Introduction
Diabetes among Aboriginal and Torres Strait Islander peoples occurs at a younger age and almost four times the rate of non-Indigenous Australians1. In a cross-country comparison of four countries whose Indigenous populations were adversely affected by British colonization, Australia stands out as having the highest age standardized mortality rates for deaths due to diabetes among Indigenous people2. While the cause for this disparity in diabetes incidence is multi-factorial, recent evidence suggests that nutrition, particularly magnesium intake, may play a role. A significant relationship between magnesium and whole-body glucose homeostasis and insulin sensitivity has been demonstrated3, with magnesium deficiency having been shown to induce insulin resistance even in normal subjects4. Dietary magnesium has also been found to be protective against the development of diabetes5,6. Low serum magnesium has been identified in both type 1 and type 2 diabetes7-9. The prevalence of low serum magnesium among outpatients with diabetes has been reported to be 38% in Switzerland and 25%-39% in the USA7,10. Other populations with diabetes might, however, be more prone to low magnesium levels. For instance, Rodriguez-Moran and Guererro-Romero11 reported 73.1% of out-patient diabetics in Mexico had low serum magnesium, while 93.9% of diabetic subjects with foot ulcers had low serum magnesium. In a separate study, the same investigators found that up to 60% of diabetic subjects had low serum magnesium12. Indeed, hypomagnesaemia is the most common electrolyte abnormality in diabetic outpatients, and may be linked to the development of both macrovascular and microvascular diabetic complications11.
While the causes of low serum magnesium are unknown, a number of factors have been proposed, including dietary intake, magnesium content in drinking water, and magnesium loss through sweat7,13. For Australia as well as the USA, the recommended dietary intakes (RDI) for magnesium is 400-420 mg/day for adult men and 310-320 mg/day for adult women14,15. Population studies now use the estimated average requirement (EAR), defined as the level needed to meet the daily requirements of half the healthy individuals in a particular life stage and gender group. The EAR for magnesium is set at 330-350 mg for adult men and 255-265 mg for adult women. The Australian National Nutrition Survey of 199516 found that the mean daily intake of magnesium was 381 mg for adult males and 283 mg for adult females, both below the RDI but still meeting the needs of half the population. Similar dietary surveys in the USA also indicated magnesium intake was frequently below the RDI14. Moreover, the Australian 1995 survey showed that dietary intake of magnesium was lower in those at greater socio-economic disadvantage, and lowest among obese persons.
Drinking water contributes to total magnesium intake and may provide 6% to 31% of the daily magnesium RDI17. Evidence associating water hardness (from magnesium and calcium salts) with cardiovascular and other chronic diseases has gathered momentum in the last three decades18,19. Not all research has supported this association20, but the balance of evidence supports a relationship between decreased chronic disease and increased water hardness21. Yet most urban water supplies remove magnesium as part of the water softening process.
Finally, blood magnesium levels are influenced by magnesium loss that can occur through sweat. Australia is called 'the sunburnt country', with vast areas of blistering desert, and the northern third of the country within the tropics. Accordingly, high temperatures and humidity are commonplace throughout much of the continent, with both of these factors expected to increase sweat loss. A review by Sawka and Montain22 indicated that heat adaptation increases sweat loss. Residents of desert climates often experience sweat rates of 0.3-1.2 L/hour while performing occupational activities. Magnesium losses in sweat are approximately 0.8 mmol/L (range: 0.2-1.5 mmol/L Mg or 19-365 mg/day)22. The only known studies on mineral sweat loss among Aboriginal people concluded that differences in sweat concentrations were not attributable to genetic differences but due to diet and lifestyle23; thus, other studies on human sweat loss may be considered applicable to Aboriginal and Torres Strait Islander peoples. With consideration of the Australian climate, there may be potential for significant magnesium loss through sweat. Indeed, Franz and Baily identified a climatic relationship to diabetes as well as heart disease, which they attributed to sweat loss of magnesium13.
In the present article, we examine the relationship between magnesium and diabetes in Australia, especially in relation to Indigenous Australians. Specifically, we explore the associations between diabetes and the magnesium content of drinking water and diet, as well as climatic and socioeconomic factors that may impact on magnesium intake, including temperature, rainfall, education, employment, and income. For an indication of dietary magnesium intake, 24 hour recalls from a convenience sample of Indigenous patients were assessed for magnesium content.
