Developmental dysplasia of the hip (DDH) is a condition occurring in infants, in which the surface of the femoral head has an abnormal relationship to the acetabulum. Developmental dysplasia of the hip is cited as the most common notifiable musculoskeletal birth defect by the South Australian Birth Defects Register at 7.0 per 1000 live births1. Failure to diagnose instability in the neonatal hip can lead to persistent excess movement in the joint, with poor development of the acetabulum and subsequent displacement of the head. If DDH is detected and treated at birth with an abduction brace the outcome is usually very favourable; however, if diagnosis is delayed until 3 months of age or older, the likelihood of requiring surgery to correct the problem is high2. If the condition is not diagnosed until the child demonstrates some motor difficulties, such as inability to bear weight on one leg or unilateral Trendelenburg gait, the child usually requires major hip surgery and immobilization to correct the position of the hip joint and gain better function. For these children there is the added risk of the comorbid effects of lessened activity including cardiovascular disease, and further surgery at an early age3.
Currently in South Australia (SA) all babies' hips are examined by a doctor in the neonatal period to screen for DDH using Barlow's test (flexion, adduction and posterior force attempting to subluxate or dislocate the infant's hip posteriorly)4, or Ortalani's manoeuvre (flexion, abduction and anterior force on the greater trochanter attempting to reduce a dislocated hip)5. If any abnormality is detected or there are risk factors (breech presentation at term, or family history), the baby may be referred for an ultrasound examination using the Graf method6. This is a static lateral-coronal view ultrasound which looks at the morphology of the developing acetabulum and specifically measures coverage of the femoral head7. Babies' hips are also re-examined at a 6 week visit to either the GP or the Child and Youth Health Services Nurse.
Despite this process, occurrences of DDH are still being missed8. One South Australian study concluded that birth in rural settings increases the risk of late diagnosis of DDH with an odds ratio of 3.89 (95% CI 1.61- 9.25, p=0.002)9. This may relate specifically to the context of maternity services in SA. Of the 19 766 births in hospitals in SA in 2009, 4.4% were delivered in one of two regional hospitals which have resident paediatric specialists and are situated more than 350 km from the city. Another 15.7% were delivered in more than 20 small rural hospitals where there are between 10 and 300 births per year, serviced almost exclusively by rural GPs with procedural skills10. In view of this context, one proposed explanation for the increase late diagnosis of DDH is that screening by physical examination in the newborn period is reliant on the clinical skills of rural generalists who perform neonatal screening examinations with less frequency than urban specialists8. This hypothesis is consistent with previous studies which have shown that less experienced clinicians have more false negative and false positive examination findings11-13.
Andersson et al showed conclusively that routine Anterior Dynamic Ultrasound (ADUS) screening for DDH in Sweden has increased sensitivity and specificity of screening for DDH in comparison with physical examination alone14,15. These findings have been confirmed in further studies in the USA, UK and Europe; however, routine screening has not been introduced in all countries for economic reasons, because even for infants with known risk factors (eg breech, family history) the number of ADUS needed to alter treatment prescribed as a result of physical examination alone can be as high as 20013,16,17.
Taking the hypothesis that rural doctors with less expertise miss more cases of DDH, a targeted screening program introducing ADUS to rural maternity units may improve the early detection rates of DDH in Australia. The aim of this study was to report on a small implementation pilot to provide ADUS screening of newborn hips in an Australian regional hospital with a view to understanding the feasibility of ADUS in rural hospitals, and the acceptability of screening to rural parents.
Anterior Dynamic Ultrasound is a dynamic, real-time scan which measures the maximum anterior-posterior movement of the femoral head within the acetabulum during hip abduction (MM)15. This study was designed as a Phase 1 effectiveness trial18 as the first step in implementing a large scale program in rural Australia to screen for DDH using ADUS. The research protocol with focussed management (Fig1) based on the ADUS results included:
- reassuring parents with infants who have MM of ≤ 3 mm
- providing focused counseling regarding the posturing of infants with hips abducted and follow-up screening for infants with MM of ≥ 3 mm
- referring infants with dislocatable hips for definitive treatment as per the current Australian protocol.
