Many published studies have established the worse health status and outcomes of rural and remote populations1, due to socioeconomic inequalities and poorer living conditions, inadequate water supply, difficulties in healthcare access and opportunities, lack of systematic health policies or insufficient education of rural populations2-4. This universal problem has most often been reported in remote areas of Australia and Canada1,3. However some communities are experiencing the reverse trend, with worsening conditions among highly urban populations and improvements in more affluent and socioeconomically favourable rural areas, especially in the UK5.
Remote and isolated populations maintain their isolation in many ways, including geographical barriers, cultural practices, language, tribal or national identity, or religion6. These populations are very interesting to genetics researchers, due to several properties. First, the so-called 'founder effect' where a historical founding group of individuals or first settlers of an isolated population possesses unfavourable genetic variants which increase in prevalence over time. The second mechanism, 'genetic drift', is defined as change in the frequency of a gene variant (allele) in a population, due to random sampling7. This mechanism can change allele frequencies or completely remove a gene variant from a population7. The third is increased homozygosity in the isolated populations, due to reduced partner choice6. All three mechanisms lead to substantial reduction in diversity in isolated populations and thus cause differences in the genetic structure of isolated populations compared with the main population or population of origin8,9.
Croatian island isolates were initially involved in genetics research studies in 1999, when an initial series of results reported an increased prevalence of cancer in some of the islands10,11. In addition, the populations of some of the islands were also shown to have increased prevalence of other diseases, including nephrolithiasis12, learning disabilities13, hypertension14,15, hyperlipidaemia16, biochemical markers of haemostasis17 or osteoporosis18. Subsequent studies aimed to understand the main health determinants in these populations6,19,20, but also converted this entire resource into a large-scale biobank capable of producing top-end genetics research results21-25.
The aim of this study was to investigate whether genetics may be considered an additional risk factor for health in isolated and remote populations, compared with their populations of origin. For this purpose, the incidence of hyperuricaemia, gout and osteoarthritis in the isolated communities on the islands of Vis and Korcula and coastal city of Split were compared.
Setting
The 10 001 Dalmatians program was initiated in 1999, and the first field work took place in 20016,19. The initial project goals were to investigate the main determinants of health in isolated populations and to understand the population genetics of the islands. Over time, the resource was developed and recently converted to a large-scale genetic resource, entitled 'The 10,001 Dalmatians'. The project currently consists of more than 4500 subjects from five isolated populations and the coastal city of Split.
All project subjects were measured for a number of clinically relevant phenotypic traits, including digital ECG, spirometry, anthropometry, eye examinations, retinal photography, hearing threshold, and biochemical and urine analyses. Furthermore, measurements included some novel phenotypes and biomarkers20,26-29, producing a nearly unique resource with over 1600 measured traits and available genotyping information. All subjects involved were aged over 18 years, and the entire project was approved by the appropriate Ethical boards in both Croatia and UK.
Sub-samples
Three sub-samples of the entire resource, obtained according to sufficient size and the existence of appropriate genotyping, were from: the island of Vis, the island of Korcula and the city of Split.
The island of Vis: The first sub-sample (N=1025) was recruited on the island of Vis, one of the most remote islands with a standing population of over 1000 inhabitants. These subjects were initially invited and involved in the project in 2003 and 2004, and later re-visited in 2007 and 2011. Most subjects from this sub-sample originated from the two main settlements on the island, Vis and Komiza, while a lesser number of subjects wase drawn from the central settlements. Population-based sampling was employed with postal invitations sent to all island inhabitants, explaining the details of the project and the benefits of participation (a number of clinically relevant traits were measured by trained measurers, and participants were immediately given personal results and interpretations).
The island of Korcula: Subjects for the second sub-sample (N=969) came from the island of Korcula, and participated in the project in 2007. Recruitment mainly focused on the eastern part of the island, including the settlements Korcula, Zrnovo, Lumbarda and Racisce. A similar sampling frame was used, with the addition of email invitations to local activity groups, and radio and local presentations to ensure maximum participation. The island of Korcula has a well established healthcare system, which includes a number of specialist and consultant visits organized from the mainland on a regular basis, offering better health opportunities and more accessible health care than on the island of Vis.
The city of Split: The third sub-sample (N=1012) was recruited from the city of Split in the same way in 2008 and 2009. The majority of the Split sample originated from highly urbanised areas, and this sample was therefore considered to be the urban control for the two island sub-samples.