Methods
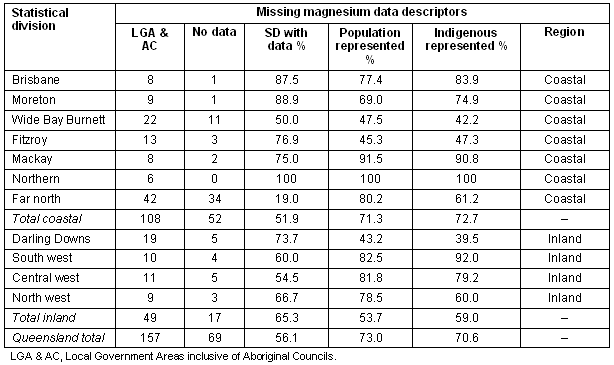
In total, 157 local government areas (LGA) and Aboriginal Councils in Queensland were identified and contacted regarding magnesium content of municipal water supplies (delivered product). Contact by telephone, email and/or facsimile was repeated a minimum of three times in an effort to obtain data from all areas. Due to difficulties many LGA had in locating water test records, data collection was limited to only magnesium levels in the drinking water for 2002. All test results were derived from chemical analysis performed as part of routine water safety monitoring and met Queensland Health laboratory standards. Australian drinking-water guidelines specify both health-related and aesthetic guideline values24. Although operational monitoring guidelines include a recommended assessment of water hardness as CaCO3, specific testing for magnesium is not required. Therefore, not all LGA were able to provide data on the magnesium content of drinking water (Table 1).
Table 1: Missing magnesium data by coastal or inland region
Age-standardized death rates for diabetes mellitus (International Classification of Diseases ICD-10 E10-E14), inclusive of diabetes as either underlying or associated cause of death in 2002, were obtained from the Australian Bureau of Statistics (ABS). All mortality data were derived from the Queensland Registrar of Births, Deaths and Marriages, where cause of death and usual residential address is recorded on death certificates. Due to small populations in some LGA, available data were limited to ABS statistical divisions (SD). Because of concerns regarding confidentiality, data were not available by Indigenous status. For additional points of comparison, the age-adjusted mortality data for the largest city in each SD and the balance of SD were obtained.
The Office of Economic and Statistical Research (OESR) provided data on the proportion of each local government area population with post-school education qualifications (diploma, certificate, advanced diploma, graduate diploma, post-graduate and bachelor degree), median weekly household income, and percent unemployment for each LGA. The original source was the ABS 2001 Census. Annual rainfall and average maximum daily temperature were also obtained from OESR SD profiles, with original data from the Australian Bureau of Meteorology. A geographically dispersed selection of sample sites was obtained for each LGA to provide a representative sample of climate. The percentage of the population identified as Indigenous was also included in the analysis.
Analysis of magnesium dietary intake was conducted on a subset of Indigenous participants in the Walkabout Together (WAT) Program, a prospective lifestyle intervention study of the Townsville Aboriginal and Islanders Health Service Ltd (TAIHS), targeting overweight (BMI >25) patients in a regional centre. Both the WAT Program and the magnesium investigation received ethics approval from the Townsville Health Service District Ethics Committee and the TAIHS Board of Directors. The TAIHS Board also served as the source of community consultation. Details on the WAT program health outcomes have been published elsewhere25. Nutrient analysis, completed as part of WAT program evaluation, served as a convenience sample for the magnesium investigation. Multiple-pass 24-hour dietary recalls were collected at baseline and at 12 month program completion. All dietary recalls were completed by two of the researchers (DL and DH) using a standardized interview process. Only Indigenous participants were included in the diet analysis. All dietary recalls were analysed using the software program 'FoodWorks Professional 2005' (Xyris Software (Australia) Pty Ltd) Brisbane, Qld, Australia;) that uses the Australian nutrient database. Dietary recalls with estimated intakes below 5000 kJ (1200 kcal) were eliminated from the analysis as implausible, because this was the lowest energy intake advised during dietary counselling. Goldberg's cut-offs were not used because this was not a weight-stable population.
