Introduction
Despite the potential lifestyle benefits of living in rural areas, rural inhabitants in the USA are at a distinct disadvantage for many health indicators. Motor vehicle accidents result in death and serious injury at rates disproportionate to their occurrence compared with urban settings (60% vs 48%) with two-thirds of deaths occurring on rural roads compared with only one-third of accidents occurring there1. Rural non-motor vehicle injuries leading to death occur at nearly twice the urban rate, despite the common perception of increased gun violence in urban settings; the opposite is true, albeit this is less gang related2,3.
Rural residents are also at higher risk for major diseases, considering both burden of disease and the severity of their complications4. Hypertension affects 27% more US rural compared with urban residents (128.8 vs 101.3 per 1000 individuals) with the attendant increased relative risk of stroke and cerebrovascular disease at 1.45 higher compared with urban residents1. For myocardial infarction, there is a decrease in compliance with recommended therapies in rural areas5 and an increased overall mortality independent of confounders6. (There is controversy about the latter statement, however, with evidence suggesting selection bias and poor internal controls to eliminate double counting7,8). Finally, retrospective chart review of rural patients in Alabama and Montana showed worse control of diabetes and its associated cardiovascular endpoints compared with non-rural patients and national averages9,10, but this was not reproduced among American Indian populations in New Mexico11.
There are several established confounders for these findings. Rural residents in the USA tend to be older, of lower socioeconomic status (SES), and without access to the services found in urban areas. For example, according to the National Rural Health Association, 18% of rural residents are aged 65 years or older, compared with 15% of non-rural residents. More are covered by Medicare (the government-provided health insurance for individuals aged 65 and older) and fewer covered by private insurance. The per capita income is US$7,417 lower in rural areas, with more rural residents living below the federal poverty level and a nearly 24% of rural children living in poverty. There are almost 2.4 times the number of Health Professional Shortage Areas in rural areas, and rural residents are 4 times as likely to lack mental health care. All of this leads to a generally sicker population, as evidenced by self-reports of health status12 and overall death rates for both men and women being 33% higher3.
Other factors that may contribute to the rural-urban disparity include a lack of specialist availability, the distance of residents from more specialized centers and even from their regular sources of care, and a lower volume of certain diagnoses or procedures for hospitals and clinicians, thereby potentially increasing the complication rates for those patients, although this latter has been debated13-15.
In the common presentation of patients with both diabetes and hypertension, however, the burden of disease in both rural and urban settings is high, the consequences of poor control are multiple and severe, and control is frequently suboptimal in almost every setting16,17. In re-examining the data of a study by the present authors that examined point-of-care provider blood pressure (BP) decision-making in patients with diabetes18, the authors wished to specifically examine rural-urban differences. The intention was to establish: (i) whether rural residents with diabetes who presented for care were more likely to have poorly controlled hypertension; (ii) for those with elevated BP, whether there is a difference in the rate of change in therapy by their healthcare provider; (iii) the frequency of providers listing 'BP at or near goal' as the reason for inaction in those patients; and (iv) what patient and provider factors predicted action taken to control BP in patients with elevated BPs. As a secondary analysis, the care provided in a further subset of the underserved was evaluated. In the USA, community health centers (CHCs) are safety-net providers of care to the uninsured and underserved within their communities. In this analysis rates of change in therapy for patients with elevated BP for patients cared for at CHCs were compared with patients at non-CHCs.
Two Colorado practice-based research networks participated in the study: the High Plains Research Network (consisting of 27 rural and frontier primary care practices throughout Northeastern Colorado) and the Colorado Research Network (32 mostly urban and underserved primary care practices). A total of 26 practices participated in those two settings: 13 in rural areas and 13 urban. Of the rural sites, 4 were CHCs and 9 were not. Of urban sites, 5 were CHCs and 8 were not.
Ethics approval
The initial study received approval from the Colorado Multiple Institutional Review Board and other ethics committees overseeing participating sites. Because this is a secondary analysis of that dataset, further ethics approval was not required.