Measurements
All three sub-samples were measured using the same approach and standard operating procedures. Laboratory measurements were performed at the same laboratory, ensuring the sub-samples were directly comparable in terms of the measured traits.
Information from the subjects' medical histories (diagnosis of gout and osteoarthritis), and biochemical measurements (serum uric acid and creatinine) were used in this study. Uric acid was converted to a binary variable and hyperuricaemia, defined as uric acid >403 mmol/L, was considered to be the upper limit of the referent laboratory range (Labor central laboratory, Zagreb, Croatia). In addition, data on BMI (obtained by anthropometric measurement) and several lifestyle indicators were included. Lifestyle indicators included self-reported intake of meat, fish, legumes and alcohol. An index was developed for meat intake, containing seven different types of meat (poultry, pork, beef, veal, meat derivates, insides and preserved meat). A similar index containing four items was developed for fish intake (blue fish, white fish, seafood, fish derivates). Legume intake was assessed from a single survey question regarding frequency of intake. Alcohol intake was based on three different alcohol types (beer, wine and spirits) to provide information on average daily alcohol intake.
Phenotypic measurements of genetic information was also used for a subset of subjects. The sample from Vis island was genotyped with Illumina HumanHap 300 v1 (Illumina Inc; San Diego, CA, USA), with 317 000 single-nucleotide polymorphisms (SNP), while Korcula and Split were genotyped with Illumina CNV370 with 346 000 SNPs. For the purposes of this study only information from three loci was used. All three loci are from the SLC2A9 gene on chromosome 4, which is considered to be the main regulator of serum uric acid and involved in determination of risk for development of gout (rs6449213, rs1014290 and rs737267)30.
Statistical analysis
In the descriptive part of the study, percentages and absolute numbers were used as categorical variables, while means and standard deviations were numeric. Since three sub-samples were compared, analysis of variance was the main analytic approach for numerical data, while the χ2 test was used for categorical data. The same test was also used for allele frequencies calculation and analyses. In order to allow direct comparison of the study results across sub-samples, direct age standardization of gout, hyperuricaemia and osteoarthritis prevalence was performed according to the new European population (based on Health for All database information31). In addition, a logistic regression was used to adjust for the commonest confounders.
A total of three separate models were made, for hyperuricaemia, gout and osteoarthritis. A set of eight predictors were used in all three models, with age and gender, thus providing adjusted odds ratio and 95% confidence intervals estimation. Analyses were performed using IBM SPSS v19 (IBM Corporation Armonk, NY) with significance set at p<0.05.
Ethics approval
This research was approved by Ethical Board of the Medical School, University of Split (#2181-198-03-04/10-11-0008).
This study included a total of 3006 participants in three cohorts: Vis (N=1025; 34.1%), Korcula (969; 32.2%) and Split (1012; 33.7%). The age and sex structure of the three sub-samples differed (Table 1). Similarly, the prevalence of gout, hyperuricaemia and arthritis were also different, with even stronger differences recorded in the age-standardized prevalence (Table 1). The mean serum uric acid showed a similar trend with the highest values in the population of Vis, intermediate in the population of Korcula and the lowest values in the population of the coastal city of Split (Table 1). A comparison of basic socioeconomic characteristics indicated that the Vis sample was least favourable on this parameter, while the Split sample was most favourable (Table 1). Multivariate analysis for hyperuricaemia, gout and osteoarthritis indicated that those on the most isolated island, Vis, had significantly higher odds for hyperuricaemia, compared with inhabitants of the city of Split, similar (insignificant) odds for gout, and increased odds for osteoarthritis (Table 2). The less isolated island of Korcula had higher odds only for osteoarthritis, compared with Split city (Table 2).
Analysis of allele frequencies of the three loci within SLC2A9 indicated significant differences across the study sample, with the highest proportion of deleterious genotypes (rare homozygotes) in Vis, an intermediate proportion in Korcula and the lowest in Split (Table 3).
