The incidence of tuberculosis (TB) in India has failed to decline sufficiently despite sustained programmatic efforts. Apart from other performance indicators such as high case detection and treatment success rate, early diagnosis and prompt initiation of treatment is essential to reduce the morbidity and mortality due to TB1. Delay in initiation of treatment has been reported extensively to have a significant impact on disease transmission, extensive progression, unfavourable treatment outcome and accentuated mortality rates2. Several studies have reported delay in initiation of treatment to be a contributory factor in development of drug-resistant TB3.
Delay in initiation of treatment can occur at various stages: (1) onset of symptoms to care seeking, (2) first contact with health system to diagnosis and (3) diagnosis to initiation of treatment4. Delay at level 1 is mainly related to patient-level factors, whereas delay at levels 2 and 3 could be due to failure on the part of patient or health system or both. Although patient-level delay and diagnostic delay have been studied extensively in India and other developing countries, literature documenting delay between diagnosis and initiation of treatment is limited. Most of the existing studies from India are from southern and western regions, and only few were from the primary healthcare setting5. Since the delay could be dependent on study setting and/or geographical region, it is imperative to identify the magnitude of delay, and programmatic factors associated with it across various regions.
The authors undertook this cohort analysis of pulmonary TB patients initiated on treatment under the Revised National Tuberculosis Control Program (RNTCP) between 2007 and 2014 to describe the delay in initiation of anti-TB treatment after diagnosis, and factors associated with it at a primary healthcare setting in the Ballabgarh block of district Faridabad, Haryana, North India.
Study setting
The study was conducted at two primary health centres (PHCs) in the Faridabad district. These two PHCs were part of the Comprehensive Rural Health Services Project (CRHSP) of the Centre for Community Medicine, All India Institute of Medical Sciences, New Delhi. These two PHCs served 28 villages with a population of nearly 95 000 individuals (as of December 2014). The entire population was a part of the Health and Demographic Surveillance System (HDSS), Ballabgarh. Details regarding HDSS Ballabgarh are available elsewhere6. The health and vital statistics information in HDSS were recorded during fortnightly domiciliary visits by village-level health workers.
Tuberculosis care in study area
Diagnosis and treatment of TB was done as per the RNTCP protocol7. Case detection of TB was done through passive surveillance. The diagnosis was done using sputum smear microscopy, and chest X-ray if the sputum smear was negative and pulmonary TB was clinically suspected. The diagnosis was made by medical officers of these PHCs. RNTCP recommends the directly observed treatment - short course (DOTS) strategy for treatment. Diagnosis of TB by microscopy was available at one PHC, whereas the other PHC provided only treatment. Residents of the study villages that were diagnosed with TB were registered and started on treatment. Patients residing outside of the study villages were transferred out to their respective PHCs. Similarly, patients that were residents of study villages but were diagnosed at any other government health facility and were transferred in to the study PHCs were registered for initiation of treatment. Tuberculosis patients taking treatment from a private health facility or from any health facility outside the purview of RNTCP were not part of the study. Before initiating the treatment, the patient was required to have their address verified by the health worker allocated to their village. In addition, a DOTS provider had to be identified who was either a community health volunteer or a health worker. Treatment was started after these two requirements were met. All the records related to TB care and treatment were maintained as per the reporting guidelines of RNTCP.
Study design
This study was a cohort analysis of TB patients started on anti-TB treatment under the national program from 2007 to 2014 from two PHCs. The overall study enrolment procedure is explained in Figure 1.
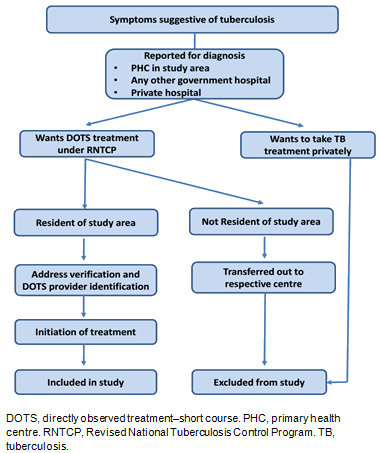
Figure 1: Study enrolment procedure for diagnosis and treatment of tuberculosis patients.