Infants born of mothers over 18 years of age at a single regional hospital in SA during the 17 week study period in 2011 were eligible to participate in the study. Exclusion criteria included: residential address outside the local district, unable to give informed consent (eg non-English speaking), and infants with an evident dislocatable hip found on clinical examination.
The ADUSs were provided on one morning per week at the regional hospital radiology department. Parents accompanied their baby to the radiology suite for the dynamic ultrasound. Collected for each infant were demographic data: sex, gestation at birth, birth weight, age at ultrasound; and relevant history of DDH, breech presentation and method of delivery. The ADUS was performed by an ultrasonographer using a 11-4 MHz curved linear array probe from an anterior position in the groin. The pelvis was stabilized and the leg flexed, abducted and pressure exerted posteriorly by a second clinician (Barlow's procedure5). Each femoral head was visualized in the acetabulum in the resting position and then as the hip joint was provoked with Barlow's test. The ADUS was captured by video, and the movement of the femoral head from a line along the femur to the pelvis across the upper joint capsule was measured from the ultrasound image (called maximum movement [MM]).
Once the ultrasound was completed the parents completed a brief survey assessing their anxiety levels. This included the short form of the Spielberger State Anxiety Scale19, which grades six statements regarding the emotions felt by the participant on a four-point likert scale (0=not at all, 1=somewhat, 2=moderate, 3=very much so). The survey included six questions about the acceptability of the procedure and convenience of attending for the ultrasound.
The research protocol then prescribed that, on completion of the survey, the health professionals performing the ADUS provided the parent/s with the results of the ultrasound. If the ultrasound indicated that movement within both hip joints was less than 3 mm, they were told that their child's hips were normal. If the ultrasound indicated the baby had a dislocated hip on the initial ADUS, the child and parents were to be immediately referred to a paediatric orthopaedic surgeon. If the ultrasound indicated that movement within one or both hips was greater than or equal to 3 mm, the parent/s were told their child's hip/s had increased hip laxity and was at risk of DDH. The parent/s were given advice about positioning the child prone with 'frog legs' during the wakeful time each day, and using a cloth nappy over their first nappy, to encourage optimal hip positioning for hip development in the neonatal period. They were asked to return at 4 weeks for follow-up investigations consisting of Graf-method ultrasound (as per current management protocols9), and repeat ADUS. The parent/infant's GP was sent a copy of the ADUS results. Those babies who still had excessive movement on the second ADUS at 4 weeks, or abnormal anatomical findings on Graf ultrasound examination were offered appropriate treatment in line with current treatment protocols. A summary of this process is provided (Fig1).
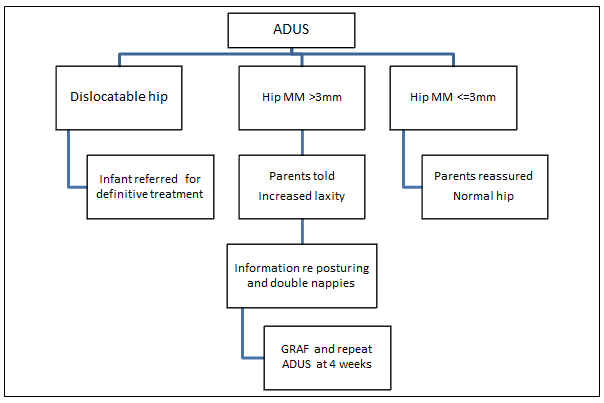
Figure 1: Anterior Dynamic Ultrasound (ADUS) research protocol.
GRAF, Graf method; MM, maximum movement of femoral head in relation to acetabulum.
Following ADUS, patient records were reviewed to determine the results of the physical examination of each infant's hips.