Table 1: Demographics, health status and behavioural pattern breakdown
within the studies samples from the Croatian islands of Vis and Korcula and coastal city of Split
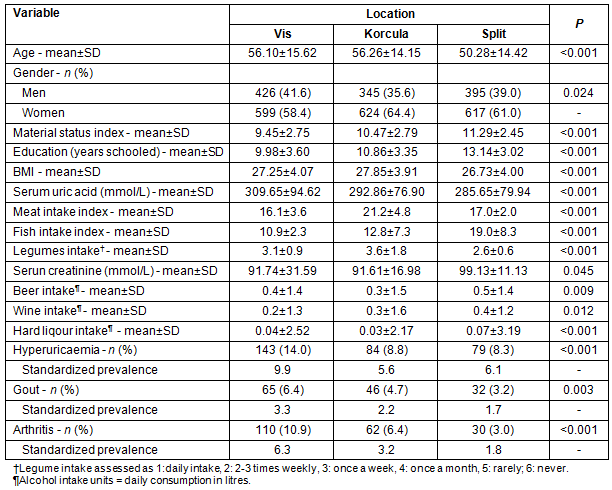
Table 2: Predictors of the hyperuricaemia, gout and osteoarthritis in logistic regression models
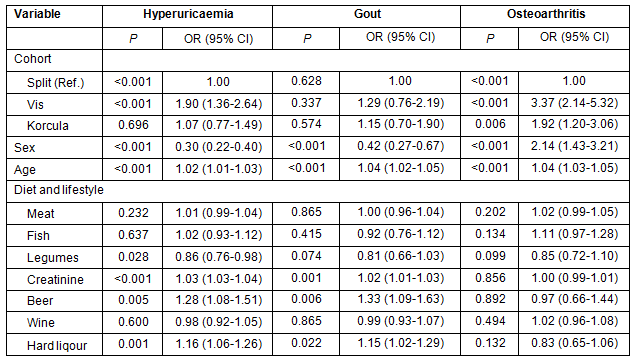
Table 3: The allele frequencies of the three loci from SLC2A9 gene in samples
from the Croatian islands of Vis and Korcula and coastal city of Split
Discussion
The results of this study show that isolated and remote populations may suffer from an unfavourable genetic structure, which may contribute to poor health status. This finding was demonstrated using the example of the prevalence of gout, hyperuricaemia and osteoarthritis, and supported by the allele frequencies of SLC2A9, a major determinant of urate metabolism30,32-36. Urate metabolism disorders in isolated and remote populations are prevalent not only in Croatia12, but also in Iceland37, Fiji38 and the Canary islands39. These findings suggest rather widespread existence of urate-disrupting mechanism in isolated populations. While this present a promising research target in the study of genetics, it has completely different implications for public health. The existence of such a mechanism demonstrates there can be no 'one size fits all' policy in addressing the healthcare needs of isolated and remote populations, and that each population requires detailed investigation, needs assessment, a specific approach and targeted activities aimed at disease-burden reduction.
As well as the effects of founder and genetic drift on isolated populations, genetic theory predicts that isolation will also increase the amount of homozygosity, due to reduced mate choice or prevalent consanguinity6. These will increase the chances of the occurrence of a range diseases in affected populations, from early-onset disorders to the complex diseases of adult life6,10,40,41. The accumulation of rare recessive alleles will lead to an increase in the genetic load and increased prevalence of deleterious alleles in the population. In the present study, the highest prevalence of deleterious homozygous for the three loci was reported in Vis, also the most genetically and geographically isolated island8,42. With a reduction in isolation and increased population mobility and mixing, allele frequencies tend to change and reduce the degree of homozygosity, which also increases population diversity. This was clearly demonstrated in a lower frequency of the most deleterious genotypes in Korcula and the lowest frequency in Split, which has a much more admixed population than either of the islands. This finding suggests that there is a dose-effect relationship between the amount of genetic isolation and disease burden, and that the smallest remote and isolated populations could have the highest disease burdens.
This and the other mechanisms that define the genetic structure of isolated populations could also be responsible for some findings in other populations. For example, an increased prevalence of metabolic and renal diseases in an Australian Aboriginal population43 could, at least partly, also be attributed to the genetic isolation of this population44. Other studies have also reported worse health status in isolates than in their populations of origin9,16,45-47. This finding becomes interesting if the main result is inverted: if isolated populations have worse health status, then the 'break-up' of isolation through urbanization should have beneficial effects on health. This notion was indeed supported by some studies, suggesting that increased population mobility and mixing could be very important mechanisms contributing to secular trends and improvements in life expectancy, intelligence and health48-50.