Methods
A brief questionnaire was completed by the provider after each encounter with a non-pregnant adult with a diagnosis of type 2 diabetes. Surveys were completed over a 2-4 week period between June 2003 and May 2004. Survey information included: (i) demographic and other data on the provider and patient; (ii) the BP reading at the visit; (iii) action taken on the BP; (iv) type of action if action was taken (medications and/or lifestyle changes); and (v) reasons given by the provider for inaction if no action was taken (multiple reasons could be selected). If more than one BP reading was taken or used in the decision-making process, the provider was asked to circle the BP result that led to the decision. All surveys were de-identified for analysis. The survey was developed by the modified Delphi technique and piloted for clarity and time of administration in selected practices within the network. 'Rural' was defined as a community of fewer than 25 000 residents at least 32 km (20 miles) from a major metropolitan area.
Statistical analysis
Bivariate Analysis: Bivariate analyses were used to describe the patient population in terms of diabetic patients seeking care from rural practices and those seeking care from urban practices. Patient demographic characteristics and socioeconomic status were considered in the analyses. Insurance status was used as a surrogate for income, combining Medicaid and discounted ('low income') versus all other payer sources ('all other income'). Medicaid refers to the government sponsored health insurance for adults under 60% of the federal poverty levels (or a monthly income of $542) and dependent children, those who are disabled, or those aged 65 or older with a monthly income of $699 or less. 'Discounted' refers to a State of Colorado program for individuals with and income less than 200% of the federal poverty level. To investigate medication costs, Medicare (which lacked medication coverage at the time of the study) only was compared with all other types of insurance combined (including patients with both Medicare and additional insurance that covered medications). Further, the total number of prescription medications (≤6 and >6) and number of hypertensive medications specifically were examined. Data from all patients were used in this analysis.
Patients were initially categorized into two BP groups. Elevated BP was defined as a systolic BP of at least 130 mmHg and/or diastolic BP of at least 80 mmHg, the target established by the American Diabetes Association19 (n = 478). The 'at goal' group consisted of all other patients (n = 298). Blood pressure was also stratified separately for systolic and diastolic levels, as well as a combined variable. In the combined categories, BP was classified as 'at goal' (systolic ≤130 mmHg and diastolic ≤80 mmHg) (n = 298), 'above previous goal' (systolic ≥140 mmHg or diastolic ≥90 mmHg, the goal for diabetes before the year 2000), or 'near goal' (not 'at goal' but systolic ≤140 mmHg and diastolic ≤90 mmHg).
Among the patients with elevated BP, bivariate analyses determined associations between clinician reasoning regarding why action was or was not taken to control patients' BP and practice setting (rural vs urban practices). For these analyses, patients with elevated BP where action was taken at the visit (n = 167) and patients with elevated BP where action was not taken at the visit (n = 311) were used, respectively. Since clinicians could select more than one reason for taking or not taking action during the visit, separate analyses were performed according to reason.
For all categorical patient and action/inaction measures, the χ2 test was used to determine associations; whereas, for continuous patient characteristics, the two-sample t-test was used. For categorical measures where χ2 was violated, associations were determined by Fishers' exact test. Results concerning these variables should be considered preliminary. Percentages, totals and p-values are reported. Because multiple tests were made, statistical significance is determined at the alpha level of 0.01.
Multivariate Analysis: Using patients with elevated BP, multivariate logistic regression analyses determined adjusted associations on action taken to control patients' BP during the visit (yes/no) and practice setting (rural vs urban). Initially multivariate models that accounted for clustering of patients within practices were explored. However, because the interclass correlation coefficient (ICC) of the clustering was low (ICC = 0.03; p = 0.2015), classic multivariate logistic regression analyses were used. The model adjusted for general demographic measures (age, sex, race and ethnicity), BP ('near goal' or 'uncontrolled'), practice level measures (practice setting, CHC) and covariates that were significantly associated at the alpha level of 0.20 with the outcome, 'action taken', from the bivariate analysis (communication problems, income level, number of prescription medications). All analyses were performed using SAS v 9.1(SAS Institute Inc; Cary, NC, USA).
Results
Twenty-six primary care practices participated with 13 sites in rural settings and 13 in urban settings. The rural clinics completed 297 surveys and urban sites completed 481. The patients were primarily females who sought care at a CHC, with rural and urban patients being approximately the same age (56.1 and 58.3 years, respectively). Rural patients were more likely to be white non-Hispanic, with Medicare, taking 8 or more prescription medications and less likely to be on Medicaid, which is, as above, a government sponsored insurance for very low income residents with significant disabilities (p<0.01). Results also indicated that rural patients on average had lower diastolic BP (p<0.001) and slightly lower systolic BP compared with patients with diabetes from urban practices (p = 0.046) (Table 1; Fig1).
