A marked resurgence of malaria in the 1990s, even in countries where DDT was previously used for various purposes, led to a renewed interest in DDT use as an indoor residual spray1,2. While still banned or severely restricted in western countries, it was made available to the global market in 20013-5. This demand for it was premised on its greater cost effectiveness and efficacy when compared with insecticide-treated bednets (ITNs) and alternatives like pyrethroids6. However, concerns about the safety or the chronic health effects from the continued use of this persistent chemical and its metabolite, dichlorodiphenyldichloroethylene (DDE), have generated much debate over the years4,7-10. Coupled with this is the seriously increasing resistance of mosquitoes to DDT11,12 and increased likelihood of users not adhering to recommended dosage as a consequence.
Having searched the Cochrane Database of Systematic Reviews (CDSR), Database of Abstracts of Reviews of Effects (DARE) and Database of Promoting Health Effectiveness Reviews (DoPHER), we ascertained that there was no pre-existing or ongoing systematic review addressing the chronic adverse effects of long-term exposure of children less than 19 years in places where DDT is used solely or mostly for malaria control. Due to this, the ongoing debates and the unclear evidence about cumulative effects of DDT on child health13, this review sought to identify, appraise and synthesise evidence about the chronic adverse effects of prolonged exposure to DDT on children of 0-18 years, in areas where IRS is practised, in order to contribute to informing policy decisions globally.
The protocol14 for this review is available from the corresponding author. A widely accepted framework for standard systematic review prepared by the Centre for Reviews and Dissemination (CRD)15 was chosen for the review design and no ethics approval was required.
Study inclusion and exclusion criteria
The PICOS (Population-Intervention-Comparator-Outcome-Study design) framework recommended by the CRD15 was used to formulate the eligibility criteria for the review. No 'comparator' was set for this study because of the focus of the review. The framework, being flexible, was therefore modified into PIOS, which is also adequately tested16.
As for the 'study design' component, the methodological challenge of combining results from experimental studies with observational ones was concluded to be balanced out in that the usefulness of randomised controlled trials (RCTs) as sources of adverse effects data is limited by their strict eligibility criteria (reducing generalisability), short period of follow-up, sample sizes that may not be enough to detect associations, and the fact that most of them report adverse effects as the secondary objective of study15,17.
The final eligibility criteria set at the end of the scoping study were as follows.
Inclusion: children, 0-18 years; use of DDT in IRS for malaria vector control; outcomes being chronic adverse effects which are (i) reproductive disorders, (ii) endocrine disorders, (iii) neurodevelopmental disorders, (iv) asthma, (v) cancers, (vi) teratogenic effects (congenital abnormalities), (vii) low birthweight, (viii) immune disorders; studies published in English between1991 and 2011; RCTs, quasi-RCTs, cohort, cross-sectional and case-control studies.
Exclusion: people 19 years and older; animals; agricultural or illegal use of DDT in pest control; other malaria control measures; cost effectiveness, efficacy and anopheline resistance measures; case reports and case series.
Search strategy
The initial search strategy developed using concepts identified during the literature review, prior knowledge of abbreviations and synonyms relevant to main concepts, was piloted on EMBASE, MEDLINE and PubMed by AO to identify limitations and then reviewed by both investigators. Search terms were used for the PIO components while for the 'study design' component manual sorting was resorted to in order to avoid leaving out relevant studies. The search and study selection summary are provided in Figure 1.
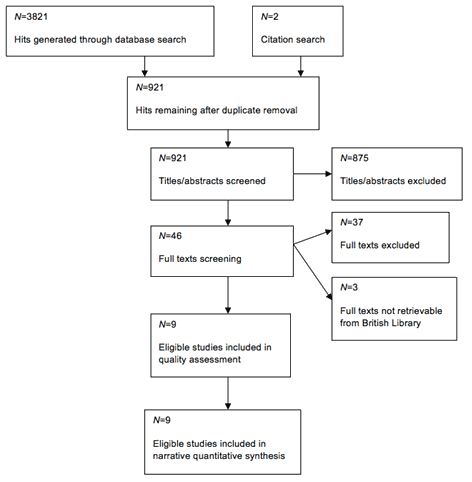 Figure 1: Study selection flow chart.
Figure 1: Study selection flow chart.
Data sources
Search was conducted on 27 databases (Table 1) and the search was managed using RefWorks. The last date of search was 30 April 2011 and access was done through the University of the West of England e-library. Table 2 presents a transcript of the free text search of EMBASE (because medical subject headings (MeSH) available for a few concepts were not very inclusive). In addition, reference lists of included studies were checked to further identify relevant studies. Citations of selected articles were searched in Web of Science, PubMed, ScienceDirect and Google Scholar. World Health Organization and System for Information on Grey Literature in Europe (SIGLE) websites were also searched. All the aforementioned generated no new eligible studies. E-mails were sent to corresponding authors of included studies, attaching a copy of the inclusion/exclusion criteria for the review and requesting them to suggest any other relevant study they knew that met the criteria. The British Library was also contacted for studies whose abstracts were indicative but whose full texts were not retrievable in any of the databases searched earlier. From databases searched, 3281 hits were generated out of which 176 were found potentially relevant.
Table 1: Databases searched
 Table 2: Transcript of search conducted in EMBASE
Table 2: Transcript of search conducted in EMBASE

Study selection
After screening the titles and/or abstracts, 121 studies were identified from all RefWorks-compatible databases; most studies appeared in more than one database. For databases incompatible with RefWorks, selection was done onsite and 55 articles were found. Fifty three of these were found already indexed in the RefWorks-compatible databases.
Having got rid of duplicates, retrieval of the full texts of the final 46 potentially relevant articles was embarked upon by AO. Out of the six that could not to be retrieved directly, only three were later retrievable on request through the British Library. On e-mail correspondence, a reply from an author in South Africa helped in re-consideration and eventual inclusion of two previously excluded studies.
Pilot: Study eligibility criteria were piloted on all of the 40 available studies. Seven of the studies were temporarily categorised as 'eligible' and then discussed by both reviewers to verify how stringently the criteria had been applied.
Final scrutiny: After proper re-scrutiny, only nine studies were included (five selected at first, then four added on reconsideration, as indicated earlier).
For the remaining 37 that were excluded (including two from Zetoc and the two from reference-tracing) and the three that could not eventually be retrieved through the British Library, a log of the reasons for exclusion of each of them was kept for future reference14 and is available from the corresponding author.
