Introduction
The regulation of health professionals and their training requirements provides essential but complex protections for the public1. In Australia, these protections include the introduction of a National Registration and Accreditation Scheme for health practitioners (NRAS) in 2010 and the establishment of the Australian Health Practitioner Regulation Agency (AHPRA)2. Other levels of control or regulation are also exerted over health professionals via mechanisms such as salaries and funding arrangements, insurance requirements, admitting rights to healthcare facilities, and legislation controlling the use of medicines and therapeutic devices2,3.
Access to essential medicines is a human right in the International Treaty on Social, Economic and Cultural Rights4. Its importance is also evidenced in the Declaration of Alma Ata5 and the Sustainable Development Goals6. Australia is a signatory to each of these instruments, and appropriate and timely access to medicine is also a central objective of Australia’s National Medicines Policy7. Australia’s Pharmaceutical Benefits Scheme (PBS) underpins affordable access to essential medicines. In 2016–2017, approximately 0.7% of the nation’s gross domestic product (A$12.06 billion) was spent on medicines subsidised through the PBS8, reflecting their critical, varied role.
In Australia, when episodes of health care involve the use of medicines it is acknowledged that the Medication Management Cycle (MMC) will occur9,10. The MMC has been described as consisting of nine steps9,10:
1. deciding to prescribe a medicine
2. recording the medication order or prescription
3. reviewing the medication order/prescription
4. issuing the medicines, which includes manufacturing and dispensing or supplying the medicine
5. providing medicines information to the consumer and those involved in storing and administering the medicine
6. appropriately distributing and storing the medicine
7. administering the medicine
8. monitoring the consumers’ response to the medicine
9. communicating or transferring verified information regarding medicines use and treatment plans.
While many healthcare professionals may be involved in different phases of the MMC, in Australia, medical practitioners and pharmacists are generally acknowledged as responsible for the first six. Nevertheless, because medications play such a vital role in health care and because other professionals, including nurses, are also expected to play at least some role in the MMC, instruments that provide a means of effecting control and coordination of use must have demonstrable relevance.
Among such instruments, legislation is key to clarifying where, by whom and how acts of prescribing, dispensing, supplying and administering medications should occur. Within Australia’s busy, federated legislative landscape, there are many opportunities for incongruence between instruments that pertain to medicines. Although this has been identified as a risk to patients and health professionals11-15, little has been published about the issue with regard to registered nurses (RNs) and no complete analysis has been undertaken from a nursing perspective since the introduction of the NRAS and AHPRA in 2010 and subsequent changes to medicines legislation14,16.
This is a significant gap because RNs and RN/midwives are the mainstay of the Australian health professional workforce, comprising more than half of its total (53.6% in 2017) and are the most evenly distributed across the nation17-19 (Fig1). Moreover, in the remote and very remote areas that constitute most of Australia’s land mass and where community health and wellbeing are generally poorest20, the role of RNs is especially significant, with many communities reliant on nurse-led services21,22. In these parts of the nation, RNs have historically been and continue to be the mainstay and therefore tend to carry more responsibility for healthcare delivery, including the MMC21-23.
The nurse practitioner (NP) role has been recognised in Australia since 2000. In Australia, NPs are RNs who have completed postgraduate training at a masters level and have applied for and hold the specific NMBA Nurse Practitioner endorsement. NPs are authorised prescribers within their individual speciality areas and are able to apply for Pharmaceutical Benefits Scheme (PBS) prescribing rights24. Despite initial hopes that NPs would fill the medical practitioner void in remote Australia, Figure 1 highlights that this has not been the case to date. Indeed, according to 2017 health workforce figures an additional 481 NPs would be required in very remote Australia to bring the combined medical practitioner-to-NP ratio in line with that of major cities17-19.
The extended role of nurses in medication management in remote Australia has been acknowledged from the earliest days of the PBS25. In 1999, amendments made to the National Health Act 1953 effectively established the Remote Area Aboriginal Health Service program, enabling the free supply of PBS medications without a prescription at designated facilities in remote areas26. This removed a barrier to accessing affordable medicines due to a lack of local medical practitioners for obtaining prescriptions and pharmacies for the dispensing of medications. Despite this, it is unclear if nurses in these areas, whose roles have in common some elements of work usually undertaken by general practitioners or pharmacists21 and may therefore include supplying and/or initiating medications, are also supported to do this by state or territory legislation governing the practices of health practitioners with regards to medicines.
Despite the significance of these advanced practice roles for RNs and the crucial position they therefore hold in the quality use of medicines there has, been little research outside of Queensland into RNs and the MMC in remote Australia or the legislative and practice frameworks that might best support quality use of medicines by nurses in remote Australia.
The objective of this study was to examine medicines and poisons legislation nationally from a nursing perspective, with a focus on the context of health service delivery in remote areas of Australia.