Differences for parental anxiety levels were tested using ANOVA, combining respondents into three groups (agreed, uncertain, disagreed) for the items concerning the acceptability of the ultrasound. The relationship between these items was tested using Spearman's Rho correlation coefficient. Differences between left and right hip were tested using a simple paired samples t-test. Significance was accepted as p<.05 for all analyses, which was undertaken using IBM SPPS Statistics 19 (www.spss.com).
Ethics approval
The study was approved by the Southern Adelaide Clinical Human Research Ethics Committee (Project 387.10).
During the 17 weeks of the study, there were 168 births in the SA regional hospital, from which 138 infants (82%) were eligible to participate in the study. Of these, 86 (62%) attended for screening. Mothers were a mean age of 29 years (SD 5.7 years).
All ADUS were provided by a single ultrasongrapher using a pre-programmed package. Her feedback was that most modern ultrasound machines would have the capability to record video clips and perform the necessary measurements; and that would it be quite an easy ultrasound examination to teach a clinician.
Only 2% of parents agreed that it was very inconvenient to attend the hospital radiology department for the ADUS, and 90% of participants reported they were pleased to see the pictures of their baby's hips (Table 1). Only 6% of participants did not agree that the ADUS was quick and did not seem to hurt the baby.
Table 1: Parents' perceptions of scan, and knowledge of baby problems
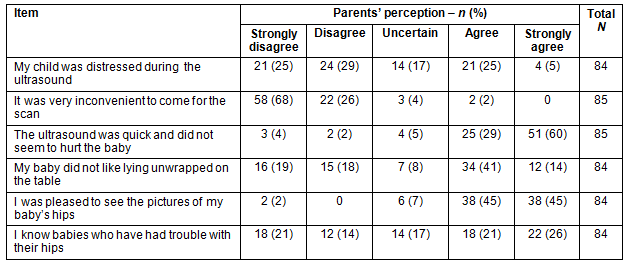
Parents who agreed or strongly agreed that their child was distressed during ADUS reported greater levels of anxiety (F=3.21, df=4 [p=0.017]) than those who were uncertain or disagreed that their child was distressed during the procedure. Parental gender was not a significant confounding factor for level of parental anxiety in this study; however, there were only 11 fathers involved in the study. There was a weak positive correlation between parents who reported knowing babies who had trouble with their hips and how pleased parents were to see the picture of their baby's hips (r=0.19, n=84, p=0.04.
In this pilot 45% of babies were female. Infants had a mean femoral head movement of 1.27 mm (SD 0.66 mm) in their left hip and a mean femoral head movement of 1.05 mm (SD 0.8 mm) in their right hip. This difference in left and right hip movement was significant (t=2.071; df=81; p [2-tailed]=0.04). There were no significant differences in hip movement by sex, gestational age, infant age at scan, method of delivery or family history.
Of the infants in the study, two were referred for follow-up scans at four weeks (Table 2). One met the criteria for a follow-up scan based on femoral head movement of both hips (left 3.4 mm; right 4.1 mm). The second met the criteria on maximum movement of the left hip (left 3.3 mm; right 0.9 mm). The second child also had a strong family history of developmental dysplasia of the hip but was not reported in the record to have a detectable abnormality on clinical examination at birth. The three hips with increased hip movement at the initial ADUS were found to be normal at the four week review.
Table 2: Maximum movement femoral head results of Anterior Dynamic Ultrasound (ADUS)
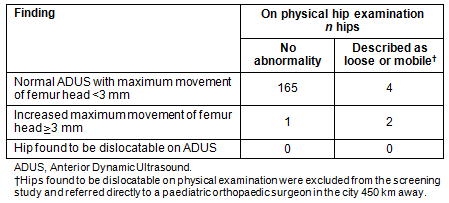
The maternity doctors identified three infants with hips 'loose/mobile' in the infants' medical records. One of these was confirmed by ultrasound (left 3.4 mm; right 4.1 mm), but the other two did not demonstrate abnormal movement under ultrasound (left 0.7 mm; right 0.6 mm vs left 2.2 mm; right 2.4 mm).