Data extraction and quality assessment
The initial data extraction form designed by us was piloted on four differently designed eligible studies. This was done alongside the quality assessment. Each data entry was done independently by AO with repeated checks to ensure correct interpretation and accuracy and thereafter moderated by DE. It enabled us to check for any necessary but missing criteria for the corresponding appraisal checklists of study types available. Our perceptions were largely similar and were resolved by consensus, where they differed.
Data were extracted on individual study characteristics including the inclusion and exclusion criteria, recruitment procedure, participant characteristics, baseline characteristics comparability, co-morbidities, number of participants enrolled, number of participants in analysis, number of withdrawals/exclusions/loss to follow-up and measurement of exposure. Data for outcome measurement and analysis included unit of assessment, outcome(s) reported, method and unit of measurement, definition of outcome, blinding of assessors, quality assurance, length of follow-up, number of times of follow-up and measurement, variables controlled for, number of participants in analysis and statistical techniques. The complete data extraction form and definitions of outcomes have been kept for future reference14 and are available from the corresponding author.
Amended versions of the validated guidelines developed for the Critical Appraisal Skills Programme (CASP) of the Public Health Resource Unit, United Kingdom, were chosen to appraise cohort and case-control studies14. Despite extensive efforts, no validated appraisal tool was found for cross-sectional studies. Therefore, a non-validated cross-sectional study appraisal checklist obtained online from Yeshiva University14,18 was adapted to suit the review, following a contact with an expert for guidance. Having removed irrelevant items, parameters added based on further guidance17,19,20 included authors' knowledge base of research done in the subject area, clear definition of study sample, provision for blinding in the assessment of outcomes, cognisance of and control for potential confounders, clear statement of measures of precision and the choice of appropriate statistics and tests.
The rationale for scores apportioned a priori by AO to each criterion in each guideline is provided in the Results section. While not standard practice to exclude studies on grounds of quality15, quality scores, both empirically and for specific criteria on the checklist, were used to assess the estimates of effects obtained from each study and determine their reliability.
Method of analyses and rationale
The relatively small number of eligible studies, the diversity of study designs and the methodological differences, even among the ones that examined similar outcome(s), precluded a meta-analysis15,16, with narrative synthesis being the only possibility.
From the nine studies included, four were cohort, one case-cohort and four cross-sectional. Seven of these eligible studies originated from Mexico (where DDT was used for IRS between 1991 and 2000), one from the US, although conducted for a Mexican population, and one from South Africa.
Summary of the quality assessment
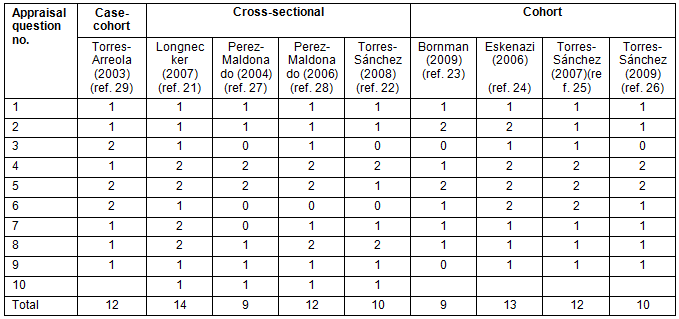
Five out of the nine included studies were of high quality while four others were moderate. During assessment, greater weights were allocated to sensitive items which focused on assessment of exposure, outcome measurement, adequate control for confounders, and clarity and accuracy of data presented. The breakdown of the quality assessment scores is provided in Table 3. For the quality assessment, all study designs had a maximum attainable score of 14. The studies were graded as high (12-14); moderate: (9-11) or poor (0-8). The guidelines used and the synopses of the critical appraisal14 are available from the corresponding author.
