy and genetic factors. Specific infections thought to be responsible include coxsackie B and enterovirus. In HIV positive individuals, non-viral infections may predominate and HIV itself may directly damage cardiac tissue3,5,6. Individuals with the HLA-DR1 and Dw10 halotypes appear to be at increased risk of developing dilated cardiomyopathy, lending further evidence for an immunologically mediated pathway to cardiac damage5. Familial cases are seen and are most commonly autosomal dominant and stem from a mutation in the gene coding for the cytoskeletal proteins of cardiac muscle.
Table 1: Causes of dilated cardiomyopathy according to a surgical sieve
Patients may present with a range of symptoms according to severity and duration of illness. Impaired left ventricular function may lead to cardiogenic shock and present with signs of congestive cardiac failure; for example, hypotension, elevated JVP, pulsatile liver, displaced and diffuse apex beat, third and fourth heart sounds, and bibasal crepitations. Pulmonary oedema and other acute signs mentioned are thought to be a result of increased hydrostatic pressure in the right side of the circulation as a result of the failing left ventricle. However, peripheral oedema, which is seen frequently in chronic sufferers, is thought to be a result of increased salt and fluid retention due to neurohormonal stimulation7. This physiological adaption takes time and may explain why the patient in this case had developed acute changes consistent with congestive cardiac failure but did not yet have any peripheral oedema. Heart murmurs may occur as a result of a disruption of the normal architecture of the heart; for example, mitral regurgitation. Shortness of breath can develop throughout the course of the disease due to fluid overload and may be accompanied by a pronounced P2 component to the second heart sound. Arrhythmias are common and patients may present with palpitations, syncope or even cardiac arrest. However, some patients may only present with vague constitutional symptoms, weight loss and fatigue.
Treatment in the acute setting is aimed at reducing the preload and afterload, and improving cardiac contractility. Relevant treatment options in the acute setting are discussed in the next section. Diuretics including furosemide and spironolactone, angiotensin converting enzyme inhibitors (ACE-i) and digoxin are currently the mainstay of long-term management8,9. The use of isosorbide dinatrate in combination with hydralazine has been used in patients intolerant to ACE-I and there is some evidence that their addition on top of standard treatments in patients with severe symptoms of congestive cardiac failure may improve mortality10. Amiodarone can be used in those who are prone to arrhythmias but consideration should be given to an implanted defibrillator. Betablockers should always be considered as they may counteract the affect of sympathetic activation on the remodelling of the heart. However, they should only be initiated once the patient is stable due to their negative ionotropic effects8.
Obstacles to treatment within a rural healthcare setting
Within a rural setting the problems of diagnosing and treating this condition are increased. Limited resources may make diagnosis difficult. Therefore, it is important to remember that the history and physical examination are paramount. As mentioned, signs of heart failure will help to differentiate a cardiac from a respiratory cause for acute onset shortness of breath. If available, a simple ultrasound scan of the heart can be very useful where diagnostic uncertainty remains. In dilated cardiomyopathy, the left ventricle will appear dilated and globally dyskinetic. Ultrasound will also help to rule out a pericardial effusion, an important differential for a dilated cardiac shadow on a chest x-ray.
Due to cultural factors and poor infrastructure, patients in a rural setting may present late. Patients are frequently malnourished and suffer from other serious co-morbidities such as HIV, giving them limited physiological reserve to cope with acute cardiac problems. As such, patients may be profoundly unwell at the time of presentation. Quick and accurate diagnosis is therefore imperative. Treatment in the acute phase is aimed at offloading the excess preload placed on the left ventricle. Intravenous furosemide is commonplace and can be used in conjunction with nitrates and small doses of morphine to achieve this end. Although infusion pumps of nitrates may not be feasible, the use of readily available sublingual alternatives can be very effective11. Patients should also be anti-coagulated, especially during the acute stage when bed rest is advocated and at discharge due to the increased chance of thrombus formation in the poorly contractile ventricle12. Within a rural setting, warfarinisation may be challenging as poor compliance and limited follow up capacity may mean the risks of bleeds outweigh the benefits. In this case, regular aspirin was used as an alternative on the advice of the local cardiology team. Intravenous fluid should be avoided unless the patient is hypotensive and then only used as a small bolus13. At all times it is important to recognise local limitations and early consultation with a referral centre is essential, especially in those patients refractory to treatment. Early initiation of an ACE-i has been shown to be beneficial providing the patient's systolic blood pressure is above 80 mmHg in the absence of any postural symptoms8. This and other long-term treatments discussed should be started at a low dose and gradually titrated up.