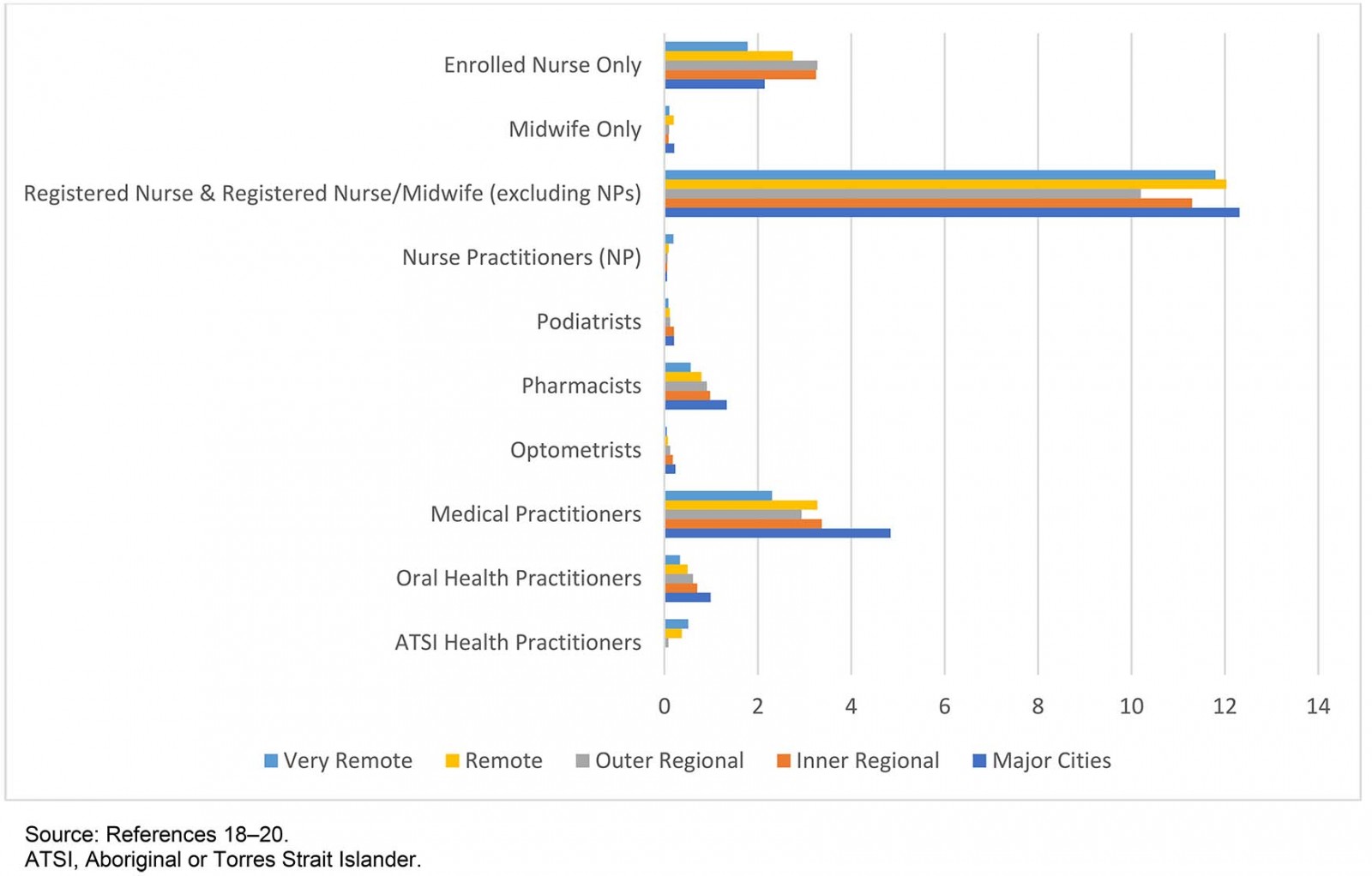 Figure 1: 2017 health professional by type per 1000 estimated resident population and Australian Statistical Geography Standard – Remoteness Area 2016.
Figure 1: 2017 health professional by type per 1000 estimated resident population and Australian Statistical Geography Standard – Remoteness Area 2016.
Methods
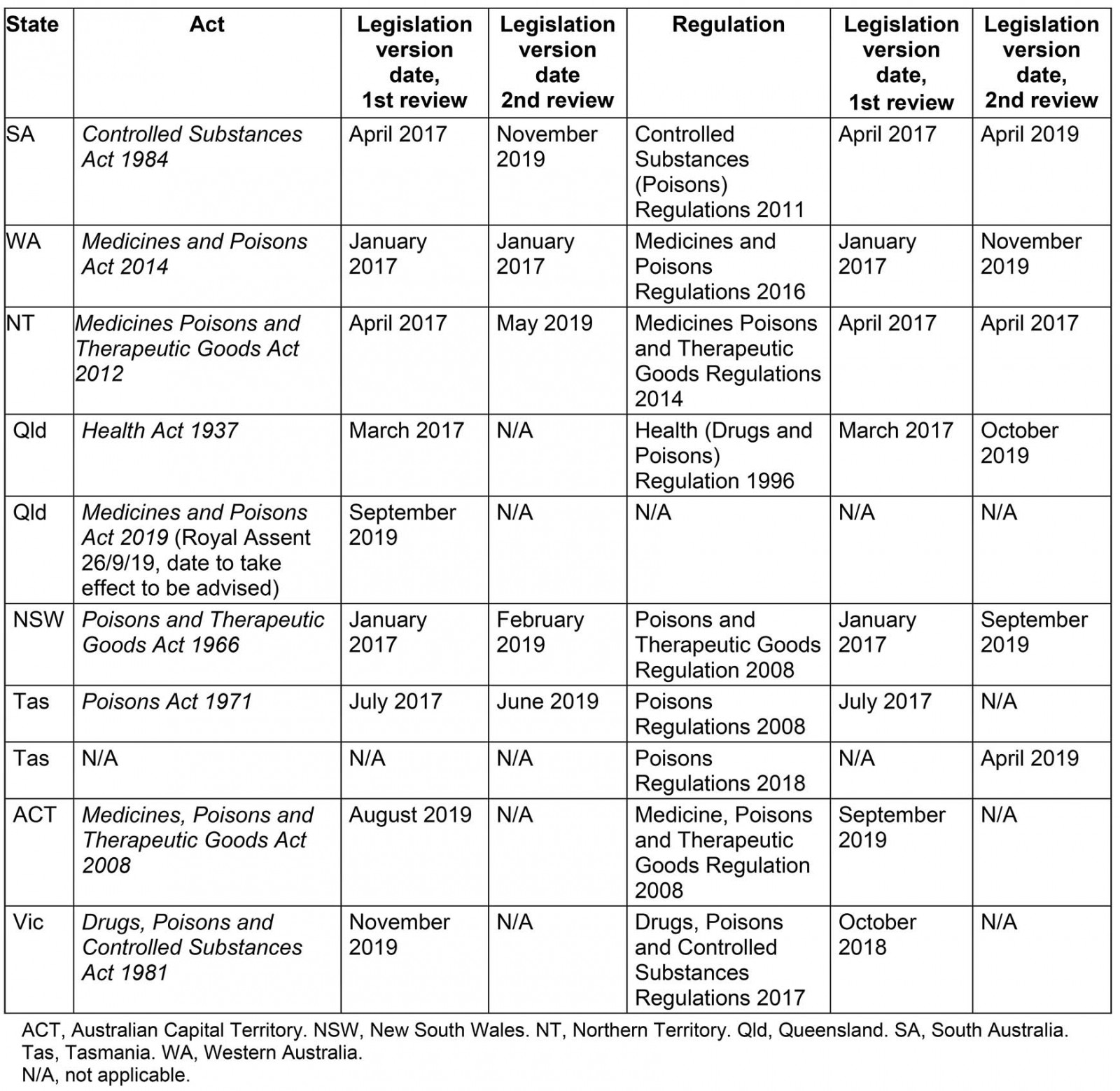
In August 2017, state and territory medicines legislation and regulations for all Australian states and territories that have areas classed as ‘very remote’ according to the Australian Statistical Geography Standard – Remoteness Area 2016 (ASGS-RA 2016)27 were interrogated for answers to the questions ‘Can an RN prescribe a medication?’, ‘Can an RN dispense a medication?’, ‘Can an RN supply or issue a medication?’ and ‘Can an RN administer a medication?’ in regards to RNs who do not hold NP endorsement. The legislation was again reviewed in 2019 for changes and amendments; at this time Victoria and the Australian Capital Territory were included in analysis for National completeness (Supplementary table 1). The medicines and poisons, or similarly titled Act and Regulations for each state and territory, will hereafter be referred to by abbreviated state or territory name and ‘Act’ or ‘Regulation’.
Findings will be presented as descriptions of the relevant definitions, authorisations and sections of legislation accessed in order to answer the identified questions for Schedule 2, 3, 4 and 8 medicines. Tables will be used where possible to summarise findings.
Ethics approval
Ethics approval was granted from the Edith Cowan University Human Research Ethics Committee (Approval 19386).
Results
Instrument and format
The title of legal instruments and location of legislative details regarding authorisations for RNs regarding medicines varies across Australian states and territories. In South Australia (SA), the Northern Territory (NT) and New South Wales (NSW) most of these details are contained within the relevant Act. In Queensland, the Australian Capital Territory (ACT), Victoria, Tasmania and Western Australia (WA), the details are predominantly located within Regulations.
In addition to Acts and Regulations, other regulatory processes such as gazetting or the issuing of individual authorisations or licences are enabled in legislation and utilised variably across the nation.
Scheduling of medicines
All Australian states and territories are party to a federal legislative instrument, the Poisons Standard (2019), also known as the Standard for the Uniform Scheduling of Medicines and Poisons (SUSMP). The terminology used to describe and refer to the different schedules of medicine within both state and territory legislation differs from the SUSMP (Table 1).