The Croatian island isolates have an added interest for public health research, due to the widely reported better health status of coastal Mediterranean populations compared with their continental counterparts51. Such differences have been reported for the last 50 years, commonly referring to unhealthy behavioural patterns, especially a fat-rich diet in continental locations and commoner adherence to healthier dietary patterns in the coastal population51. However, studies have suggested that this might not be true for the islands, where much worse health indices were recorded, including higher a prevalence of incident hypertension, unregulated hypertension and overweight than in continental locations of the country52. The results of this study reflect the less favourable situation of the more remote island populations, supporting the idea that islanders require more specific and targeted public health approaches. This finding was confirmed by the multivariate analysis, where even after adjustment some results retained their significance. This was especially true for osteoarthritis, which was found to have a much higher disease burden in the isolated populations. A possible explanation could lie in the way of life, where more agricultural activities and greater physical activity was likely among islanders, compared with those in the city of Split. Additionally, the more isolated island of Vis had greater odds for hyperuricaemia, thus also confirming the previous finding and supporting the idea of an increased burden of disease in more genetically isolated populations.
Limitations
The limitations of this study include the possibility that these results could be limited to the Croatian population and may not be replicated elsewhere. However, the similarity of the results of this study with those previously published suggests that similar mechanisms also could at work in different populations.
While researchers in genetics have used this information in numerous studies previously, genetics has only recently begun to enter the field of public health and health policy. The findings from this study are especially interesting for rural and remote policy-makers. Until now, most policies for the improvement of rural and remote health have involved interventions in access to, and the organization of health care or mobility, without consideration of a population's genetic structure. Based on the results reported here and other studies, it is now evident that genetics will not only change clinical medicine and introduce a personalized approach, but it will also have a substantial effect on public health in the future. Future policies for remote and isolated populations should therefore consider and include at least the estimated amount of genetic isolation as a potential factor that can affect population health.
Attempts to provide equal health opportunities in genetically isolated populations may be even more demanding than was previously believed. If such populations are provided with even the same level of health care as their population of origin, this may not be sufficient to offset their genetic load. Paradoxically, in order to achieve equal health opportunities across the entire population, it appears that genetically isolated populations require an even greater level of basic research and needs assessment before any intervention and healthcare improvement can be deployed .
Acknowledgements
This work was supported by grants from the Medical Research Council UK and the Ministry of Science, Education and Sports of the Republic of Croatia.
References
1. Ryan-Nicholls K. Health and sustainability of rural communities. Rural and Remote Health 4: 242. (Online) 2004. Available www.rrh.org.au (Accessed 16 January 2013).
2. Polasek O, Kolcic I, Voncina L, Strnad M, Vuletic S, Kern J. Breast, colon, and prostate screening in the adult population of Croatia: does rural origin matter? Rural and Remote Health 7: 749. (Online) 2007. Available www.rrh.org.au (Accessed 16 January 2013).
3. McDonald E, Bailie R. Hygiene improvement: essential to improving child health in remote Aboriginal communities. Journal of Paediatrics and Child Health 2010; 46: 491-496.
4. Mukhopadhyay A. Tackling health determinants in rural India: the KHOJ initiative. Global Health Promotion 2010; 17: 69-72.
5. Watt IS, Franks AJ, Sheldon TA. Health and health care of rural populations in the UK: is it better or worse? Journal of Epidemiology and Community Health 1994; 48: 16-21.
6. Rudan I. Health effects of human population isolation and admixture. Croatian Medical Journal 2006; 47(4): 526-531.
7. Masel J. Genetic drift. Current Biology 2011; 21: 837-838.
8. Vitart V, Biloglav Z, Hayward C, Janicijevic B, Smolej-Narancic N, Barac L et al. 3000 years of solitude: extreme differentiation in the island isolates of Dalmatia, Croatia. European Journal of Human Genetics 2006; 14(4): 478-487.
9. Igl W, Johansson A, Gyllensten U. The Northern Swedish Population Health Study (NSPHS) - a paradigmatic study in a rural population combining community health and basic research. Rural and Remote Health 10: 1363. (Online) 2010. Available www.rrh.org.au (Accessed 16 January 2013).
10. Rudan I. Inbreeding and cancer incidence in human isolates. Human Biology 1999; 71: 173-187.
11. Rudan I. Ancestral kinship and cancer in Lastovo island. Human Biology 2001; 73: 871-884.
12. Rudan I, Padovan M, Rudan D, Campbell H, Biloglav Z, Janicijevic B et al. Inbreeding and nephrolithiasis in Croatian island isolates. Collegium Antropologicum 2002; 26(1): 11-21.
13. Rudan I, Rudan D, Campbell H, Biloglav Z, Urek R, Padovan M et al. Inbreeding and learning disability in Croatian island isolates. Collegium Antropologicum 2002; 26(2): 421-428.