Data source and study variables
A TB register was maintained at both the PHCs. Demographic details such as age, sex, caste and place of residence (PHC village or non-PHC village), date of diagnosis and initiation of treatment, initial sputum smear results, site of TB (pulmonary or extrapulmonary), category of TB treatment (new or retreatment), and outcome of TB treatment was available in the TB register. The data available in the TB register were extracted and entered in EpiInfo v3.5.4 (Centers for Disease Control and Prevention; https://www.cdc.gov/epiinfo). Data cleaning was done, and logical checks were done to ensure correctness of the data.
Definitions
Initiation of treatment after diagnosis was considered to be delayed if the gap between diagnosis and treatment was greater than 7 days8. This definition was based on operational guidelines of RNTCP in India. Patients who had never had TB treatment or had taken it for less than 4 weeks were considered as 'new'; patients who had taken TB treatment for 4 weeks or more were categorised as 'retreatment'. Outcome of TB treatment was defined as per the guidelines laid by RNTCP:
- 'cure': patients who completed the full course of treatment and the sputum smear result was negative at the end of the treatment period
- 'treatment completed': patients who completed the full course of treatment but the sputum smear at the end of the treatment period was either not done or the result was not available
- 'default from treatment': patients who took the treatment for at least 4 weeks and then discontinued the treatment for at least two consecutive months
- 'failure': patients who remained or became smear positive at 5 months or later during the course of the treatment
- 'death': patients who died during the course of the treatment.
'PHC village' was defined as the village in the study area where either of the two PHCs were located.
Statistical analysis
Statistical analysis was done using Stata v13.0 (StataCorp; http://www.stata.com). Descriptive analysis was conducted to examine the demographic and clinical characteristics of the patients and presented as a proportion (95% confidence interval (CI)) and mean (±2 standard deviations (SD)) wherever applicable. Treatment outcome was dichotomised into favourable (cure or treatment completed) and unfavourable (default from treatment or failure) treatment outcome. Initial sputum smear results were categorised as negative, low positive ('1+' and '2+') and high positive ('3+' and '4+'). Bivariate and multivariate logistic regression was performed to identify independent predictors of delay in initiation of treatment (>7 days), with age, sex, caste, village of residence, treatment category and initial sputum smear positivity taken as explanatory variables. Multivariate analysis was done to study the effect of delay in initiation of treatment on treatment outcome, unadjusted as well as adjusted for all other explanatory variables. Results were reported as odds ratios (OR) with 95%CI. p<0.05 was considered statistically significant.
Ethics approval
Ethical clearance for the study was obtained from the Institute Ethics Committee of All India Institute of Medical Sciences (AIIMS), New Delhi (Ref No. IEC-345/01.07.2016, RP-3/2016).
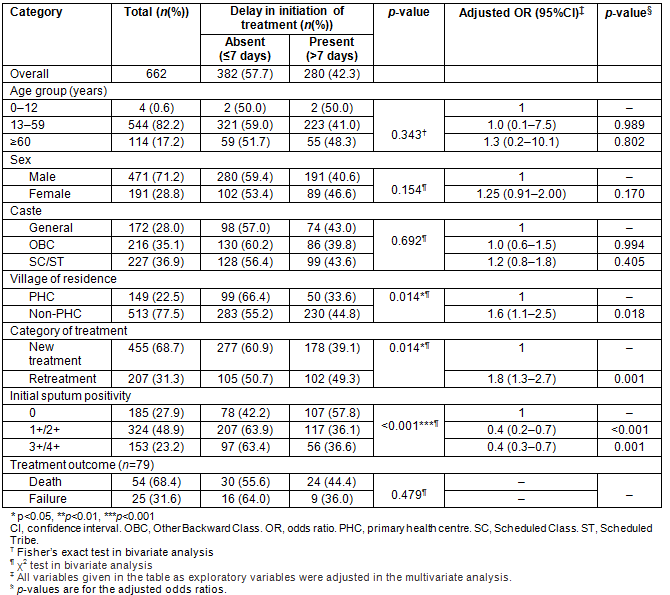
A total of 885 patients were started on TB treatment at two PHCs during the study period, ie from January 2007 to December 2014. Summary of performance indicators as recommended by RNTCP are shown in Table 1. Out of 885 patients, a total of 662 (74.8%) pulmonary TB patients for whom information regarding date of diagnosis and initiation of treatment was available were included in the analysis. Mean time interval between diagnosis and initiation of treatment was 8.95 days. Only 57.7% of pulmonary TB patients were started on treatment within 7 days of diagnosis, and an additional 24.5% were started on treatment 8-14 days after diagnosis (Table 2). Distribution of patients by time interval between diagnosis and treatment is shown in Table 2. Basic demographic and clinical characteristics of the patients included in this analysis are shown in Table 3.