Table 3: Breakdown of scores for the quality assessment of included studies
Results of individual studies
Summaries of the results of individual studies are given in Table 4.
Table 4: Summary of study results
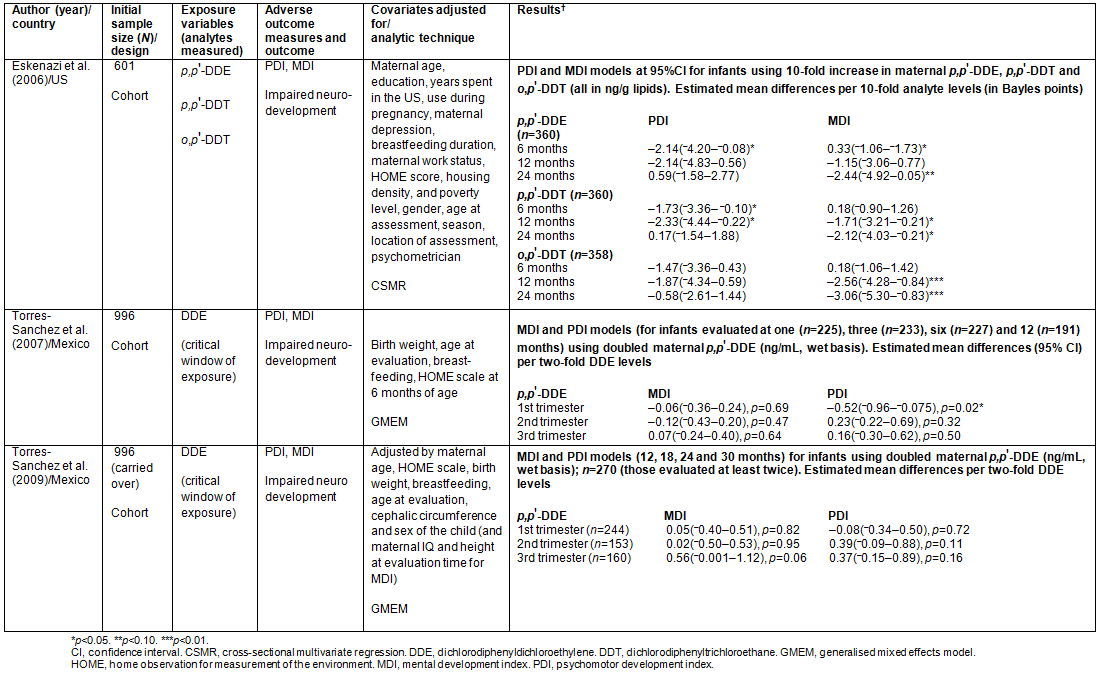
4a: Results from studies on impairment in infant neurodevelopment
 4b: Results from studies dealing with reproductive/endocrine outcomes and preterm birth
4b: Results from studies dealing with reproductive/endocrine outcomes and preterm birth
 4c: Results from studies on negative effects on immunity
4c: Results from studies on negative effects on immunity
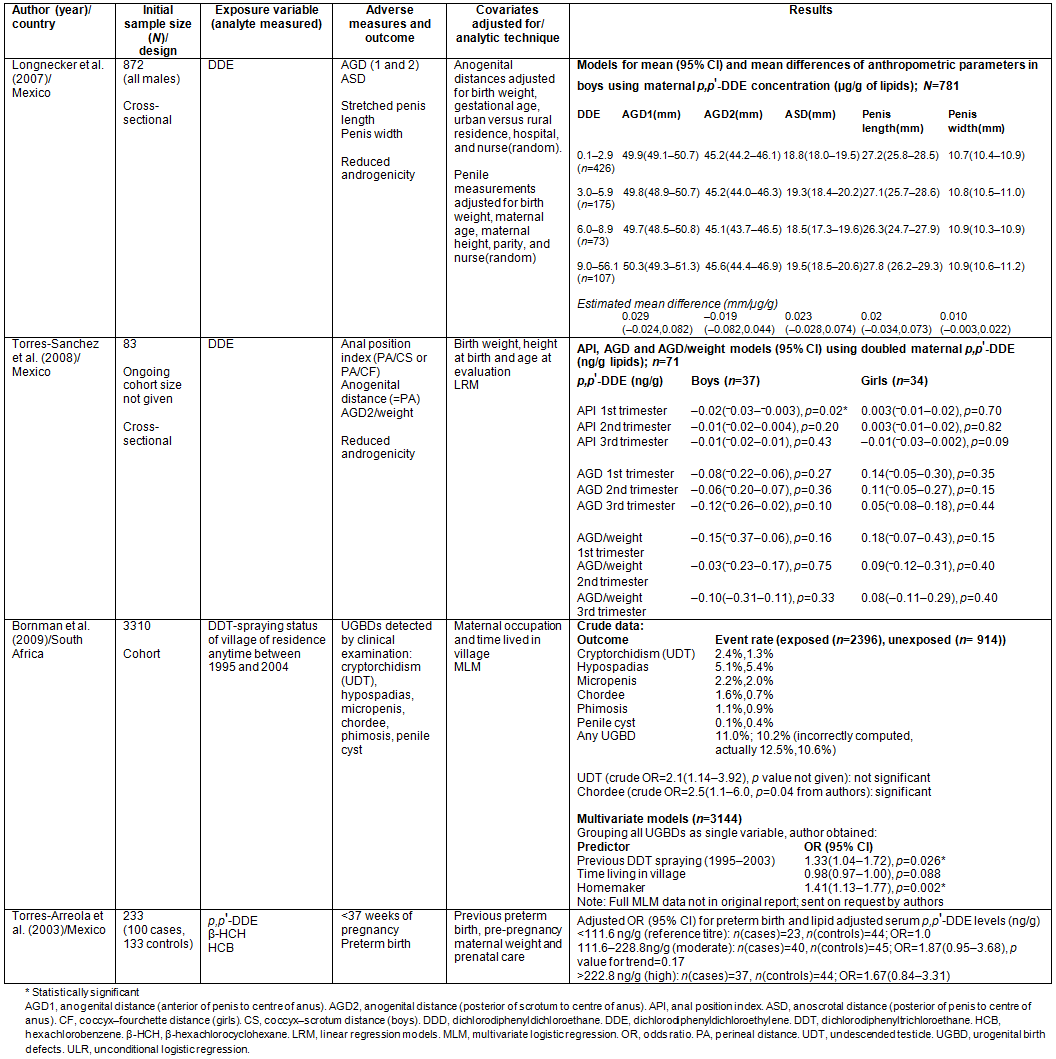
Studies on adverse endocrine/congenital outcomes (n=3): Longnecker et al. examined the hypothesis that in utero exposure to DDE decreases anogenital distance (AGD, a measure of foetal androgen action or endocrine disruptor activity) in newborn males (amidst other outcomes studied)21. Modelling DDE concentration as a continuous variable, no association was found between quantiles of DDE concentration and any of the outcomes.
In a cross-sectional study where Torres-Sanchez et al. sampled 71 infants (37 males and 34 females) from an ongoing perinatal cohort study on neurodevelopment that started in 2001 in Morelos, Mexico, they evaluated the potential anti-androgenic effects of prenatal p,p'-DDE and p,p'-DDT exposures on infant AGD, using anal position index (API)22. Maternal serum levels of DDT and DDE before pregnancy and during each trimester of pregnancy were used to 'characterise' in utero exposure. Analysis was conducted only for p,p'-DDE because very few mothers had their p,p'-DDT levels above detection limits. A significant reduction in API (estimated difference (mm)= -0.02; 95% confidence interval (CI)= -0.03, -0.003; p=0.02) in boys was found to be associated with a doubling of maternal serum DDE levels only during the first trimester of pregnancy. No associations were found between p,p'-DDE and the age-dependent parameters (AGD and AGD/weight). For girls, no significant association was found between the parameters and DDE levels in all three trimesters.
In a retrospective cohort study where Bornman et al. determined the association between six different external urogenital birth defects (UGBDs) in newborn boys and exposure to DDT from spraying in Limpopo, South Africa, both cryptorchidism and chordee were found associated with village having ever been sprayed (with wide confidence intervals)23. However, only chordee was significantly associated (crude odds ratio (OR)=2.5; 95% CI=1.1 to 6.0; p=0.04). Hypospadias, phimosis, micropenis and penile cyst showed no association.
In a multivariate logistic model involving 3144 out of the 3310 recruited, and where they grouped all six UGBDs as one variable (more detailed data provided on request14), authors claimed that the chance of male babies from villages sprayed between 1995 and 2003 having any UGBDs was 33% (OR=1.33; 95% CI=1.04 to 1.72) higher than with counterparts in villages never sprayed. Mother being a homemaker increased the risk of the child having a UGBD by 41% (OR=1.41; CI=1.13 to 1.77) compared to employed or studying mothers.
Studies on adverse neurodevelopmental outcomes (n=3): As part of a US multiyear birth cohort study that started in 1999/2000 with 601 mothers, a study was done by Eskenazi et al. on the effects of in utero exposure to two isomers of DDT (p,p'-DDT and o,p'-DDT) and p,p'-DDE on the neurodevelopment of infants of mothers recently immigrated from Mexico24. Maternal serum samples collected at the second or third trimester of pregnancy helped 'characterise' the foetal exposure having adjusted for lipid levels. The psychomotor development index (PDI) and the mental development index (MDI) of infants were measured on Bayles' scales at 6, 12 and 24 months, as indicators of neurodevelopment.