In the univariate analysis, action rates were higher among urban patients, compared with rural patients. Among patients with elevated BP, 27.5% of rural patients had some action taken by their physician and 39.1% of urban patients had action taken (p = 0.0108) (Table 2). However, in a multivariate logistic regression model, after adjusting for patient and practice characteristics, practice setting was not a significant predictor of action taken to lower BP among diabetic patients with elevated BP; that is, neither rural setting (OR 0.75 [0.45-1.25], p 0.27) nor receiving care at a CHC versus another source of care in the community (OR 0.75 [0.45-1.23] p 0.26) were associated with higher or lower action rates. Having 6 or more prescription medications (p = 0.004) and the patient's BP being 'above goal' (p <0.0001) were significant predictors of clinicians taking action to control high BP among diabetic patients with elevated BP (Table 3; Fig2).
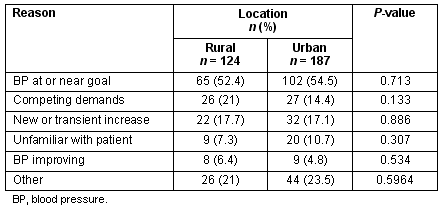
The type of action and reasons for inaction for those patients with elevated BP were also not significantly different according to practice location (Tables 4, 5).
Table 1: Patient and practice characteristics
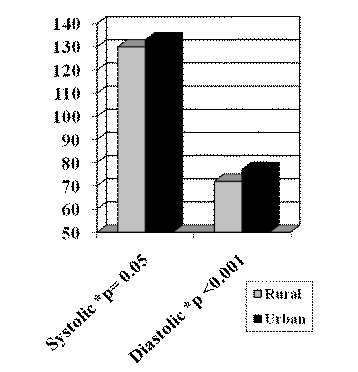
Figure 1: Mean blood pressure.
Table 2: Action taken among patients with elevated blood pressure (unadjusted)
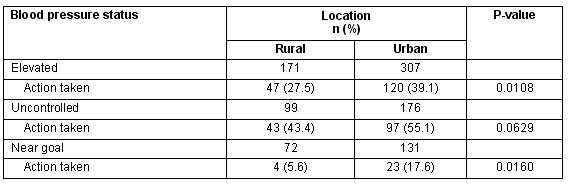
Table 3: Patients with elevated blood pressure. Multivariate logistic regression: action taken at visit
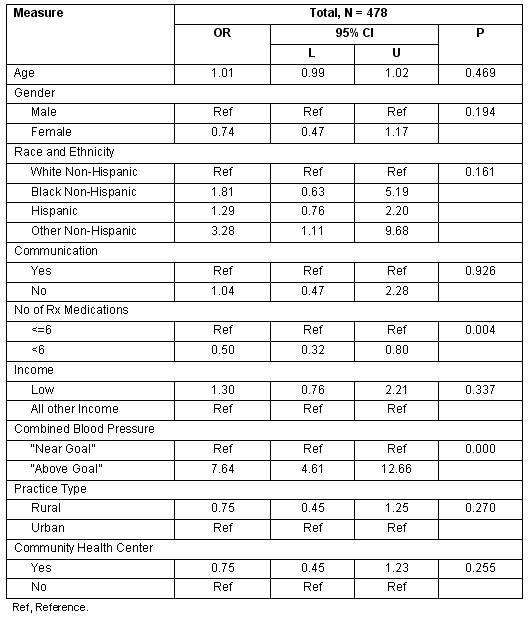
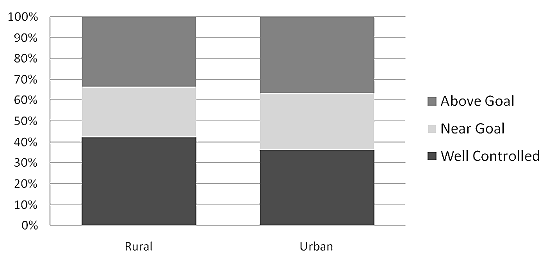
Figure 2: Blood pressure control by variation from goal.