Data analysis
Available water data were evaluated at the local level with regard to mean, median and range of magnesium content. Since age-adjusted mortality data was only available for the largest city in each SD and the balance of SD, mean magnesium was aggregated to the same areas. All data were tested for normal distribution using the Kolmogorov-Smirnov test with Lilliefors correction. Where log-transformation successfully normalized distributions, Pearson's correlation was used. For non-normal distributions, non-parametric correlation of Spearman's rho was used to verify the bivariate unadjusted linear associations. An independent sample t-test was used to examine differences between the cities and townships and the balance of the SDs. Additionally, replicating the statistical approach of Franz and Bailey11, we determined z-scores for rainfall and mean maximum daily temperature, and summed the z-scores to create a climate variable. A sum of z-scores for post-school qualifications, percent of population employed (inverse of unemployment), and mean weekly household income were used as a variable for socioeconomic environment. A non-parametric Mann-Whitney test was used to compare nutrient intakes per 1000 kJ against age- and sex-matched 1995 National Nutrition Survey nutrient intakes per 1000 kJ. All statistical analysis was done using SPSS v13.0 (SPSS Inc; Chicago, IL, USA;). For spatial analysis, 'Map Info Professional' v 7, a map software program used by the Australian Bureau of Statistics, was used to geo-code diabetes mortality and water magnesium levels (MapInfo; New York, USA).
Results
Age standardized death rates for diabetes in 2002 for the SD of Queensland ranged between 52 and 191 per 100 000 population (Fig1). Highest incidence occurred in the northern tropical SD while the lowest incidence occurred in the coastal divisions in the southern half of the state.
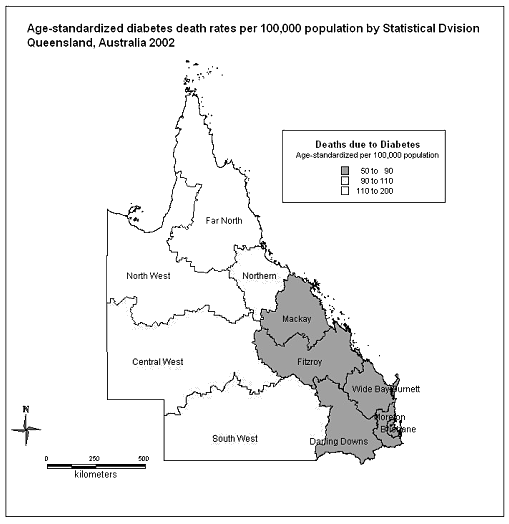
Figure 1: Age-standardized diabetes death rates per 100 000 population by statistical division, Queensland, Australia 2002.
Magnesium data from 2002 were available from 88 of the 157 municipal councils and Aboriginal councils contacted. Of the total population, 73% was represented by councils who had data on water levels of magnesium; 70.6% of the total Indigenous population was represented, although this was skewed toward coastal regions.
The amount of magnesium in the drinking water ranged from barely detectable at 0.01 mg/L to as high as 150 mg/L (Fig2). Outliers were validated by repeat inquiries to the original data sources and were retained in the analysis. The mean magnesium level was 13.0 mg/L and the median was 9.5 mg/L.
The areas with the lowest reported levels of magnesium in the water were those with the highest reported Indigenous population (Table 2).
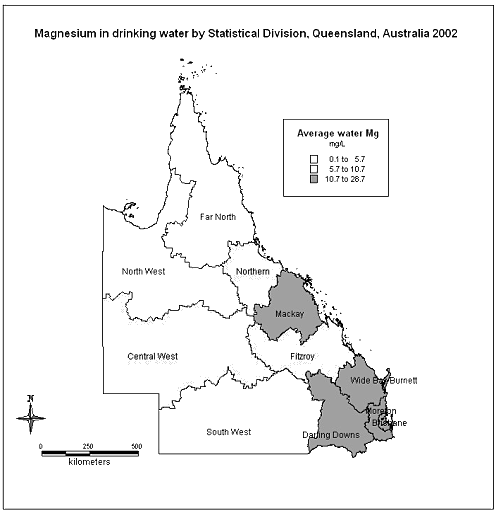
Figure 2: Magnesium in drinking water by statistical division, Queensland, Australia 2002.
Table 2: Population percentage identified as Indigenous in areas of low, medium, and high water magnesium (Mg)

Log transformation for water magnesium did not result in a normal distribution, so non-parametric methods were used for subsequent data analysis. The correlation between diabetes mortality rate and water magnesium levels was significant (r = -.422, p = 0.028). No significant correlation was found between rainfall and the magnesium content of drinking water.