Geometric means of the serum levels of analytes (which were found to be strongly correlated, r= 0.8-0.9) were used. Statistically significant decreases of 1.73 and 2.14 points in the 6-month PDI score of infants were found to be associated with 10-fold increases in serum levels of p,p'-DDT (p< 0.05) and p,p'-DDE (p<0.05), respectively. However, only p,p'-DDT remained significantly associated with decreases in PDI scores (estimated difference (points)= -2.33 (95% CI= -4.44 to -0.22)) afterwards, at 12 months but not beyond. On a subgroup analysis (data not provided), authors claimed a negative association between o,p'-DDT level and PDI score for boys only, at 12 months (estimated difference (points)= -3.78; 95% CI= -7.47 to -0.09; p<0.01). This suggests that the non-association observed when combined was due to girls.
For MDI scores, no analyte demonstrated any association at 6 months. However, at 12 months, authors observed statistically significant decreases of 1.71 and 2.56 points in MDI scores associated with 10-fold increases in serum levels of p,p'-DDT (p<0.05) and o,p'-DDT (p< 0.01), respectively. By 24 months, 10-fold increases in serum levels of p,p'-DDT, o,p'-DDT and p,p'-DDE were found to be associated with decreases of 2.12 (p<0.05), 3.06 (p<0.01) and 2.44 (p<0.10) points, respectively, in MDI scores. Noteworthily, a consistent negative trend, as age increased, was found with mental development.
Among covariates adjusted for, breastfeeding was the only correlate found to be significantly positively associated with 12- and 24-month MDI scores whether DDT/DDE levels were kept in the model or not. Each month of breastfeeding was associated with increases of approximately 0.20 points in MDI scores at 12 and 24 months (p<0.05).
Torres-Sanchez et al., within a birth cohort, assessed the critical window of prenatal DDE exposure (for foetuses of mothers not occupationally exposed to DDT) and the effect it may have on PDI and MDI of infants in the first year of life25. To this effect, maternal serum levels of DDE before and during each trimester of pregnancy were measured and values were log-transformed to achieve normality. Adjusting for selected covariates, only with the first trimester was every doubling of serum DDE levels found to be significantly associated with a reduction in PDI scores, by 0.52 points in the first year (95% CI= -0.96 to -0.075; p=0.02). This is approximately a two-point decrease for every 10-fold increase in first-trimester maternal DDE levels. In the case of MDI, no significant association was observed with maternal DDE levels in all three trimesters.
Although a high-quality study, the very high attrition rate (approx. 75%) could have made those eventually studied different from those from the larger population. They only demonstrated a level of comparability in very few selected baseline characteristics among those excluded, those lost to follow-up and those included in analysis. Also, no cut-offs were stated as regards the normal values for the two outcome indices.
As a follow-up to the study above, a study was done by Torres-Sanchez et al. to determine whether the effect persists between 12 and 30 months of age in a similar set of children in Morelos, Mexico26. Maternal serum levels of p,p'-DDE and p,p'-DDT in each trimester were evaluated on a wet basis. Only 21.2% of cohort members had p,p'-DDT levels above the detection limit and therefore not analysed. No significant association was found between DDE and either MDI or PDI scores of children at between 12 and 30 months of age, even when DDE values were adjusted for lipids, as the authors claimed (data not provided).
Compared with the earlier study, the numbers of mothers whose serum DDE levels were measured at each trimester were similar. However, the residual high attrition rate from the earlier study might have had a carryover effect. Also, the authors did not adequately account for the differences between the '333' claimed to be in the original cohort and the 244 that actually remained in this 'existing' cohort. If new members had been allowed into the cohort, findings may be affected.
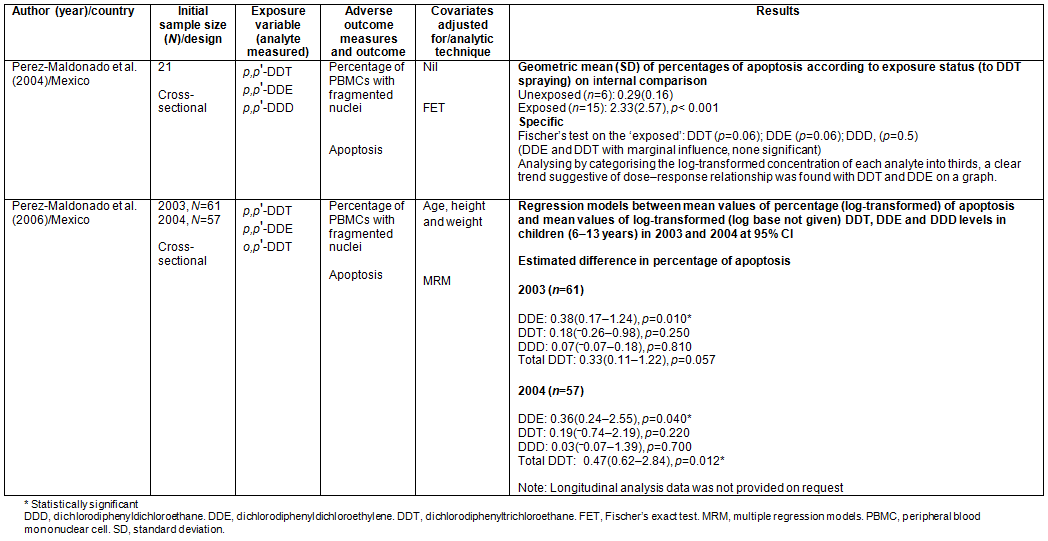
Studies investigating adverse immunity-related outcomes (n=2): A pilot cross-sectional study was performed by Perez-Maldonado et al. examining whether DDT and its metabolites are able to induce apoptosis of human peripheral blood mononuclear cells (PBMCs) both in vitro and in vivo27. The levels of DDT, DDE, dichlorodiphenyldichloroethane (DDD) and the percentage of in vivo apoptotic cells were measured from whole blood samples of the participants, and values log-transformed to achieve normality.
For an 'internal comparison', children recruited from an unsprayed community were classified as unexposed. The geometric mean of the percentages of apoptosis was significantly higher in the exposed than in the unexposed children (p<0.001). Categorising children into three groups - the unexposed children being the lowest while the second and third were those with medium and high exposures respectively - a clear trend of higher apoptosis with higher exposures was found for all analytes (only graphs provided). Although without any statistical adjustments, only DDT and DDE were able to show a weak but insignificant association. However, the authors neither described their 'randomisation' nor accounted for the numbers recruited into both groups of this extremely small study but introduced them at the less rigorous analyses stage.