Table 1: Standard for the Uniform Scheduling of Medicines and Poisons definition and terminology used to describe medicine schedules in Australian legislation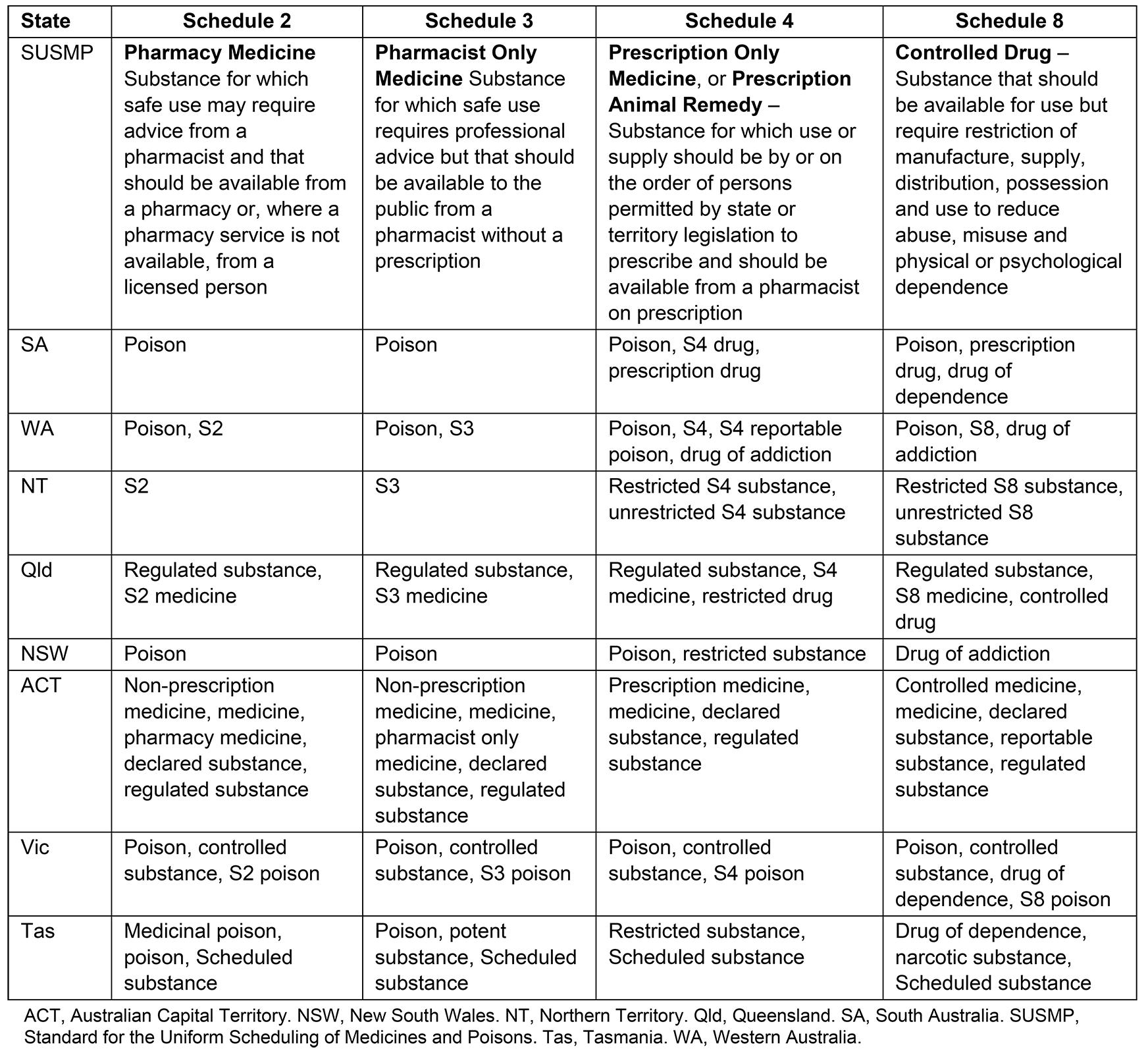
Endorsements, training, authorities and protocols
A variety of mechanisms across Australia make provisions for RNs to work more broadly through the MMC. The National Health Practitioner Law Act 2010, as enacted by each state and territory and enabling the powers and responsibilities of NRAS and AHPRA, makes provision for suitably qualified, registered health professionals to apply for an endorsement in a certain area of practice28. RNs who have undertaken approved training can apply to the Nursing and Midwifery Board of Australia (NMBA) for an endorsement for scheduled medicines for RNs (rural and isolated practice) (Registration Standard for Endorsement of Registered Nurses (Rural and Isolated Practice) –RIPEN)29. This endorsement is recognised in legislation in all states and territories with the exception of WA and Tasmania29. However, Queensland and Victoria are the only states in which RIPEN is actually needed for an RN to undertake broader aspects of medication management practice. The NMBA is preparing to discontinue RIPEN29.
In NSW, a letter of appointment may allow RNs to supply and/or initiate listed medications according to Standing Orders for the Supply or Administration of Medications under the Nurse Designated Emergency Care (NDEC) model and related Guidelines and Medication Standing Orders30. This entails training and certification that is only available and recognised in a limited number of NSW rural and remote facilities. In WA, RNs providing sexual health services are required to undertake additional training to be able to initiate medications according to specific protocols. There is no requirement in WA for RNs in remote nursing roles to undertake additional education prior to initiating and supplying medications according to remote area nursing protocols31. Similarly, in SA, the NT, ACT and Tasmania, there are no requirements for RNs to have undertaken additional education to supply medications according to a protocol.
Definitions and the terminology describing protocols and standing orders also vary across the country, and different protocols are authorised through state or territory legislation to allow RNs to undertake extended MMC roles. Both Queensland and Victoria utilise the Primary Clinical Care Manuals (PCCM) for most nurse-managed care for acute presentations in remote contexts. In the NT and at multiple remote health sites in SA32 the Remote Primary Healthcare Manual (RPHCM) suite has been adopted. The NDEC Nursing Management Guidelines (NDEC NMG) and Medication Standing Orders are utilised at limited sites in NSW30. The PCCM, RPHCM and NDEC NMG provide guidance on the assessment and management for common presentations and protocols for the use of specific drugs. South Australian and Tasmanian legislative mechanisms do not stipulate the use of specific manuals or protocols. WA utilises a system of Structured Administration and Supply Arrangements (SASAs) to authorise administration and supply of medications by RNs in certain contexts. A variety of SASAs are issued by the state, and mechanisms exist for health organisations or individual medical practitioners to develop their own SASAs for use by RNs31. Unlike the RPHCM, PCCM and NDEC NMG, SASAs only relate to a specific medication, its indication for use and dose. SASAs do not provide guidance for RNs around patient assessment, differential diagnoses or clinical decision-making guidance in respect to the condition being treated. Endorsements and training requirements relating to RNs who are immunisation providers were not included in this analysis due to their multitude and complexity.
Prescribe, prescriber, prescription
Prescribing traditionally relates to stages 1–3 of the MMC.