14. Rudan I, Smolej-Narancic N, Campbell H, Carothers A, Wright A, Janicijevic B et al. Inbreeding and the genetic complexity of human hypertension. Genetics 2003; 163(3): 1011-1021.
15. Rudan I, Rudan D, Campbell H, Carothers A, Wright A, Smolej-Narancic N et al. Inbreeding and risk of late onset complex disease. Journal of Medical Genetics 2003; 40(12): 925-932.
16. Polasek O, Kolcic I, Smoljanovic A, Stojanovic D, Grgic M, Ebling B et al. Demonstrating reduced environmental and genetic diversity in human isolates by analysis of blood lipid levels. Croatian Medical Journal 2006; 47(4): 649-655.
17. Pulanic D, Polasek O, Petrovecki M, Vorko-Jovic A, Pericic M, Barac Lauc L et al. Effects of isolation and inbreeding on human quantitative traits: An example of biochemical markers of hemostasis and inflammation. Human Biology 2008; 80(5): 513-533.
18. Rudan I, Skaric-Juric T, Smolej-Narancic N, Janicijevic B, Rudan D, Klaric IM et al. Inbreeding and susceptibility to osteoporosis in Croatian island isolates. Collegium Antropologicum 2004; 28(2): 585-601.
19. Rudan I, Biloglav Z, Vorko-Jovic A, Kujundzic-Tiljak M, Stevanovic R, Ropac D et al. Effects of inbreeding, endogamy, genetic admixture, and outbreeding on human health: A "1001 dalmatians" study. Croatian Medical Journal 2006; 47(4): 601-610.
20. Rudan I, Marusic A, Jankovic S, Rotim K, Boban M, Lauc G et al. "10001 Dalmatians: " Croatia launches its national biobank. Croatian Medical Journal 2009; 50(1): 4-6.
21. Ehret GB, Munroe PB, Rice KM, Bochud M, Johnson AD, Chasman DI et al. Genetic variants in novel pathways influence blood pressure and cardiovascular disease risk. Nature 2011; 478(7367): 103-109.
22. Lango Allen H, Estrada K, Lettre G, Berndt SI, Weedon MN, Rivadeneira F et al. Hundreds of variants clustered in genomic loci and biological pathways affect human height. Nature 2010; 467(7317): 832-838.
23. Repapi E, Sayers I, Wain LV, Burton PR, Johnson T, Obeidat M et al. Genome-wide association study identifies five loci associated with lung function. Nature Genetics 2010; 42(1): 36-44.
24. Speliotes EK, Willer CJ, Berndt SI, Monda KL, Thorleifsson G, Jackson AU et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nature Genetics 2010; 42(11): 937-948.
25. Sotoodehnia N, Isaacs A, de Bakker PIW, Dorr M, Newton-Cheh C, Nolte IM et al. Common variants in 22 loci are associated with QRS duration and cardiac ventricular conduction. Nature Genetics 2010; 42(12): 1068-1076.
26. Elks CE, Perry JR, Sulem P, Chasman DI, Franceschini N, He C et al. Thirty new loci for age at menarche identified by a meta-analysis of genome-wide association studies. Nature Genetics 2010; 42: 1077-1085.
27. Knezevic A, Polasek O, Gornik O, Rudan I, Campbell H, Hayward C et al. Variability, Heritability and Environmental Determinants of Human Plasma N-Glycome. Journal of Proteome Research 2009; 8(2): 694-701.
28. Knezevic A, Gornik O, Polasek O, Pucic M, Redzic I, Novokmet M et al. Effects of aging, body mass index, plasma lipid profiles, and smoking on human plasma N-glycans. Glycobiology 2010; 20: 959-969.
29. Pucic M, Knezevic A, Vidic J, Adamczyk B, Novokmet M, Polasek O et al. High throughput isolation and glycosylation analysis of IgG-variability and heritability of the IgG glycome in three isolated human populations. Molecular Cell Proteomics 2011; 10: M111.010090.
30. Vitart V, Rudan I, Hayward C, Floyd J, Knott S, Kolcic I et al. SLC2A9 is a newly identified urate transporter influencing serum urate concentration, urate excretion and gout. Nature Genetics 2008; 40(4): 437-442.
31. UN. Health for All Database. (Online) 2012. Available: www.euro.who.int/hfadb (Accessed 16 January 2013).
32. Polasek O, Gunjaca G, Kolcic I, Zgaga L, Dzijan S, Smolic R et al. Association of nephrolithiasis and gene for glucose transporter type 9 (SLC2A9 ): study of 145 patients. Croatian Medical Journal 2010; 51: 48-53.