Two cross-sectional studies were conducted by Perez-Maldonado et al. to verify whether the blood DDT and DDE concentrations are associated with the frequency of apoptosis of PBMCs in exposed healthy children28. In 2003, 61 children were recruited, and, in 2004, 57 children were recruited differently; all from La Ciguena, El Ramonal and La Ventinella in Mexico. As a secondary objective, a cohort of 34 children was followed from 2002 to 2004.
Using blood samples to assess the concentrations of DDT and DDE, and the percentage of apoptosis, only DDE demonstrated a significant association with apoptosis frequencies (p=0.10 and p=0.04 for 2003 and 2004, respectively).
Study investigating adverse birth/survival outcome (n=1): A case-cohort study was performed by Torres-Arreola et al. in Mexico in 1995 to examine the associations that maternal serum level of p,p'-DDE and that of two other organochlorine pesticides not under review may have with preterm birth29. A total of 100 preterm births were recruited as cases and 133 full-term births as controls from the same cohort.
Having categorised each organochlorine into tertiles using the distribution among the controls, the adjusted ORs for preterm birth in the middle and uppermost tertiles of the p,p'-DDE concentration were 1.87 (95% CI=0.95 to 3.68, p(trend)=0.17) and 1.67 (95% CI=0.84 to 3.31), respectively.
Although differences in baseline risk factors were adequately adjusted for, no explanation was given for the randomisation and blinding. The relatively wide confidence intervals also suggest that the fraction of the sample size for each category of DDE concentration might have been too small to detect an association if one exists. Being a case-cohort study, only perinatal maternal serum DDE level was used, and this might not have represented the average found in cohort studies.
Comparison of studies and methodological variations detected
Broadly, no study discussed any pre-study power calculation and this was reflected in the level of robustness of many studies. All other studies except three27-29 ensured blinding of outcome assessors to maternal exposure parameters. All studies on neonates/infants were performed on singleton births. Except for a pilot study27, all other studies analysed effects and adjusted for confounders using regression analyses. The heterogeneity discovered across studies is indicated below.
Studies on endocrine/reproductive/congenital outcomes: A statistically significant association was only demonstrated between first-trimester maternal serum level and API in males, in Torres-Sanchez et al., where the sample size was very small (n=37) and all mothers primiparous22. The opposite was found with the well-powered study (a very high response rate (95%) and low attrition rate (10%)) of Longnecker et al. where most mothers had given birth previously21. Significant amounts of DDE excreted through previous births might have influenced outcomes here. Unlike Longnecker et al21 however, participants' recruitment by Torres-Sanchez et al22 might not be representative of the source population, possibly influencing the direction and magnitude of effect because infants for this cross-sectional study were recruited from an ongoing cohort. Furthermore, only one observer measured outcomes in Torres-Sanchez et al22, and, as such, a bias might be introduced.
Bornman et al. demonstrated a significant association between a DDT-sprayed village and the occurrence of urogenital malformations, although in this study the exposure status was characterised by information in patient's file about the spraying status of their village of residence23. This could have predisposed the study to misclassification of participants, considering migration possibilities. There were also inadequacies in analysis and in data computed for event rates and no account was made for 166 neonates missing in their analysis, although this might not eventually alter effects sizes.
Studies on neurodevelopmental outcomes: DDE consistently demonstrated a significant association in two of these high-quality studies that investigated possible association between maternal DDE levels during pregnancy and MDI or PDI although it is noteworthy that all studies had very high attrition rates. However, in their studies Torres-Sanchez et al25,26, unlike Eskenazi et al24, did not adjust for whether mothers smoked during pregnancy. Although estimates of the mean for PDI and MDI scores at 6 months by Eskenazi et al24 were similar to that of Torres-Sanchez et al25, estimates at 12 months were much higher in the former (PDI=106±12.5 vs 91.0±7.4; MDI=100.9±9.0 vs 94.1±7.1). Differences that might account for these are that, unlike in the study of Eskenazi et al24, where mothers had migrated out of Mexico, more of the mothers in the latter were primiparous, homemakers, much younger and still getting residual doses from the environment.
Studies on immunity-related outcomes: Out of these two successive studies conducted by Perez-Maldonado et al27,28, only the latter, with the greater sample size, detected statistically significant association only for DDE in 2003 and 2004. The analysis in the much smaller preliminary study was less rigorous (using Fischer's test), while in the later study regression analysis was used, controlling for few confounders, though claiming that participants had similar socioeconomic backgrounds.
Discussion
Even though DDT and its metabolite, DDE, have been considered separately in the results section for clarity, both are treated as a single putative risk factor in this section for a holistic risk assessment.
Endocrine disruptors affect the release, binding and metabolism of endogenous hormones30,31. Prenatal effects of these are reflected in reduced AGD (an age-dependent parameter whose clinical significance on human reproduction remains vague30,32,33), penile dimensions (less reliable than AGD34) and other developmental anomalies35. The two related cross-sectional studies included may have reliably showed no association between foetal DDE exposure and hormone-related anthropometric parameters in boys and girls, given that slight differences exist between serum levels at birth and during pregnancy36. However, Torres-Sanchez et al., with a very small sample size, demonstrated a negative association with API (an age-independent parameter calculated using AGD) in boys22. The main difficulty here is the limited ability to detect the temporal sequence in associations detected in cross-sectional studies37. There is therefore insufficient reason to conclude whether there is an association or not.
However, Bornman et al. demonstrated an association between the occurrence of any UGBD and a DDT-sprayed village23. A major weakness in their findings is the categorisation of all different congenital defects studied as a single variable (apparently multiple testing19). Such finding lacks any specificity because each birth defect may be related to other risk factors apart from DDT. Also, the retrospective nature of the study makes it vulnerable to misclassification, possibly explaining the marginal differences in event rates observed between the 'exposed' and the 'unexposed' groups (aggregates being 11.0% and 10.2% respectively). Otherwise, such a high event rate within the 'unexposed' group observed in the study should attract urgent well-planned research attention because cryptorchidism, especially, has been found to be both the most frequent abnormality of male sexual differentiation and the main risk factor for testicular cancer and male infertility38,39.