Definition: Definitions of terms, where provided, are in the Act for WA, the NT and Queensland. SA defines only prescriber in the Regulations. The ACT defines prescribe and prescription in the Act and prescriber in the Regulations. No clear definitions are provided by NSW or Tasmania. Victoria defines prescription by listing two provisions of what a prescription is not within the Regulations (Table 2).
Authorised actions for RNs: RNs without an NP’s endorsement are not authorised to prescribe medications in any practice context. However, under the new Queensland Medicines and Poisons Act 2019, there are provisions for individuals to apply for a prescribing approval, and this could include RNs.
Table 2: Definitions and authorised actions for prescribe and dispense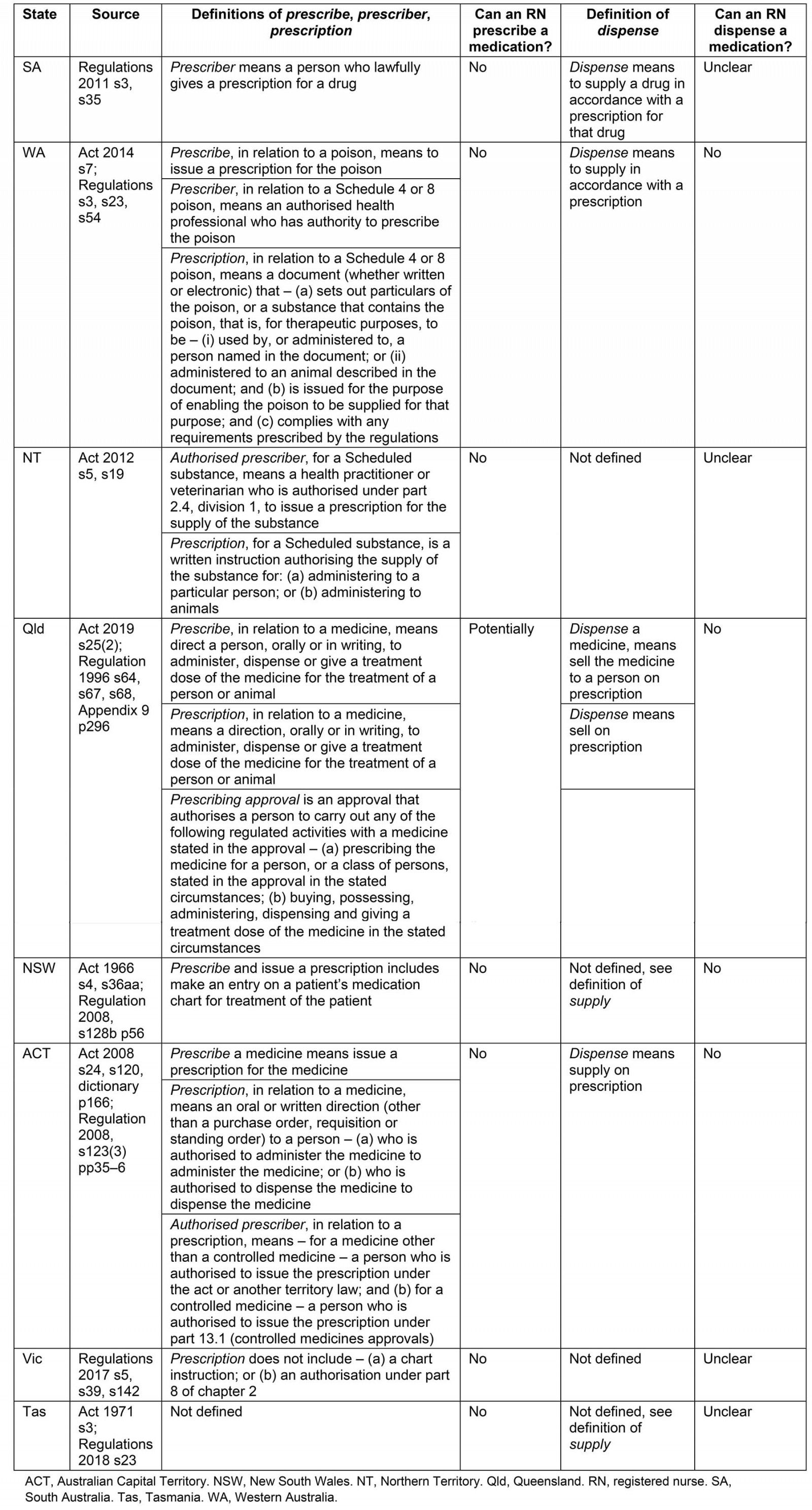
Dispense
The dispensing of medications traditionally involves stages 4–6 of the MMC.
Definition: The ACT defines dispense in the Act, and SA and WA within the Regulations similarly as being to supply a drug according to a prescription. Queensland also defines the term within its Regulations but specifies that dispensing is to sell a drug in accordance with a prescription. The NT makes no reference to the term. Victoria does not define the term. NSW and Tasmania do not define the term but in these states supply includes the act of dispensing, with Tasmania specifying that dispensing occurs in accordance with a prescription (Table 3).
Authorised actions for RNs: SA only mentions pharmacists and medical practitioners in the more than 12 provisions for actions that must be undertaken when dispensing. RNs in WA and Queensland are not authorised to dispense. Dispensing is not a specific action regulated in the NT. Pharmacists are the only health professionals authorised to dispense in NSW. The situation in Tasmania is unclear because person is undefined when referring to the dispensing of narcotic substances and RNs’ status related to dispensing is not clarified. Dispense is not defined by Victoria; however, authorised (RIPEN) RNs may sell Schedule 3, 4 and 8 medications to patients under their care if there is a therapeutic need; there is no stipulation that a prescription is required.