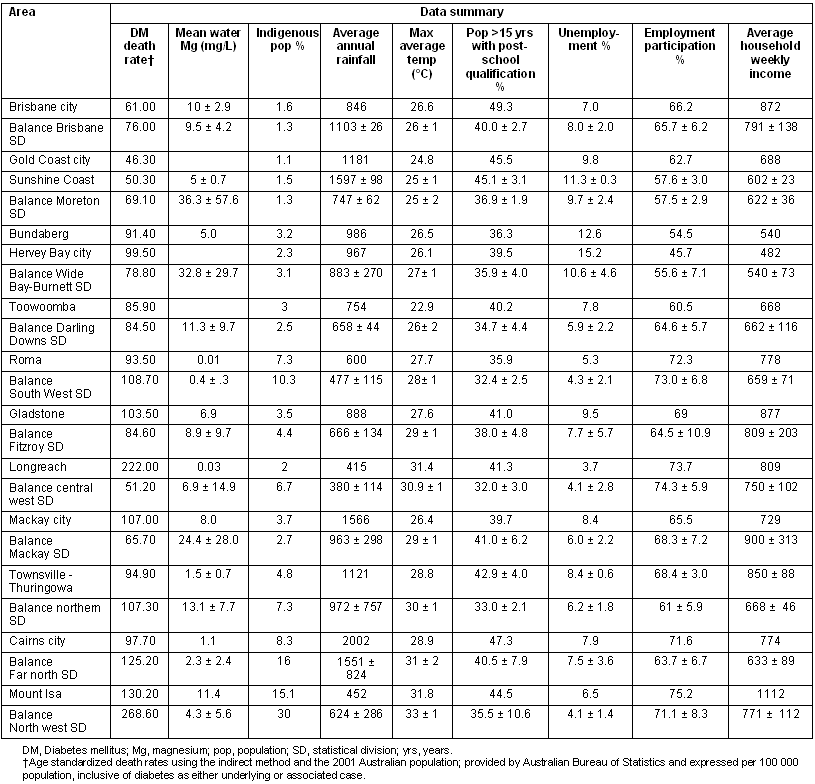
The diabetes mortality rate was positively correlated to the average maximum daily temperature (r = 0.579, p = 0.002), and to the percent of the population identified as Indigenous (r = 0.673, p < 0.001; Table 3). The percent of the population identified as Indigenous was also positively correlated to the average maximum temperature (r = 0.733, p < 0.001), and negatively correlated to unemployment (r = -.451, p = .027). A higher proportion of the population with post-school qualifications correlated with higher incomes (r = 424, p <.039). Comparing cities and townships to rural areas revealed only one significant difference; post-school qualifications were more common in the urban areas (t = 3.9, 95% confidence interval 3.03-9.89, p = .001). Using sum z-score variables for climate and socioeconomic factors did not reveal any additional correlations.
Table 3: Data summary by area
A total of 100 Indigenous participants completed the twelve-month WAT follow-up assessment. There were a total of 210 dietary recalls on file as 10 participants also completed a dietary recall at 6 months. A total of 167 records (79.5%) were included in analysis after culling for implausible intake. The participant ages ranged from 18 to 69 years old, with a mean age of 44.4 ± 1.3 standard error of the mean. There were 88 female and 12 male participants, with a mean body mass index (BMI) of 36.6 ± 0.65 SEM. Over 41.3% had hypertension, 34.7% had type 2 diabetes, and 68.7% had additional co-morbidities such as osteoarthritis or hyperlipidaemia. The distribution of months in which the recalls were collected was skewed towards February, the month the program began. The other 11 months of the year were evenly represented, allowing for adequate seasonal representation. The majority of recalls were performed on Tuesdays through to Fridays; weekend dietary intakes were not adequately addressed. Distribution throughout the month allowed for adequate representation between weeks with and without pension payments. The average daily magnesium intake was 248 mg (men: 267 mg ± 17; women: 245 mg ± 6 mg), significantly less than the intakes observed in the 1995 National Nutrition Survey (p< .001). These figures are 36% and 23% below the RDI for magnesium for males and females, respectively. Noting that the EAR is set to meet the needs of half the population, the reported intake was 24% and 8% below the EAR for males and females, respectively.
Discussion
In the present study, we found the 2002 incidence of diabetes-related mortality in Queensland, Australia, was positively correlated to the percentage of the population that was of Aboriginal and Torres Strait Islander descent. These results are consistent with ABS reports that Indigenous deaths in Australia due to diabetes (2001-2002) accounted for 7.6% of the total deaths, compared with 2.4% of total non-Indigenous deaths1. We also report a correlation between the incidence of diabetes-related mortality and the concentration of magnesium in drinking water. When considered in the context of lower magnesium intake that was observed in the regional Indigenous diet, the total daily magnesium intake may be low in the Australian Indigenous population.