On bivariate analysis, pulmonary TB patients residing in a non-PHC village (45% vs 35% p=0.01) were more likely to have a delay in initiation of treatment after diagnosis (>7 days). Similarly, those on the retreatment regimen (50% vs 39%, p=0.01) were more likely to have delay in initiation of treatment as compared to patients on the new treatment regimen (Table 3). On multivariate analysis, similar results were obtained with those on the retreatment regimen (OR=1.82 (1.3-2.7, p=0.001)) and those residing in non-PHC villages (OR=1.62 (1.1-2.5, p=0.01)) for delay in initiation of treatment (Table 3). Patients with smear-positive pulmonary TB had a lower odds (OR=0.42, 95% CI 0.2-0.7) of delay in initiation of treatment than those with smear-negative pulmonary TB. Among 595 patients where information on treatment outcome was available, delay in initiation of treatment was associated with unfavourable treatment outcome, ie default, failure or death during treatment (OR=1.87, 95%CI 1.11-2.93; Table 3).
Table 1: Key program performance indicators in the study area, 2007-2014 (n=885)
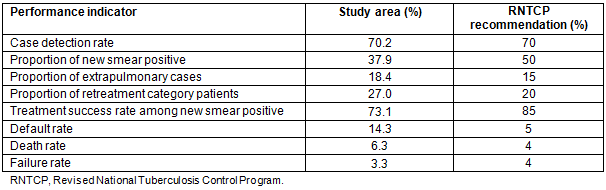
Table 2: Time interval between diagnosis and initiation of treatment among pulmonary tuberculosis patients (n=662)

Table 3: Distribution of delay in initiation of treatment by patient characteristics
Discussion
RNTCP recommends initiation of treatment within 7 days of diagnosis among 80% of patients. In comparison to the recommended norm, it was found that only 57.7% of patients were started on DOTS within 7 days of diagnosis. Thus, in the study area a high proportion of pulmonary TB patients had a delay in initiation of treatment. Literature shows that early initiation of treatment renders direct benefit to the patients in terms of improved rate of favourable treatment outcome, and indirect benefit to the community by reducing the transmission of TB3. One large study recently conducted in two districts of West Bengal and Andhra Pradesh reported 65% of pulmonary TB patients starting treatment within 7 days of diagnosis1.
The mean delay between diagnosis and initiation of treatment was 8.95 days in the present study and the median delay was 6 days. Another study done in two districts of West Bengal and Andhra Pradesh reported a median delay of 8.0 days, which was similar to the present study8. That study had also gathered information from the TB register. While the present study was in the primary healthcare setting, the other study included patients initiated on treatment at all the levels of care: primary, secondary and tertiary. Four studies, done in Karnataka, Maharashtra and Himachal Pradesh, reported a range of delay of 1.0-4.0 days. These studies were cross-sectional surveys of patients registered under DOTS2-5.
A primary survey to identify delay might introduce a social desirability bias, resulting in patients underreporting the delay. There is a possibility of recall bias depending upon time lag between initiation of treatment and primary survey. Moreover, most of these studies had small sample sizes ranging from 39 to 266 pulmonary TB patients. A survey of patients taking treatment from private practitioners outside the purview of DOTS reported a delay of 3.4 days6. Private practitioners do not need to undertake address verification of patient prior to the start of treatment, as is required in a public health facility. Therefore, a shorter time interval between diagnosis and initiation of treatment could be expected in private healthcare settings.