Discussion
The 2010 Australian Cochrane review on screening for DDH found insufficient evidence to recommend routine ADUS screening of all neonates20 but the study did not take into account the higher rates of late diagnosis in rural areas. As the cause for this finding is presumed to be reduced expertise in physical examination by rural generalist doctors8, the authors considered additional clinical training as a potential intervention21. However this intervention was considered unlikely to be cost-effective and efficient, because rural doctors already have significant ongoing professional development demands to maintain their broad range of clinical skills22. Recognizing this, it is important to consider the potential for a ADUS screening program in rural areas in Australia.
This pilot study of neonates for DDH has demonstrated that it is logistically feasible in an Australian regional hospital to introduce an ADUS screening program. During this small pilot study there was no cost to participants; however, with no Medicare (universal government health insurance) rebate currently available for ADUS screening in rural Australia, the financial cost of ultrasonography, particularly as an assistant is required to perform Barlow's manoeuvre, is inhibiting. Costs could potentially be reduced through role substitution. A recent study showed that clinical staff could, with focussed training, perform a Graf ultrasound with equivalent accuracy of results to experienced ultrasonographers23.
The ADUS was found to be an accessible screening test, with the small minority of study participants reporting attendance to be inconvenient. One weakness of this implementation pilot is that further information was not sought from parents who did not bring their infant for screening. However, having 62% of eligible patients attend an ADUS session provided on only one day per week suggests that the prevalence of neonatal screening would be increased should this service be available throughout the week. Providing this access in a multi-site trial will require careful measurement of human and financial resource implications for rural health services.
Acceptability of the ADUS was high, with the majority of participants finding the scan to be quick, perceiving it as comfortable for their baby (except that their baby did not like lying unwrapped), and being pleased to see the picture of their baby's hips. A total of 30% of babies demonstrated some level of distress during the ADUS and this did increase parental anxiety for a short time, but not to an unacceptable level. These results are consistent with previous study findings which highlighted the importance of the provision of clear information prior to ultrasound screening and effective communication during screening24.
The incidence of hips found to have increased maximum femoral head movement (>3 mm) in this study was 1.7%, which is close to the incidence of hip instability found on clinical examination in the neonatal population in a previous Australian regional study (19 per 1000 births)25; however, it is important to recognise that this represents reduced hip stability, requiring further follow up, not DDH. The likelihood of false positive ADUS results in this study was considerable because the researchers were deliberately very conservative with MM threshold of 3 mm, due to the initial inexperience of the ADUS screening team. In previous studies by Andersson and others the threshold for diagnosis of hip instability has been a maximum movement of 4 mm to 6.5 mm14,26. The increased risk of false positives was deemed acceptable in the pilot because the intervention experienced by these neonates was non-invasive, consisting only of double nappies, posturing and further ultrasound examination at 4 weeks (ADUS and Graf). If a future, large-scale study is conducted, it would be possible to increase the MM threshold for diagnosis provided an adequate quality assurance program existed for all rural ADUS providers. Electronic recording and storage of scans used in this implementation pilot demonstrates that this would be feasible. The optimal cut-off value for MM would have to be determined prospectively during any large scale study in order to be reliable.
In this pilot there were four hips (2.3%) reported as loose or mobile on physical examination which were found to be normal on ADUS. This suggests a high number of clinical examination false positives. This might be explained as a Hawthorn effect, with GP obstetricians more likely to report instability during the study period when their examinations were being scrutinized. It is more likely that this result confirms the findings of other studies that clinical examination can result in false positives when the pelvis is not fully stabilised with the Barlow manoeuvre4, and that movement is misinterpreted as intra-articular by less experienced clinicians12. There is, however, the possibility that these four hip results represented ADUS false negative results, but the study design did not allow the researchers to calculate the rate of these. They are unlikely to represent false negatives because the MM threshold in this pilot was so low (3 mm). Previous studies have not reported significant false negative rates7.