Extensive epidemiological evidence exists, suggesting that a high magnesium diet may protect against the development of diabetes. The Women's Heart Study, with a cohort of 39 345 women and a 6 year follow up, demonstrated a significant inverse relationship between dietary magnesium intake and the risk of developing diabetes26. The risk reduction was 22% for those women with a BMI of 25 or higher. An analysis that combined the Nurses Health Study and the Health Professionals' Follow-up Study, including 85 060 women (18 years follow up) and 42 872 men (12 years follow up), demonstrated that after adjusting for confounding variables, a magnesium-rich diet reduced the relative risk of developing diabetes by 34% in women and 33% in men5. Similarly, an analysis of the Iowa Women's Health Study and the Honolulu Heart Program also showed inverse correlations between magnesium intake and diabetes risk27,28. In contrast, the Atherosclerosis Risk in Communities (ARIC) Study reported that while low serum magnesium levels were associated with an increased risk of diabetes, there was no correlation between dietary magnesium and diabetes risk. Researchers attributed this contradiction to the difference in the population studied compared with the other studies29. Clearly, most studies confirm that a high magnesium diet has a beneficial effect on reducing the risk of developing diabetes. Both Australian and US dietary surveys identified dietary intake as low in magnesium14,16, and this study identified an Australian Indigenous cohort with intakes significantly lower than the average Australian diet. Magnesium deficiency has been shown to induce insulin resistance, even in normal subjects4, and may be the underlying common mechanism by which insulin resistance links hypertension, lipid disorders, impaired glucose tolerance, type 2 diabetes, and metabolic syndrome3,12, all of which are prevalent among Australian Indigenous people.
The dietary component of this study is limited by the that fact we used a convenience sample which over-represented people with diabetes (35%) and hypertension (41%), was predominantly female, and is therefore not representative of all Indigenous people. Nonetheless, the sample was taken from subjects who were actively attempting to improve their dietary intake. The fact that the magnesium intake in the diet was inadequate in this cohort is highly suggestive that there is an even more profound nutrient deficiency in the broader population. Although a standardized technique was used for the triple-pass dietary recall, under-reporting is typical of all diet assessment methods outside a highly-controlled research setting.
This study confirmed prior studies that have correlated the level of magnesium in drinking water to diabetes mortality30,31. This does not imply causality, because ecological bias cannot be ruled out, and the association observed at area level may be different from that at the individual level. For example, the location of an individual's water consumption may not be reflected in the usual residence indicated on the death certificate. Furthermore, the water provided to one municipal area may be a blend from several sources with varying amounts of magnesium, and with seasonal variations (especially during times of drought). Finally, not all people living in an area receive their water from a municipal water system. While most Indigenous people live in cities and towns, and thus have access to municipal water, approximately one-quarter of the Indigenous population (108 085 of 458 520 in 2001) live in discrete Indigenous communities. Of these, 89 861 (20%) live in communities not connected to a municipal water supply32.
However, the fact that this study found a correlation between the incidence of diabetes-related mortality and the concentration of magnesium in drinking water suggests that further studies are warranted. When considered in the context of the lower magnesium dietary intake observed in a regional Indigenous cohort, total daily magnesium intake may be low. This deficit might be exacerbated in an environment where magnesium sweat loss is increased. This supposition is supported by a highly significant correlation between average daily high temperature and incidence of diabetes-related mortality observed in the present study. The correlations observed with lower post-school qualification among indigenous people, with subsequent association of lower socioeconomic status, may also contribute in that these individuals might be less likely to have access to heat relief such as air-conditioning, either at home or in the workplace, and that their dietary choices may be limited by income.
The evidence presented here are like pieces of a puzzle that build a case for magnesium as a potential contributor to diabetes in Australia, especially among Aboriginal and Torres Strait Islander peoples. It is plausible that a combination of lower magnesium intake in diet, increased loss of magnesium through sweat, and an inability to adequately replace lost magnesium through drinking water, may create a unique situation predisposing an individual to hypomagnesaemia and diabetes. Indeed, Australia has the unenviable reputation of having the highest diabetes-related mortality in the world among its Indigenous population (Fig. 3).