Table 3: Definitions and authorised actions for supply
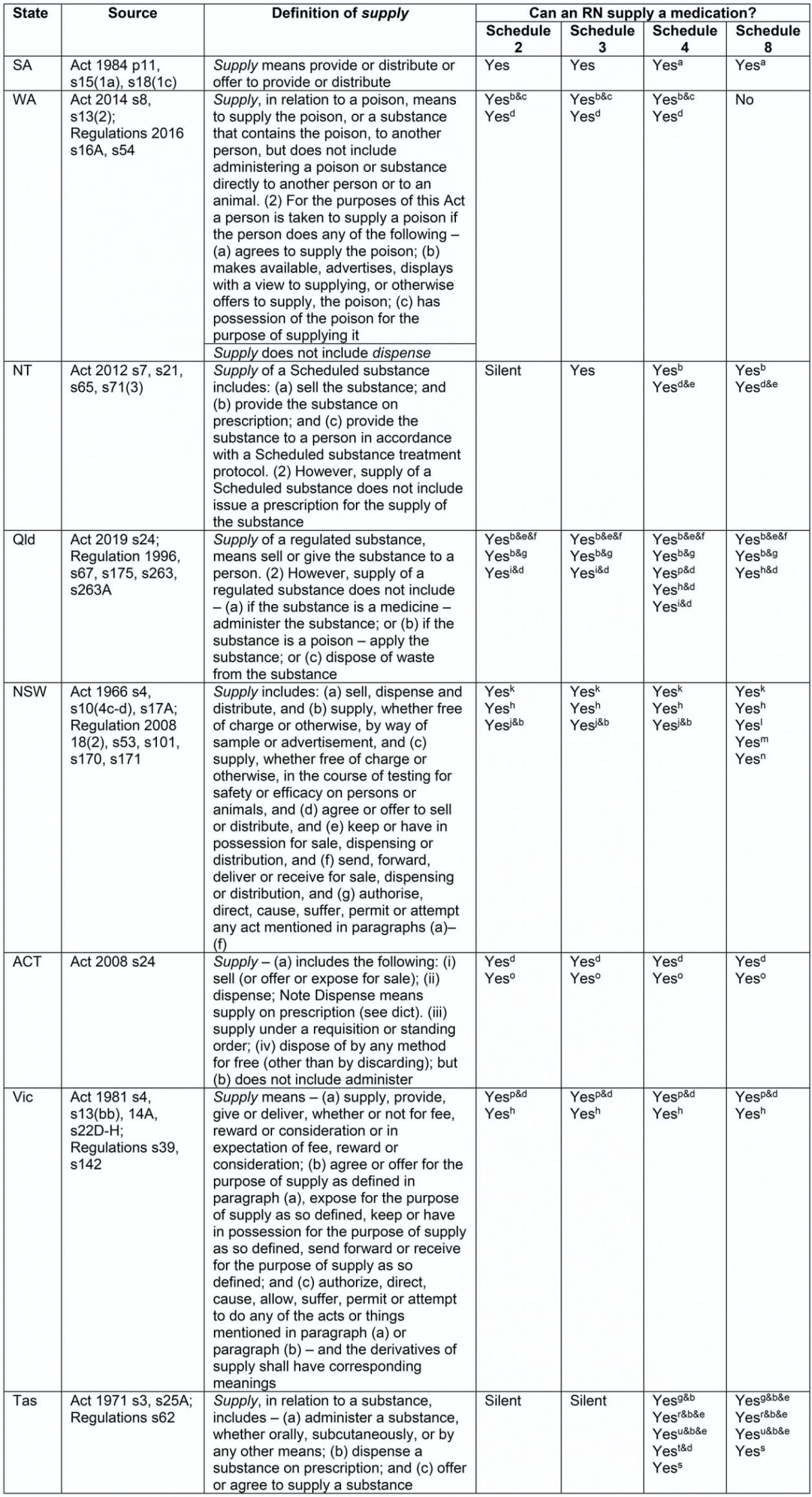

Supply
The supply of medications may relate only to stages 4–6 of the MMC. However, where supply is authorised without a prescription, stages 1–3 of the MMC will also be occurring. For example, where an RN is authorised to supply any medication without an order from a prescriber, the RN is then required to assess and use clinical reasoning to determine the need for medication; make a decision about appropriate medication use either independently or according to a protocol; record information about the medication route, dose and timing; and the supply and/or administration of the medication to the patient.
Definition: Although the Acts in SA, WA, NT, Queensland, NSW, ACT, Victoria and Tasmania all define supply, the definitions are different. Acts in WA, Queensland and the ACT specifically mention that supply does not include administer, whereas in Tasmania the term supply includes administer.
Authorised actions for RNs: Table 3 summarises differences in authorised actions for RNs in relation to the supply of medicines. Twenty-one variable provisions relating to supply authorisations were identified.
Schedule 2 and 3 medications There is no national consistency relating to the supply of Schedule 2 or 3 medications by RNs (Table 3).
Schedule 4 medications There is no national consistency relating to the supply of Schedule 4 medications by RNs (Table 3).
Schedule 8 medications There is no national consistency relating to the supply of Schedule 8 medications by RNs (Table 3).