Finally, Andersson proposes that there is no evidence that DDH can occur if hip instability is not present at birth14. Despite this bold claim, ADUS screening has not eliminated the late diagnosis of DDH in some countries14,20,27. It is possible that the late diagnosis of DDH is caused by a mechanism other than failure of diagnosis at birth. This is supported by the evidence that while the majority of infants with high hip laxity at birth improve spontaneously, a few do not, and to date it has not been possible to predict these reliably7,27. An alternative explanation for the high frequency of late diagnosis of DDH in rural compared with urban SA areas is that high compliance with Sudden Infant Deaths (SIDS) nursing protocols (to posture infants swaddled on their back during sleep) potentially increases hip instability and results in poor femoral head coverage by the acetabulum in the postnatal period28,29. It is certainly true that babies wrapped with their legs held in adduction and extension are more at risk30. These issues complicate the story and demonstrate the importance of rigorous follow-up studies in rural Australia.
This implementation pilot has demonstrated that a research protocol is feasible for a large scale rollout of Anterior Dynamic Ultrasound (ADUS) in regional Australia to test whether more effective neonatal screening and a focused education program can reduce the number of infants with DDH late diagnosis and associated considerable morbidity.
Acknowledgements
This project was supported by: an 'In Front Out Back' grant from Toowoomba Hospital Foundation awarded at the National Rural Health Alliance Conference in Perth in 2011. The researchers also acknowledge Benson Radiology which supported staff member Lyn Muir to contribute her time and expertise to the project, including performing all ADUS screening examinations.
References
1. Gibson CS, van Essen PB, Scott H, Baghurst P, Chan A, Scheil W. 2007 Annual Report of the South Australian Birth Defects Register, incorporating the 2007 Annual Report of Prenatal Diagnosis in South Australia. Adelaide: SA Birth Defects Register Children, Youth & Women's Health Service, 2010.
2. Paton RW, Hopgood PJ, Eccles K. Instability of the neonatal hip: the role of early or late splintage. International Orthopaedics 2004; 28(5): 270-273.
3. Ekelund U, Luan J, Sherar L, Esliger D, Griew P, Cooper A. Moderate to Vigorous Physical Activity and Sedentary Time and Cardiometabolic Risk Factors in Children and Adolescents. JAMA 2012; 307: 704-712.
4. Barlow T. Early diagnosis and treatment of congenital dislocation of the hip. Journal of Bone and Joint Surgery 1962; 44-B: 292-301.
5. Rosen S. Diagnosis and treatment of congenital dislocation of the hip joint in the newborn. Journal of Bone and Joint Surgery 1962; 44: 284-291.
6. Graf R. Classification of hip joint dysplasia by means of sonography. Archives Orthopaedic and Trauma Surgery 1984; 102: 248-255.
7. Finnbogason T. Dynamic Ultrasonography in neonatal hip instability and acetabular dysplasia. PhD Thesis; Department of Women and Child Health Karolinska Institutet, 2008.
8. Lisle R, Boekelaar M, Stannage K, Whitewood C. Delayed diagnosis of developmental dislocation of the hip: the Western Australian experience. Australian and New Zealand Journal of Surgery 2012; DOI: 10.1111/j.1445-2197.2012.06110.
9. Sharpe P, Mulpuri K, Chan A, Cundy P. Differences in risk factors between early and late diagnosed developmental dysplasia of the hip. Archives of Disease in Childhood (Fetal and Neonatal edn) 2006; 91: F158-F162.
10. Chan A, Scheil W, Scott J, Nguyen A-M, Sage L. Pregnancy outcomes in South Australia, 2009. Adelaide, SA: Pregnancy Outcome Unit, SA Health, Government of South Australia, 2011.
11. Finne PH, Dalen I, Ikonomou N, Ulimoen G, Hansen TW. Diagnosis of congenital hip dysplasia in the newborn. Acta Orthopaedica 2008; 79(3): 313-320.
12. Dezateau C, Brown J, Arthur R, Karnon J, Parnaby A. Performance, treatment pathways, and effects of alternative policy options for screening for developmental dysplasia of the hip in the United Kingdom. Archives of Disease in Childhood 2003; 88: 753-759.