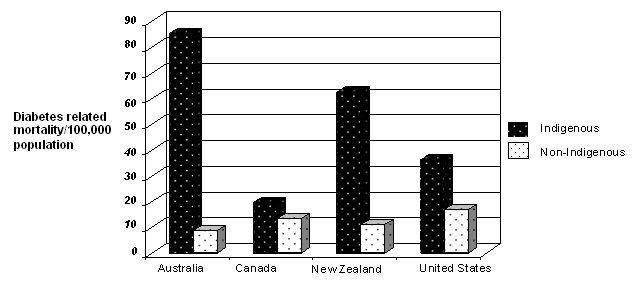
Figure 3: Diabetes related mortality in Indigenous and non-Indigenous populations in Australia, Canada, New Zealand and the USA, adapted from2.
Whether such hypomagnesaemia exists in this Indigenous population has not been determined. However, an association between hypomagnesaemia and diabetes has been well described in other populations7-10,33. Despite the growing body of evidence supporting the involvement of magnesium in diabetes, the need to consider magnesium status has not been integrated into primary health care for diabetes. This article presents the case that there is an urgent need to further investigate the potential relationship between magnesium and diabetes in the Australian Indigenous population, given that a deficiency might be readily addressed.
Acknowledgements
The authors gratefully acknowledge Dr David King, Director of the Centre for Tropical Urban and Regional Planning, and Ms Janis Puce, MA, for assistance with MapInfo, and Domonique Pinata for assistance in water data collection. The dietary component of this paper was derived from the Walkabout Together Program, funded by the Royal Australian College of General Practice.
References
1. Queensland Health. Health Indicators for Queensland: Northern Zone 2001. Brisbane, Qld: Public Health Services, Queensland Health, 2001.
2. Bramley D, Hebert P, Jackson R, Chassin M. Indigenous disparities in disease-specific mortality, a cross-country comparison: New Zealand, Australia, Canada, and the United States. New Zealand Medical Journal 2004; 117(1207): U1215.
3. Barbagallo M, Dominguez LJ, Galioto A, Ferlisi A, Cani C, Malfa L et al. Role of magnesium in insulin action, diabetes and cardio-metabolic syndrome X. Molecular Aspects of Medicine 2003; 24(1-3): 39-52.
4. Nadler JL, Buchanan T, Natarajan R, Antonipillai I, Bergman R, Rude R. Magnesium deficiency produces insulin resistance and increased thromboxane synthesis. Hypertension 1993; 21(6-2): 1024-1029.
5. Lopez-Ridaura R, Willett WC, Rimm EB, Liu S, Stampfer MJ, Manson JE et al. Magnesium intake and risk of type 2 diabetes in men and women. Diabetes Care 2004; 27: 134-140.
6. Song Y, Manson JE, Buring JE, Liu S. Dietary magnesium intake in relation to plasma insulin levels and risk of type 2 diabetes in women. Diabetes Care 2004; 27: 59-65.
7. Walti MK, Zimmermann MB, Spinas GA, Hurrell RF. Low plasma magnesium in type 2 diabetes. Swiss Medical Weekly 2003; 133(19-20): 289-292.
8. Saggese G, Federico G, Bertelloni S, Baroncelli G, Calisti L. Hypomagnesemia and the parathyroid hormone-vitamin D endocrine system in children with insulin-dependent diabetes mellitus: effects of magnesium administration. Journal of Pediatrics 1991; 118: 220-225.
9. Elamin A, Tuvemo T. Magnesium and insulin-dependent diabetes mellitus. Diabetes Research and Clinical Practice 1990; 10: 203-209.
10. Nadler J, Rude R. Disorders of magnesium metabolism. Metabolic Clinics of North America 1995; 24: 623-641.
11. Rodriguez-Moran M, Guerrero-Romero F. Low serum magnesium levels and foot ulcers in subjects with type 2 diabetes. Archives of Medical Research 2001; 32: 300-303.
12. Guerrero-Romero F, Rodriguez-Moran M. Hypomagnesemia is linked to low serum HDL-cholesterol irrespective of serum glucose values. Journal of Diabetes and its Complications 2000; 14: 272-276.
13. Franz KB, Bailey SM. Geographical variations in heart deaths and diabetes: effect of climate and a possible relationship to magnesium. Journal of the American College of Nutritionists 2004; 23: 521S-524S.