Administer
Traditionally administering a medication is stage 8 of the MMC. However, similar to the situation with supply of medications, where administration is authorised without an order from an authorised prescriber then steps 1–6 of the MMC may also be occurring.
Definition: Administer is a frequently used term in legislation. It is not defined in the Acts or Regulations of SA, WA, NSW, ACT, Victoria or Tasmania (Table 4).
Authorised actions for RNs: Significant variations exist regarding administration authorisations for RNs across Australian states and territories, summarised in Table 4. Due to their complexity and multitude, provisions regarding RNs and the administration of Schedule 4 medicines that are also vaccines have been excluded from this analysis. All findings regarding authorised actions in Queensland relate to authorisations made under the Health (Drugs and Poisons) Regulation 1996 which remains in effect until the new Medicines and Poisons (Medicines) Regulations 2019 have been finalised and enacted.
Schedule 2 and 3 medications There is no national consistency relating to the administration of Schedule 2 or 3 medications by RNs (Table 4).
Schedule 4 medications Nationally, RNs are consistently authorised to administer a Schedule 4 medication on the order of an authorised prescriber. Several variations exist nationally in additional authorisations for RNs relating the administration of Schedule 4 medicines (Table 4).
Schedule 8 medications Nationally, RNs are consistently authorised to administer a Schedule 8 medication on the order of an authorised prescriber. Several variations exist nationally in additional authorisations for RNs relating the administration of Schedule 8 medicines (Table 4).
Table 4: Definitions and authorised actions for administer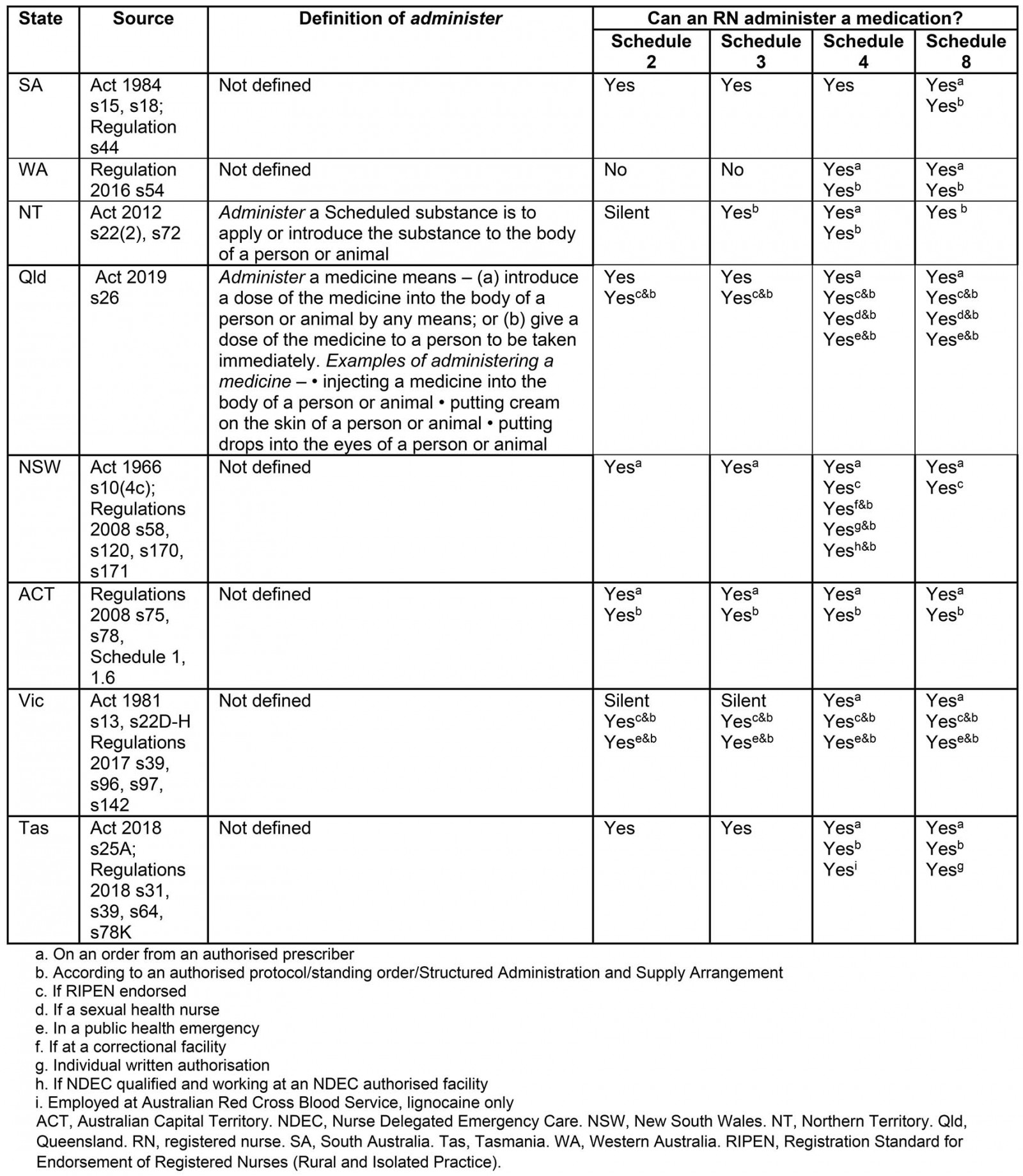
Discussion
Nationally inconsistent medicines and poisons legislation and related legal instruments affect the capacity for RNs in Australia to understand and therefore practise confidently within their legal scope. With the burgeoning demand for healthcare services, changing models of health service delivery, and new and emerging health issues, it is vital that systems regulating health professional practice adapt quickly.