33. Rudan D, Polasek O, Kolcic I, Rudan I. Uric acid: the past decade. Croatian Medical Journal 2010; 51: 1-6.
34. Gunjaca G, Boban M, Pehlic M, Zemunik T, Budimir D, Kolcic I et al. Predictive value of 8 genetic loci for serum uric acid concentration. Croatian Medical Journal 2010; 51: 23-31.
35. Polasek O, Jeroncic I, Mulic R, Klismanic Z, Pehlic M, Zemunik T et al. Common variants in SLC17A3 gene affect intra-personal variation in serum uric acid levels in longitudinal time series. Croatian Medical Journal 2010; 51: 32-39.
36. Merriman TR, Dalbeth N. The genetic basis of hyperuricaemia and gout. Joint, Bone and Spine 2011; 78: 35-40.
37. Edvardsson V, Elidottir H, Indridason OS, Palsson R. High incidence of kidney stones in Icelandic children. Pediatric Nephrology 2005; 20(7): 940-944.
38. Holmes G. Urinary calculi in Fiji Indians. The curry kidney. Medical Journal of Australia 1971; 2: 755-756.
39. Melian JS, Nieto VG, Sosa AM. Inheritance and prevalence of hypercalciuria in the children from the island of La Gomera. Nefrologia 2000; 20(6): 510-516.
40. Campbell H, Carothers AD, Rudan I, Hayward C, Biloglav Z, Barac L et al. Effects of genome-wide heterozygosity on a range of biomedically relevant human quantitative traits. Human Molecular Genetics 2007; 16(2): 233-241.
41. Polasek O, Leutenegger AL, Gornik O, Zgaga L, Kolcic I, McQuillan R et al. Does inbreeding affect N-glycosylation of human plasma proteins? Molecular Genetic Genomics 2011; 285: 427-432.
42. Polasek O, Hayward C, Bellenguez C, Vitart V, Kolcic I, McQuillan R et al. Comparative assessment of methods for estimating individual genome-wide homozygosity-by-descent from human genomic data. BMC Genomics 2010; 11: 139.
43. McDonald S, Maguire G, Duarte N, Wang XL, Hoy W. Homocysteine, renal disease and cardiovascular disease in a remote Australian Aboriginal community. Internal Medical Journal 2005; 5(35): 289-294.
44. Huoponen K, Schurr TG, Chen Y, Wallace DC. Mitochondrial DNA variation in an aboriginal Australian population: evidence for genetic isolation and regional differentiation. Human Immunology 2001; 62: 954-969.
45. Igl W, Johansson A, Wilson JF, Wild SH, Polasek O, Hayward C et al. Modeling of environmental effects in genome-wide association studies identifies SLC2A2 and HP as novel loci influencing serum cholesterol levels. PLoS Genetics 2010; 6: e1000798.
46. Pattaro C, Aulchenko YS, Isaacs A, Vitart V, Hayward C, Franklin CS. Genome-wide linkage analysis of serum creatinine in three isolated European populations. Kidney International 2009; 76: 297-306.
47. McWhirter RE, McQuillan R, Visser E, Counsell C, Wilson JF. Genome-wide homozygosity and multiple sclerosis in Orkney and Shetland Islanders. European Journal of Human Genetics 2012; 20: 198-202.
48. Rudan I, Carothers AD, Polasek O, Hayward C, Vitart V, Biloglav Z et al. Quantifying the increase in average human heterozygosity due to urbanization. European Journal of Human Genetics 2008; 16(9): 1097-1102.
49. Mingroni MA. Resolving the IQ paradox: heterosis as a cause of the Flynn effect and other trends. Psychology Review 2007; 114(3): 806-829.
50. Mingroni MA. The secular rise in IQ: Giving heterosis a closer look. Intelligence 2004; 32(1): 65-83.
51. Bergman Markovic B, Vrdoljak D, Kranjcevic K, Vucak J, Kern J, Bielen I et al. Continental-Mediterranean and rural-urban differences in cardiovascular risk factors in Croatian population. Croatian Medical Journal 2011; 52: 566-575.
52. Kolcic I, Biloglav Z, Zgaga L, Jovic AV, Curic I, Curic S et al. Prevalence of increased body weight and hypertension in the population of Croatian mainland and Adriatic Islands--are islanders really healthier? Collegium Antropologicum 2009; 33(Suppl1): 135-140.