14. National Academy. Dietary reference intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. Washington, DC: National Academy Press, 1997.
15. Australian Government. Nutrient reference values for Australia and New Zealand including recommended dietary intakes. Canberra, ACT: Commonwealth of Australia, 2006; 193-196.
16. McLennan W, Podger A. National nutrition survey: nutrient intakes and physical measurements. Report no 4805.0. Canberra, ACT: Australian Bureau of Statistics; 1998.
17. Azoulay A, Garzon P, Eisenberg MJ. Comparison of the mineral content of tap water and bottled waters. Journal of General Internal Medicine 2001; 16: 168-175.
18. Seelig MS. Epidemiology of water magnesium: evidence of contributions to health. (Online) 2002. Available from: http://www.mgwater.com/epidem.shtml (Accessed 16 July 2004).
19. Zhao HX, Mold MD, Stenhouse EA, Bird SC, Wright DE, Demaine AG et al. Drinking water composition and childhood-onset type 1 diabetes in Devon and Cornwall, England. Diabetes Medicine 2001; 18: 709-717.
20. Nerbrand C, Agreus L, Lenner RA, Nyberg P, Svardsudd K. The influence of calcium and magnesium in drinking water and diet on cardiovascular risk factors in individuals living in hard and soft water areas with differences in cardiovascular mortality. BMC Public Health 2003; 3: 21.
21. Ferrandiz J, Abellan JJ, Gomez-Rubio V, Lopez-Quilez A, Sanmartin P, Abellan C et al. Spatial analysis of the relationship between mortality from cardiovascular and cerebrovascular disease and drinking water hardness. Environmental Health Perspectives 2004; 112: 1037-1044.
22. Sawka MN, Montain SJ. Fluid and electrolyte supplementation for exercise heat stress. American Journal of Clinical Nutrition 2000; 72(2 Suppl): 564S-572S.
23. National Health and Medical Research Council. Nutrition in Aboriginal and Torres Strait Islander peoples: an information paper. Canberra: NHMRC, 2000; 24
24. National Health and Medical Research Council, and National Resource Management Ministerial Council. National Water Quality Management Strategy Australian Drinking Water Guidelines. Canberra, ACT: NHMRC and National Resource Management Ministerial Council, 2004.
25. Heath D, Longstreet D, Malouf P, Panaretto K. Walkabout together: a lifestyle intervention program developed for Townsville's overweight Indigenous people. In Touch, Public Health Association of Australia 2006; 23(1): 18-19. [Newsletter].
26. Song Y, Manson JE, Buring JE, Liu S. A prospective study of red meat consumption and type 2 diabetes in middle-aged and elderly women: the women's health study. Diabetes Care 2004; 27: 2108-2115.
27. Abbott RD, Ando F, Masaki KH, Tung KH, Rodriguez BL, Petrovitch H et al. Dietary magnesium intake and the future risk of coronary heart disease (the Honolulu Heart Program). American Journal of Cardiology 2003; 92: 665-669.
28. Meyer KA, Kushi LH, Jacobs DR Jr, Slavin J, Sellers TA, Folsom AR. Carbohydrates, dietary fiber, and incident type 2 diabetes in older women. American Journal of Clinical Nutrition 2000; 71: 921-930.
29. Kao WH, Folsom AR, Nieto FJ, Mo JP, Watson RL, Brancati FL. Serum and dietary magnesium and the risk for type 2 diabetes mellitus: the Atherosclerosis Risk in Communities Study. Archives of Internal Medicine 1999; 159: 2151-2159.
30. Yang CY, Chiu HF, Cheng MF, Tsai SS, Hung CF, Tseng YT. Magnesium in drinking water and the risk of death from diabetes mellitus. Magnesium Research 1999; 12: 131-137.
31. Mahaba H. Prevalence of diabetes mellitus and hypertension in relation to chemical composition of drinking water - does magnesium protect against diabetes mellitus? Saudi Medical Journal 1998; 19: 469-474.
32. Australian Institute of Health and Welfare. Australia's Health 2004: 9th Biennial Health Report of the Australian Institute of Health and Welfare. Report no. #Aus44. Canberra, ACT: AIHW, 2004.
33. Walti MK, Zimmermann MB, Spinas GA, Jacob S, Hurrell RF. Dietary magnesium intake in type 2 diabetes. European Journal of Clinical Nutrition 2002; 56: 409-414.