Mild, severe or total debilitating adverse effects on the nervous system in prenatal or childhood life may not be obvious until later in life40. Different analyses performed to suit the various research objectives of the three studies on neurodevelopment make comparability quite challenging. However, the negative associations demonstrated between the foetal exposure to DDT/DDE and PDI scores in infants in two studies24,25 and the consistent negative trend, as age increased, found with mental development in Eskenazi et al24, call for deep attention. Gradual withdrawal from breastfeeding at such ages might have caused increasing expression of possible detrimental effects of DDT and its metabolites. Alternatively, the beneficial effect of breastfeeding on neurodevelopment observed in the studies (regardless of DDT/DDE exposure) might have attenuated any possible damage DDT/DDE could cause, something consistent with literature41. From another viewpoint, postnatal exposure could have added to the effects sizes observed because children assessed for neurodevelopmental outcomes over the course of 12-30 months would have had incremental exposure through environmental residues and breastfeeding (which increases organochlorines transfer42). Therefore, if the effects sizes are to be reckoned with, then they become really clinically significant at a population level, if not in individuals. Therefore, in the light of Bradford Hill criteria37, there is a suggestive evidence on impaired mental development but no obvious causal link, as issues of specificity (although they controlled for lead), dose-response relationship and strength of association are still to be resolved.
PBMCs help in mediating inflammatory and immunological functions in the body through a release of chemical factors43,44. If immunosuppression should set in through much-faster-than-normal rates of apoptosis of these cells, infection susceptibility would increase. The low-precision negative associations found by Perez-Maldonado et al28 between DDT and DDE exposure in children 6-13 years and the frequency of apoptosis of PBMCs, at different concentrations that existed in 2003 and 2004, make the evidence less reliable. There is also a difficulty in comparing this with the earlier inadequately designed study. Being cross-sectional studies, the feasibility of establishing temporal sequence between variables is dim. Also, it is difficult to know whether DDE is the only risk factor for increased frequency of apoptosis in such populations or assess how consequential it is amidst other risk factors, thus making a concrete conclusion impossible.
Preterm birth has been identified as a major determinant of mortality and long-term morbidities like respiratory ailments, neurological impairment and developmental delay in infants although its pathogenesis has remained unclear45. The only included study that looked into spontaneous preterm birth29 demonstrated no associations between sublevels of p,p'-DDE concentration in the population and preterm birth.
In a similar retrospective study in the US46, where the median DDE level was 25µg/L (less than half than the WHO average for zones where DDT is in current use47), they found strong associations between the variables, following a dose-response trend. Another small case-control study48 in Canada found no association between the variables although the median serum DDE levels were just half of that in the study under review.
Going by the contradictory evidence from the wider literature, we cannot conclude that DDE is not a risk factor for preterm birth, seeing that the study by Torres-Arreola et al29 lacked sufficient statistical power. If there is a strong association or causal link, it should be addressed in future studies, as preterm birth implies serious consequences, especially in high-risk areas that experience very high neonatal mortality.
Of the studies included in this review, 89% were carried out on Mexican populations of varying social classes. The differences between the socioeconomic context of Mexico and that of countries most affected by exposure to DDT (from IRS), and the 'noncurrent' status of DDT spraying at the time most studies were conducted limit the generalisability of the findings. Therefore, more studies are needed from areas where DDT is currently used in IRS in order to better assess effects.
Limitations
Language bias, through the restriction of the inclusion criteria to studies written in English, was a major challenge. A relevant study found was written in Spanish, and despite extensive efforts made, no English version of it was found. This clearly indicates that a number of studies reporting in non-English-speaking countries might have been missed. Also, this English bias could have reduced the chances of indexing or citing studies reported only in their original language in many databases. Further still, some unretrieved studies could have been eligible. As well, poor technology in some parts of the world might not have enabled indexing studies performed there in electronic databases. In all, the review was based on varying levels and qualities of evidence from observational studies, with inferences that are weak for policy decisions.
Only very limited evidence exists about the safety of such a widely used and persistent chemical, DDT, for IRS purposes after approximately seven decades of its insecticidal use. In addition, many of the outcomes reviewed could be counted as 'mere surrogates'. Long-term, well-coordinated birth cohort studies that document outcomes until at least school age or adolescence may be required to start getting the 'true endpoints' for possible effects of DDT/DDE.
Eight out of nine included studies emanated from parts of the world where there was no 'current' use of DDT at the time of study. That, besides the methodological concerns about the types and quality of studies included, could add to explaining the weak associations found. In areas where there are sustained higher titres from current use in IRS over many years, the consequences might be more serious on children. It is therefore imperative that the WHO policy for the use of DDT for IRS be broadened to accommodate periodical evaluation of its safety in countries currently using it, with national legal frameworks monitoring the circulation and the application of DDT.
Acknowledgements
We thank Dr Jon Pollock for his guidance in improvising an appraisal tool.
References
1. Smith A. How toxic is DDT? Lancet 356(9226): 267-268. (Online). 2000. Available: http://www.thelancet.com/journals/lancet/article/PIIS0140-6736%2800%2902497-1/fulltext (Accessed 12 May 2015).
2. Sadasivaiah S, Tozan Y, Breman JG. Dichlorodiphenyltrichloroethane (DDT) for indoor residual spraying in Africa: how can it be used for malaria control? American Journal of Tropical Medicine and Hygiene 77(6_Suppl): 249-263. (Online). 2007. Available: http://www.ajtmh.org/cgi/content/abstract/77/6_Suppl/249 (Accessed 27 December 2010).
3. Ecobichon DJ. Pesticide use in developing countries. Toxicology 160(1-3): 27-33. (Online). 2001. Available: http://www.sciencedirect.com/science/article/B6TCN-42H7PP3-5/2/94e89cb21f1757a9026e1362df0f2ef7 (Accessed 28 December 2010).
4. Mabaso MLH, Sharp B, Lengeler C. Historical review of malarial control in southern African with emphasis on the use of indoor residual house-spraying. Tropical Medicine & International Health 9(8): 846-856. (Online). 2004. Available: http://dx.doi.org/10.1111/j.1365-3156.2004.01263.x. (Accessed 27 December 2010).
5. World Health Organization. WHO position on DDT use in disease vector control under the Stockholm Convention on Persistent Organic Pollutants (Online) 2004. Available: http://whqlibdoc.who.int/hq/2004/WHO_HTM_RBM_2004.53.pdf (Accessed 14 June 2011).
6. World Health Organization. Indoor residual spraying: use of indoor residual spraying for scaling up global malaria control and elimination (Online). 2006. Global Malaria Programme. Available: http://whqlibdoc.who.int/hq/2006/WHO_HTM_MAL_2006.1112_eng.pdf (Accessed 16 June 2011).
7. Carlson R. Silent spring. New York: Fawlett Crest Books, 1962.
8. Longnecker MP, Klebanoff MA, Dunson DB, Guo X, Chen Z, Zhou H, et al. Maternal serum level of the DDT metabolite DDE in relation to fetal loss in previous pregnancies. Environmental Research 97(2): 127-133. (Online). 2005. Available: http://www.sciencedirect.com/science/article/pii/S0013935103001087 (Accessed 12 May 2015).