Primary health care provision, as well as poor patient understanding and attendance, may hamper long-term follow up and treatment. Patient education is of vital importance here. Having a basic understanding of their diagnosis and key warning symptoms will aid follow up and ensure early re-presentation. Translators and pictographic representations may be helpful. If possible, follow up care at a specialist centre should be arranged. Regular weight and renal function monitoring in the community ensure optimal diuresis is achieved.
Conclusion
Cardiomyopathy is a significant cause of morbidity and mortality throughout Africa. Diagnosis and treatment can often be challenging, especially within a rural setting. Focusing on history and examination and the use of simple, readily available treatments can help to overcome this. Prognosis in patients with dilated cardiomyopathy is variable and 5 year survival is estimated at 40%14. This has improved but continues to rely on prompt diagnosis and appropriate treatment. It is suggested that a diagnosis of dilated cardiomyopathy be considered in all patients presenting with symptoms of fatigue and dyspnoea and signs of cardiac failure.
References
1. Sliwa K, Damasceno A, Mayosi B. Heart disease in Africa. Epidemiology and etiology of cardiomyopathy in Africa. Circulation 2005; 112: 3577-3583.
2. Sunderman T, Olithselvan M, Sethuraman KR, Narayan KA. Scorpion envenomation as a risk factor for development of dilated cardiomyopathy. Journal of the Association of Physicians of India 1999; 47(11): 1047-1050.
3. Jameson JN, Kasper DL, Harrison TR, Braunwald E, Fauci AS, Hauser SL et al. Harrison's principles of internal medicine, 16th edn. New York: McGraw-Hill Medical Publishing Division, 2005.
4. Lilli A, Baratto MT, Del Meglio J, Chioccioli M, Magnacca M, Svetlich C et al. Three-dimensional simultaneous strain volume analysis describes left ventricular remodelling and its progression: a pilot study. European Heart Journal - Cardiovascular Imaging 2011; 12(7): 520-527.
5. Kumar V, Abbas AK, Mitchell R, Fausto N. Robbins basic pathology, 8th edn. Philadelphia: Saunders, 0000.
6. Barbaro G, Di Lorenzo G, Grisorio B, Barbarini G. Incidence of dilated cardiomyopathy and detection of HIV in myocardial cells of HIV-positive patients. New England Journal of Medicine 1998; 339: 1093-1099.
7. Navas JP, Martinez-Maldonado M. Pathophysiology of edema in congestive cardiac failure. Heart Disease and Stroke 1993; 2(4): 325-329.
8. Elliott P. Diagnosis and management of dilated cardiomyopathy. Heart 2000; 84: 106.
9. Massen E. Current treatment of dilated cardiomyopathy. Journal of the Texas Heart Institute 1991; 18(1): 41-49.
10. Cohn JN, Archibald DG, Ziesche S, Franciosa JA, Harston WE, Tristani FE et al. Effect of vasodilator therapy on mortality in chronic congestive cardiac failure. Results of a Veterans Administration Cooperative Study. New Engand Journal of Medicine 1986; 314(24): 1547-1552.
11. Antman E. Cardiovascular therapeutics: a companion to Braunwald's Heart Disease. Philadelphia: Elsevier Health Sciences, 2007; 79.
12. Bakali A, Georgievska-Ismail L, Kocinal D, Musliu N, Krasniqi A, Pllana E. Prevalance of left chamber thrombi in patients with dilated left ventricle at sinus rhythm: the role of transesophageal echocardiography. Journal of Clinical Ultrasound. (Online) 2012. doi 10.1002/jcu21953.
13. Longmore M, Wilkinson IB, Davidson EH, Foulkes A, Mafi AR. Oxford handbook of clinical medicine, 8th edn. Oxford: Oxford University Press, 2010, 812-815.
14. Ogasawara S, Sekiguchi M, Hiroe M, Morimoto S, Hirosawa K. Prognosis of dilated cardiomyopathy. Heart and Vessels 1985; 1(2): 78-82.