Table 4: Types of action taken among patients with elevated blood pressure

Table 5: Reasons for inaction among patients with elevated blood pressure
Discussion
Given the national data that shows higher rates of hypertension, increased workload for rural clinicians, and decreased access of care for rural residents, it is not hard to assume that the care provided to rural patients might be of lower quality or in some other way inferior to that in urban settings. Similarly, with the workloads and barriers to care in CHCs, one might assume the same. This study, however, suggests the opposite. Rural rather than urban residents had lower average BPs (a small yet statistically significant difference that is unlikely to be clinically important). Patients of CHCs had similar BP control compared with non-CHC patients.
When adjusted for confounders, action rates for patients with elevated BP were not different between rural or urban settings, nor in CHCs. What predicts action is the degree of variance from goal BP and the number of medications used by the patient. It is also important to note that the reasons for inaction were remarkably similar among rural and urban providers. If one group of providers were not aware of current recommendations, one might expect to see them list 'BP at or near goal' more frequently, and that did not occur. What was notable was the listing of 'competing demands' 50% more frequently as the reason for inaction among rural providers. This was not significant but may reflect the higher workload typically seen in rural settings20,21.
All of this suggests that patient characteristics, not demographics nor rurality nor low SES nor other external factors, seem to be what providers base their treatment decisions on. In this study, in Central and Northeastern Colorado providers seemed to care for the patients in front of them and treat them on objective criteria, regardless of setting.
Limitations
While this study offers strengths both in content and study design, it has several limitations. In any study, and possibly more likely in point-of-care studies, there is potential for the Hawthorne effect. This seems less operative in this study for several reasons. First, participating providers were instructed that this study was to determine how decisions were made and not how closely guidelines were followed. Second, the data were de-indentified and could not be tracked back to the provider. Third, practices in these two research networks participate regularly in similar studies, making the novelty of being in a study, and the attendant change in behavior potentially somewhat less. Fourth, this study shows similar rates of action compared with other studies with different designs22,23.
A second limitation is bidirectional - does measuring provider behavior once reflect usual practice and, similarly, does measuring BP once truly measure a patient's usual BP? In answer, this is a limitation of every cross-sectional study. One data point does not equal a trend in either practice patterns or BP control. What is studied though is how providers make decisions on the BP value presented to them. It is likely that they considered the patient's past BP readings and, in fact, 'BP improving' was one of the reasons listed for inaction that a provider might choose. Regarding identifying the usual practice patterns of a provider, we also must consider the benefit of large numbers. If the Hawthorne effect is minimized, provider behavior is proximate to baseline if one studies that same action over and over. Behaviors, as do statistics, regress towards the mean or usual behavior. A further potential limitation is that the practices included are not representative of usual practices in the USA. The urban practice-based research network is heavily weighted towards the underserved. Similarly, the number of surveys from rural settings were predominantly from CHCs. However, CHCs were neither positively or negatively associated with action rates, suggesting that the quality of care among safety-net providers is similar to that of non-safety-net providers. As medicine becomes more urbanized, that is, with an increasing proportion of physicians choosing to practice in urban settings, rural areas will more strongly depend on the CHC in their area. Finally, it is notable that urban practices contributed substantially more surveys than did rural ones. This is likely to be secondary to smaller practice size overall in rural settings. While the patient mix was not controlled for in each setting and the data were de-identified, there is no evidence that urban providers see more patients with diabetes proportionally. This theory is supported by the recent work of Weeks and Wallace who show similar patient panels among primary care physicians in rural and urban settings21.
Conclusion
When comparing rural and urban clinicians, there was no evidence of a difference in the quality of care in the management of elevated BP among their patients with diabetes. This was also so for CHCs. However, elevated BP is common in both settings and diligence is required among providers, regardless of urban or rural setting, in lowering BP among patients with diabetes.
References
1. Gamm LD, Hutchison LL, Dabney BJ , Dorsey AM (Eds). Rural Healthy People 2010: a companion document to Healthy People 2010. College Station, TX: The Texas A&M University System Health Science Center, School of Rural Public Health, Southwest Rural Health Research Center, 2003.
2. Cherry DC, Huggins B, Gilmore K. Children's health in the rural environment. Pediatric Clinics of North America 2007; 54(1): 121-33; ix.
3. National Rural Health Association. 'What's different about rural health care?' 2007-2009. Available: http://www.ruralhealthweb.org/go/left/about-rural-health (Accessed 3 February 2009).