9. Beard J. DDT and human health. Science of The Total Environment 355(1-3): 78-89. (Online). 2006. Available: http://www.sciencedirect.com/science/article/pii/S0048969705001749 (Accessed 12 May 2015).
10. Aneck-Hahn NH, Schulenburg GW, Bornman MS, Farias P, De Jager C. Impaired semen quality associated with environmental DDT exposure in young men living in a malaria area in the Limpopo Province, South Africa. Journal of Andrology 28(3): 423-434 (Online). 2007. Available: http://www.andrologyjournal.org/cgi/content/abstract/28/3/423 (Accessed 28 December 2010).
11. Oduola AO, Olojede JB, Ashiegbu CO, Adeogun AO, Otubanjo OA, Awolola TS. High level of DDT resistance in the malaria mosquito: Anopheles gambiae S.L from rural, semi urban and urban communities in Nigeria. Journal of Rural and Tropical Public Health 9: 114-120. (Online). 2010. Available: http://www.jcu.edu.au/jrtph/vol/JRTPH_vol9_p114-120_Oduola.pdf. (Accessed 08 March 2010).
12. World Health Organisation. The use of DDT in malaria vector control: WHO position statement. Global Malaria Programme (Online). 2011. Available: http://whqlibdoc.who.int/hq/2011/WHO_HTM_GMP_2011_eng.pdf (Accessed 16 June 2011).
13. van den Berg H. Global status of DDT and its alternatives for use in vector control to prevent disease. Environmental Health Perspectives 117: 1656-1663. (Online). 2009. Available: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2801202/pdf/ehp-117-1656.pdf (Accessed 14 April 2011).
14. Osunkentan AO. Chronic adverse effects of long-term exposure of children to dichlorodiphenyltrichloroethane (DDT) through indoor residual spraying: a systematic review. University of the West of England MSc Public Health dissertation (unpublished) 2011.
15. Centre for Reviews and Dissemination (CRD). Systematic reviews: CRD's guidance for undertaking reviews in health care. (3rd ed.) (Online). 2008. Available: http://www.york.ac.uk/inst/crd/pdf/Systematic_Reviews.pdf (Accessed 17 March 2011).
16. Khan KS, Kunz R, Kleijnen J, Antes G. Systematic reviews to support evidence-based medicine: how to review and apply findings of healthcare research. London: Royal Society of Medicine Press, 2003.
17. Greenhalgh T. How to read a paper. (3rd ed.) Oxford: Blackwell-BMJ, 2003.
18. Yeshiva University. 11 questions to help you make sense of descriptive/cross-sectional studies (Online). 2011. Available: http://www.yu.edu/aecomdb/dfsm/Uploads/ugclerkshipinfo/cross-sectional study appraisal tool.pdf. (Accessed 31 May 2011).
19. Elwood M. Critical appraisal of epidemiological studies and clinical trials. (2nd ed.) New York: Oxford University Press, 1998.
20. Young JM, Solomon MJ. How to critically appraise an article. 6(2): 82-91. (Online). 2009. Nature Clinical Practice Gastroenterology and Hepatology. Available: http://dx.doi.org/10.1038/ncpgasthep1331 (Accessed 6 June 2011).
21. Longnecker MP, Gladen BC, Cupul-Uicab L, Romano-Riquer S, Weber JP, Chapin RE, et al. In utero exposure to the antiandrogen 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene (DDE) in relation to anogenital distance in male newborns from Chiapas, Mexico. American Journal of Epidemiology 2007; 165(9): 1015-1022.
22. Torres-Sanchez L, Zepeda M, Cebrian ME, Belkind-Gerson J, Garcia-Hernandez RM, Belkind-Valdovinos U, et al. Dichlorodiphenyldichloroethylene exposure during the first trimester of pregnancy alters the anal position in male infants. Annals of New York Academy of Sciences 1140: 155-162. (Online). 2008. Available: http://dx.doi.org/10.1196/annals.1454.004 (Accessed 29 April 2011).
23. Bornman R, De Jager C, Worku Z, Farias P, Reif S. DDT and urogenital malformations in newborn boys in a malarial area. BJU International (Online). 106(3): 405-411. 2010. Available: http://dx.doi.org/10.1111/j.1464-410X.2009.09003.x (Accessed 28 April 2011).
24. Eskenazi B, Marks AR, Bradman A, Fenster L, Johnson C, Barr DB, et al. In utero exposure to dichlorodiphenyltrichloroethane (DDT) and dichlorodiphenyldichloroethylene (DDE) and neurodevelopment among young Mexican American children Pediatrics 118(1): 233-241. (Online). 2006. Available: http://pediatrics.aappublications.org/content/118/1/233.full.pdf+html (Accessed 30 April 2011).
25. Torres-Sánchez L, Rothenberg SJ, Schnaas L, Cebrian ME, Osorio E, Del Carmen Hernandez M, et al. In utero p,p'-DDE exposure and infant neurodevelopment: a perinatal cohort in Mexico. Environmental Health Perspectives 115(3): 435-439. (Online). 2007. Available: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1849908/pdf/ehp0115-000435.pdf (Accessed 30 April 2011).
26. Torres-Sánchez L, Schnaas L, Cebrián ME, Hernández MdC, Valencia EO, García Hernández RM, et al. Prenatal dichlorodiphenyldichloroethylene (DDE) exposure and neurodevelopment: a follow-up from 12 to 30 months of age. Neurotoxicology 30(6): 1162-1165. (Online). 2009. Available: http://www.sciencedirect.com/science/article/pii/S0161813X0900182X. (Accessed 29 April 2011).
27. Perez-Maldonado IN, Diaz-Barriga F, de la Fuente H, Gonzalez-Amaro R, Calderon J, Yanez L. DDT induces apoptosis in human mononuclear cells in vitro and is associated with increased apoptosis in exposed children. Environmental Research 94(1): 38-46. (Online) 2004. Available: http://www.sciencedirect.com/science/article/pii/S0013935103001129. (Accessed 29 April 2011).
28. Perez-Maldonado IN, Athanasiadou M, Yanez L, Gonzalez-Amaro R, Bergman A, Diaz-Barriga F. DDE-induced apoptosis in children exposed to the DDT metabolite. Science of the Total Environment 370(2-3): 343-351. (Online). 2006 .Available: http://www.sciencedirect.com/science/article/pii/S0048969706004815. (Accessed 29 April 2011).