Features of the Australian legal system and the processes involved in effecting change to Acts and Regulations hamper the ease with which adaptations can be made to accommodate changing healthcare delivery needs, such as in the management of a public health outbreak or health professional staffing crisis.
The format and content of legislation pertaining to the use of medicines also vary considerably. Authorised actions for health professionals are difficult to locate, collate and interpret where they are not presented by profession, authorised action and the schedule of medicine, or where summary tables of authorised actions by profession are not provided.
Inconsistent definitions in the legislation also affect the ease with which legislation can be interpreted. Queensland is the only jurisdiction that defines all key terms. Nationally, terms such as supply are used inconsistently or used in some contexts but not others. Moreover, the use of several terms to describe a single schedule of medicine both within and across jurisdictions makes interpretation more complex and confusing.
The 28 identified variants in how RNs are authorised to work with medicines seems an unnecessary burden on nurses who need to be able to work consistently across Australia. The greatest impact of these inconsistencies is likely to be in remote areas, where RNs have responsibility for more of the MMC than their urban counterparts. Where RNs are authorised to undertake extended practice with regards to medications, such as supplying medications according to authorised protocols, differences in required training and preparation are significant and these impact on workforce mobility and quality of care.
These are important policy issues in a context of greater equity of access to health care and medications for people living in remote Australia. A lack of consistency in legislation relating to how RNs work with medications in Australia is a hidden, systemic cause of inequity impacting on nursing practice and directly affecting the health of those who live in remote parts of the nation. By far the greatest proportion of the people affected are Aboriginal and Torres Strait Islanders. Eliminating this hidden, systemic inequity will, therefore, contribute to closing the gap in life expectancy between Aboriginal and Torres Strait Islander and non-Indigenous Australians. Harmonisation of nursing practice in relation to all aspects of the medication management cycle via nationally consistent medicines legislation will improve equity of access to health care and medications. This will enhance the quality of care provided to those Australians whose health is generally poorest. However, this demands of decision makers a nationally collaborative approach and a willingness for policy and legislative reform efforts. An appetite and ambition for such reform has been characteristic in some areas of Australian health care, so it is really now just a question of the desire of decision-makers to act on this practical issue that will lead to improvements in health outcomes in remote Australia.
Limitations
This analysis has neither explored nor compared the legislative provisions relating to RNs and the possession of scheduled substances, nor the multitude of legislative requirements that exist for storing, labelling and destroying medicines. Also not explored are the requirements and provisions related to RNs and the administration of vaccines. The differing requirements of the state and territories regarding verbal orders from authorised prescribers have similarly not been explored. Although warranted, analysis of authorisations for both RNs who hold NP endorsement and authorisations for Aboriginal health workers and practitioners was not possible within the scope of this project.
Conclusion
Administrations of Schedule 4 and 8 medications according to an order from an authorised prescriber are the only actions authorised consistently for RNs across Australia. Other actions relevant to a specific schedule supported in one state or territory are not necessarily supported in others, and the regulated educational requirements for extended roles with medicines differ. This lack of consistency poses a significant risk to not only RNs working in remote areas but all RNs whose roles with medications exceed these two defined actions. A lack of uniformity between Australia’s medicines and poisons legislation means RN practice will vary across Australia and patients may not receive a standard or universal level of care.
It is imperative to acknowledge that in Australia a health professional’s individual scope of practice is defined jointly by legislation; their level of educational preparation, knowledge, skills and experience; the needs of the community; and the policies and procedures of their employing organisation. Any or all of these elements may be either broader or more limited than legislated roles with medicines. With Australia’s highly mobile workforce and high turnover rates in remote areas33, inconsistencies in legislated authorisations regarding medicines may result in RNs working either suboptimally or outside their legal scope.
Little has been published about the effects of legislative requirements on nursing practice and health service delivery in remote Australia, where health status is generally poorer and where access to health services and health professionals is more limited than in other parts of the country20. Additionally, although RNs who are not NPs are known to have been working in extended roles in remote Australia, little is known about how they are supported to undertake these roles involving medications. Further research is warranted to investigate the legislative models that best facilitate timely access to medicines in remote Australia and the education and experience that best prepare RNs to work in extended MMC roles within a quality use of medicines framework.
Access to essential medicines is a key component of the National Medicines Policy7. A lack of consistency between state and territory legislative instruments may affect people’s timely access to medicines in remote Australia and have significant implications for their health.
Health service delivery involving medicines in remote areas needs to be consistent across Australia to ensure health outcomes and health professionals in remote areas are not unnecessarily compromised. The present situation poses a considerable risk to the health and rights of many Australians, particularly those living in very remote areas where the vast majority of the population is Aboriginal or Torres Strait Islander. It also poses a considerable risk to the nursing workforce who may knowingly or not be undertaking aspects of medication management that are not authorised in their state or territory. Significant collaborative effort cognisant of models of healthcare delivery and the needs of people in remote Australia is required to address the issues identified.
Acknowledgements
KP received support from an Australian Government Research Training Program Scholarship. Dr David Scrimgeour reviewed the initial manuscript and provided writing assistance. Dr Ann-Claire Larsen provided technical advice on legal terms and reviewed the initial research proposal and manuscript.
References
Supplementary material is available on the live site https://www.rrh.org.au/journal/article/6020/#supplementary
You might also be interested in:
2003 - The meaning of health and well-being: voices from older rural women