4. Weeks WB, Wallace AE, Wang S, Lee A, Kazis LE. Rural-urban disparities in health-related quality of life within disease categories of Veterans. Journal of Rural Health 2006; 22(3): 204-211.
5. Sheikh K, Bullock C. Urban-rural differences in the quality of care for Medicare patients with acute myocardial infarction. Archives of Internal Medicine 2001; 161(5): 737-743.
6. Baldwin LM, MacLehose RF, Hart LG, Beaver SK, Every N, Chan L. Quality of care for acute myocardial infarction in rural and urban US hospitals. Journal of Rural Health 2004;20(2): 99-108.
7. Westfall JM, McGloin J. Impact of double counting and transfer bias on estimated rates and outcomes of acute myocardial infarction. Medical Care 2001; 39(5): 459-468.
8. Westfall JM, Kiefe CI, Weissman NW, Goudie A, Centor RM, Williams OD et al. Does interhospital transfer improve outcome of acute myocardial infarction? A propensity score analysis from the Cardiovascular Cooperative Project. BMC Cardiovascular Disorders 2008; 8: 22.
9. Andrus MR, Kelley KW, Murphey LM, Herndon KC. A comparison of diabetes care in rural and urban medical clinics in Alabama. Journal of Community Health 2004; 29(1): 29-44.
10. Coon P, Zulkowski K. Adherence to American Diabetes Association standards of care by rural health care providers. Diabetes Care 2002; 25(12): 2224-2229.
11. Moore K, Roubideaux Y, Noonan C, Goldberg J, Shields R, Acton K. Measuring the quality of diabetes care in urban and rural Indian health programs. Ethnicity and Disease 2006; 16(4): 772-777.
12. Weeks WB, Kazis LE, Shen Y, Cong Z, Ren XS, Miller D et al. Differences in health-related quality of life in rural and urban veterans. American Journal of Public Health 2004; 94(10): 1762-1767.
13. Christian CK, Gustafson ML, Betensky RA, Daley J, Zinner MJ. The Leapfrog volume criteria may fall short in identifying high-quality surgical centers. Annals of Surgery 2003;238(4): 447-455.
14. Saposnik G, Baibergenova A, O'Donnell M, Hill MD, Kapral MK, Hachinski V. Hospital volume and stroke outcome: does it matter? Neurology 2007; 69(11): 1142-1151.
15. Dimick JB, Welch HG, Birkmeyer JD. Surgical mortality as an indicator of hospital quality: the problem with small sample size. JAMA 2004; 292(7): 847-851.
16. Frijling BD, Spies TH, Lobo CM, Hulscher ME, van Drenth BB, Braspenning JC et al. Blood pressure control in treated hypertensive patients: clinical performance of general practitioners. British Journal of General Practice 2001; 51: 9-14.
17. Berlowitz DR, Ash AS, Hickey EC, Friedman RH, Glickman M, Kader B et al. Inadequate management of blood pressure in a hypertensive population. New England Journal of Medicine 1998; 339: 1957-1963.
18. Hicks PC, Westfall JM, Van Vorst RF, Bublitz Emsermann C, Dickinson LM, Pace W et al. Action or inaction? Decision making in patients with diabetes and elevated blood pressure in primary care. Diabetes Care 2006; 29(12): 2580-2585.
19. Anon. Executive summary: standards of medical care in diabetes-2009 Diabetes Care 2009; 32(suppl1): S6-S12.
20. Baldwin LM, Rosenblatt RA, Schneeweiss R, Lishner DM, Hart LG. Rural and urban physicians: does the content of their Medicare practices differ? Journal of Rural Health 1999; 15(2): 240-251.
21. Weeks WB, Wallace AE. Rural-urban differences in primary care physician's practice patterns, characteristics, and incomes. Journal of Rural Health 2008; 24(2): 161-170.
22. Oliveria SA, Lapuerto P, McCarthy BD, L'Italien GJ, Berlowitz DR, Asch SM: Physician-related barriers to the effective management of uncontrolled hypertension. Archives of Internal Medicine 2002; 162: 413-420.
23. Cotton A, Aspy C, Mold J, Stein H. Clinical decision-making in blood pressure management of patients with diabetes mellitus: an Oklahoma Physicians Resource/ Research Network (OKPRN) study. Journal of the American Board Family Medicine 2006; 19: 232-239.