29. Torres-Arreola L, Berkowitz G, Torres-Sanchez L, Lopez-Cervantes M, Cebrian ME, Uribe M, et al. Preterm birth in relation to maternal organochlorine serum levels. Annals of Epidemiology 13(3): 158-162. (Online). 2003. Available: http://www.sciencedirect.com/science/article/pii/S1047279702004246. (Accessed 30 April 2011).
30. Wang M, Baskin LS. Endocrine disruptors, genital development, and hypospadias. Journal of Andrology 29(5): 499-505. (Online). 2008. Available: http://www.andrologyjournal.org/cgi/content/abstract/29/5/499 (Accessed 16 June 2011).
31. Wolf C, Lambright C, Mann P, Price M, Cooper RL, Ostby J, et al. Administration of potentially antiandrogenic pesticides (procymidone, linuron, iprodione, chlozolinate, p,p′-DDE, and ketoconazole) and toxic substances (dibutyl- and diethylhexyl phthalate, PCB 169, and ethane dimethane sulphonate) during sexual differentiation produces diverse profiles of reproductive malformations in the male rat. Toxicology and Industrial Health 15(1-2): 94-118. (Online). 1999. Available: http://tih.sagepub.com/content/15/1-2/94.abstract
32. Swan SH. Environmental phthalate exposure in relation to reproductive outcomes and other health endpoints in humans. Environmental Research 108(2): 177-184. (Online) 2008. Available: http://www.sciencedirect.com/science/article/pii/S0013935108001862 (Accessed 18 June 2011).
33. Hsieh M, Breyer B, Eisenberg M, Baskin L. Associations among hypospadias, cryptorchidism, anogenital distance, and endocrine disruption. Current Urology Reports 9(2): 137-142. (Online) 2008. Available: http://dx.doi.org/10.1007/s11934-008-0025-0. (Accessed 12 June 2011).
34. Romano-Riquer SP, Hernández-Ávila M, Gladen BC, Cupul-Uicab LA, Longnecker MP. Reliability and determinants of anogenital distance and penis dimensions in male newborns from Chiapas, Mexico. Paediatric and Perinatal Epidemiology 21(3): 219-228. (Online). 2007. Available: http://dx.doi.org/10.1111/j.1365-3016.2007.00810.x. (Accessed 14 June 2011).
35. Jarrett RE. Endocrine disruptor chemicals as a rising compliance issue. Federal Facilities Environmental Journal 11(1): 25-39. (Online). 2000. Available: http://dx.doi.org/10.1002/ffej.3330110104 (Accessed 14 June 2011).
36. Longnecker MP, Klebanoff MA, Gladen BC, Berendes HW. Serial levels of serum organochlorines during pregnancy and postpartum. Archives of Environmental Health 54(2): 110-114. (Online). 1999. Available: http://dx.doi.org/10.1080/00039899909602244 (Accessed 16 June 2011).
37. Gerstman BB. Epidemiology kept simple: an introduction to traditional and modern epidemiology. New Jersey: Wiley-Liss, 2003.
38. Thonneau PF, Candia P, Mieusset R. Cryptorchidism: Incidence, risk factors, and potential role of environment; an update. Journal of Andrology 24(2): 155-162. (Online). 2003. Available: http://www.andrologyjournal.org (Accessed 16 June 2011).
39. Thorup J, McLachlan R, Cortes D, Nation TR, Balic A, Southwell BR, et al. What is new in cryptorchidism and hypospadias - a critical review on the testicular dysgenesis hypothesis. Journal of Pediatric Surgery 45(10): 2074-2086. (Online). 2010. Available: http://www.sciencedirect.com/science/article/pii/S0022346810006172 (Accessed 16 June 2011).
40. Colborn T. A case for revisiting the safety of pesticides: a closer look at neurodevelopment. Environmental Health Perspectives 114: 10-17. (Online). 2006. Available: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1332649/pdf/ehp0114-000010.pdf (Accessed 14 June 2011).
41. Ribas-Fito N, Cardo E, Sala M, Eulalia de Muga M, Mazon C, Verdu A, et al. Breastfeeding, exposure to organochlorine compounds, and neurodevelopment in infants. Pediatrics 111(5): 580-585. (Online). 2003. Available: http://pediatrics.aappublications.org/cgi/content/abstract/111/5/e580 (Accessed 16 June 2011).
42. Waliszewski SM, Aguirre AA, Infanzon RM, Silva CS, Siliceo J. Organochlorine pesticide levels in maternal adipose tissue, maternal blood serum, umbilical blood serum, and milk from inhabitants of Veracruz, Mexico. Archives of Environmental Contamination and Toxicology 40(3): 432-438. (Online). 2001. Available: http://dx.doi.org/10.1007/s002440010194 (Accessed 16 June 2011).
43. Alegría-Torres JA, Díaz-Barriga F, Gandolfi AJ, Pérez-Maldonado IN. Mechanisms of p,p′-DDE-induced apoptosis in human peripheral blood mononuclear cells. Toxicology in Vitro 23(6): 1000-1006. (Online). 2009. Available: http://www.sciencedirect.com/science/article/pii/S0887233309001490. (Accessed 10 June 2011).
44. Beck Z, Brown BK, Matyas GR, Polonis VR, Rao M, Alving CR. Infection of human peripheral blood mononuclear cells by erythrocyte-bound HIV-1: effects of antibodies and complement. Virology 412(2): 441-447 (Online). 2011. Available: http://www.sciencedirect.com/science/article/pii/S0042682211000602 (Accessed 10 June 2011).
45. Pathak R, Ahmed RS, Tripathi AK, Guleria K, Sharma CS, M, a, khijani SD, et a, l. Maternal and cord blood levels of organochlorine pest, icides: association with preterm labor. Clinical Biochemistry 42(7-8): 746-749. (Online). 2009 Available: http://www.sciencedirect.com/science/article/pii/S0009912008005997. (Accessed 18 June 2011).
46. Longnecker MP, Klebanoff MA, Zhou H, Brock JW. Association between maternal serum concentration of the DDT metabolite DDE and preterm and small-for-gestational-age babies at birth. Lancet 358(9276): 110-114. (Online). 2001. Available: http://www.sciencedirect.com/science/article/pii/S0140673601053296 (Accessed 16 June 2011).
47. World Health Organization. DDT in indoor residual spraying: human health aspects. (Online). 2011. Available: http://www.who.int/ipcs/publications/ehc/ehc241.pdf (Accessed 19 June 2011).
48. Wood SL, Jarrell JJ, Swaby C, Chan S. Endocrine disruptors and spontaneous premature labour: a case-control study. Environmental Health 6(35) (Online). 2007. Available: http://www.ehjournal.net/content/6/1/35 (Accessed 20 June 2011).