Patients in most of the previous studies were smear-positive pulmonary TB patients. In the present study, smear-negative pulmonary TB (n=185, 27.9%) was included as well. The authors found that smear-negative patients were twice as likely to have delay in initiation of treatment than smear-positive patients. Thus, the longer median delay in the present study compared to previous studies could be a consequence of inclusion of smear-negative pulmonary TB patients in the present study. None of the previous studies have addressed this issue. Smear-positive patients are more likely to have symptoms that are more severe (eg haemoptysis) than those of the smear-negative patients. Thus, they would be more eager to start the treatment early in order to alleviate their symptoms. Smear-negative patients rarely transmit the disease. Therefore, delay in initiation of treatment in smear-negative patients would have a limited role in spreading the disease, although treatment outcomes for those patients may be less favourable.
Patients residing in villages where PHCs were located were half as likely to have delays in initiation of treatment. Physical proximity of patient residence to the PHC and its attached subcentre is likely to shorten the time period required for address verification and DOTS provider identification. This in turn could lead to a shortened time interval between diagnosis and initiation of treatment.
Retreatment category patients had a greater likelihood and longer median duration of delay in initiation of treatment after diagnosis. Patients on retreatment regimens are generally at higher risk of being drug resistant. Delay in initiation of treatment in these patients could further increase the chances of transmission of drug-resistant strains. Retreatment patients have also been found to go 'doctor shopping' even after diagnosis at an RNTCP facility. Pretreatment counselling could help in reducing unnecessary visits to multiple healthcare providers even after diagnosis is established. Association of these factors with delayed initiation of treatment may have an adverse impact, resulting in unfavourable treatment outcome. The authors found that delayed initiation of treatment was associated with unfavourable treatment outcome. Successful treatment is the mainstay of the TB control program in India. Hence, unfavourable treatment outcome defeats the objectives of the RNTCP.
The study was based on data recorded in the TB register. Records of some TB patients may not have been entered in the TB register. The TB program was accorded high priority at the PHCs. Therefore, the number of patients not recorded in the TB register is likely to be small. The authors could find no plausible reason to suspect that those who might be missing from TB register would be systematically different from those who were included. Therefore, the findings are considered likely to be representative of all TB patients that reported at the PHC.
The study cohort of TB patients included only those who were taking treatment from the PHCs. Patients treated at private health facilities were not included in the study. Hence, the findings of this study are applicable only to those pulmonary TB patients who received treatment at government-owned primary healthcare facility.
A significant proportion of pulmonary TB patients had delay in initiation of treatment. Physical proximity of residence to health facility and being a new patient starting on treatment were associated with lower chances of delay in initiation of treatment.
References
1. Paul D, Busireddy A, Nagaraja SB, Satyanarayana S, Dewan PK, Nair SA, et al. Factors associated with delays in treatment initiation after tuberculosis diagnosis in two districts of India. PLoS ONE 2012; 7(7): e39040. https://doi.org/10.1371/journal.pone.0039040
2. Goel K, Kondagunta N, JoyceeSoans S, Ramachandra Bairy A, Goel P. Reasons for patient delays and health system delays for tuberculosis in South India. Indian Journal of Community Health 2012; 23(2): 87-89.
3. Tamhane A, Sathiakumar N, Vermund S, Kohler CL, Karande A, Ambe G. Pulmonary tuberculosis in Mumbai, India: factors responsible for patient and treatment delays. International Journal of Preventive Medicine 2012; 3(8): 569-580.
4. Uplekar M, Juvekar S, Morankar S, Rangan S, Nunn P. Tuberculosis patients and practitioners in private clinics in India. International Journal of Tuberculosis and Lung Disease 1998; 2(4): 324-329.
5. Thakur R, Murhekar M. Delay in diagnosis and treatment among TB patients registered under RNTCP Mandi, Himachal Pradesh, India, 2010. Indian Journal of Tuberculosis 2013; 60(1): 37-45.
6. Kant S, Misra P, Gupta S, Goswami K, Krishnan A, Nongkynrih B, et al. The Ballabgarh Health and Demographic Surveillance System (CRHSP-AIIMS) International Journal of Epidemiology 2013; 42(2): 758-768.
7. Dhanvij P, Joshi R, Kalantri S. Delay in diagnosis of tuberculosis in patients presenting to a tertiary care hospital in rural central India. Journal of Mahatma Gandhi Institute of Medical Sciences 2009; 14(2): 56-63.
8. Central Tuberculosis Division, Ministry of Health and Family Welfare, Government of India. Technical and operational guidelines for tuberculosis control, Revised National Tuberculosis Control Programme. New Delhi: Government of India, 2005.