13. Clarke N, Reading I, Corbin C, Taylor C, Bochmann T. Twenty years experience of selective secondary ultrasound screening for congenital dislocaton of the hip. Archives of Disease in Childhood (Fetal and Neonatal edn) 2012; 97: 423-429.
14. Andersson JE. Neonatal Hip Instability: results and experiences from ten years of screening with the anterior-dynamic ultrasound method. Acta Paediatrica 2002; 91(8): 926-929.
15. Andersson J, Funnemark P. Neonatal hip instability: screening with anterior-dynamic ultrasound method. Journal of Pediatric Orthopedics 1995; 15(3): 322-324.
16. Mahan S, Katz JN, Kim Y. To Screen or not to screen? A decision analysis of the utility of screening for developmental dysplasia of the hip. Journal of Bone and Joint Surgery 2009; 91(1): 705-719.
17. Afaq A, Stokes S, Fareed H, Zadeh H, Watson M. Ultrasound in the selective screening of developmental dysplasia of the hip. European Review of Medical Pharmacological Science 2011; 15(4): 394-398.
18. Stanton B, Kaljee L, Lunn S, Deveaux L, Li X, Chen X, et al. Implementation of effective health innovations and pediatricians. Clinical Pediatrics 2011; 50(11): 995-1000.
19. Marteau TM, Bekker H. The development of a six-item short form of the Spielberger state-trait anxiety inventory (STAI). British Journal of Clinical Psychology 1992; 31: 301-306.
20. Shorter D, Hong T, Osborn DA. Screening progra mmes for developmental dysplasia of the hip in newborn infants (Review). Cochrane Database of Systematic Reviews 2011; (9): art. no: CD004595. DOI: 004510.001002/14651858.CD14004595.
21. Brown J, Dezateau C, Karnon J, Parnaby A, Arthur R. Efficiency of alternative policy optios for screening for developmental dysplasia of the hip in the United Kingdom. Archives of Disease in Childhood 2003; 88: 760-766.
22. McGrail Mr, Humphreys JS, Joyce CM, Scott A, Kalb G. How do rural GPs' workloads and work activities differ with co mmunity size compared with metropolitan practice? Australian Journal of Primary Care. (Online) 2011. Available: http://dx.doi.org/10.1071/PY11063 (Accessed 10 September 2012).
23. Ramwadhdoebe S, Sakkers R, Uiterwaal C, Boere-Boonekamp M, Beek F. Evaluation of a training program for general ultrasound screening for developmental dysplasia of the hip in preventive child health care. Pediatric Radiology 2010; 40(10): 1634-1639.
24. Witting M, Boere-Boonekramp M, Fleuren M, Sakkers R, IjJzerman M. Determinants of parental satisfaction with ultrasound hip screening in child health care. Journal of Child Health Care 2012; 16(2): 178-189.
25. Goss P. Successful screening for neonatal hip instability in Australia. Journal of Paediatric Child Health 2002; 38: 469-474.
26. Andersson J. Noenatal hip instability: normal values for physiological movement of the femoral head determined by an anterior-dynamic ultrasound method. Journal of Pediatric Orthopaedics 1995; 15(6): 736-740.
27. Roovers E, Boere-Boonekamp M, Mostert A, Castelein R, Zielhuis G, Kerkhoff T. The natural history of developmental dysplasia of the hip: sonographic findings in infants of 1-3 months of age. Journal of Pediatric Orthopaedics 2005;14(5): 325-330.
28. van Sleuwen B, Engelberts A, Boere-Boonekamp M, Kuis W, Schulpen T, L'Hoir M. Swaddling: a systematic review. Pediatrics 2007; 129: e1097-e1106.
29. Gilbert R, Salanti G, Harden M, See S. Infant sleeping position and the sudden infant death syndrome: systematic review of observational studies and historical review of reco mmendations from 1940 to 2002. International Journal of Epidemiology 2005; 34(4): 874-87.
30. Storer S, Skaggs D. Developmental dysplasia of the hip. American Family Physician 2006; 74(8): 1310-1316.




