Introduction
Australians living in rural communities have higher overall death rates and lower life expectancy than their urban counterparts1. Mortality data for various cancers show disadvantage to rural people1-4. Malignancies are likely to be diagnosed at a more advanced stage, or not diagnosed prior to death in rural populations3-5. Reasons for poor outcomes for rural oncology patients have been explored, including reduced levels of cancer screening and different management in regional hospitals2,5-7. However health attitudes and behaviours of rural people may also impact on these worrying statistics. Rural people have been shown to have different health beliefs and values compared with urban populations with emphasis placed on role fulfilment8,9 rather than comfort or cosmetic fulfilment for some conditions. There is also a perception that rural people are more stoic and fatalistic than urban people, although this is difficult to formally assess10.
Other studies have investigated health attitudes in cancer patients. One study involving newly diagnosed cancer patients compared depression, perception of seriousness of illness and health locus of control beliefs11. Patients with strong internal belief (ie, believe that their health is under their personal control) had a weaker relationship between perceived severity of disease and depression. A study into health locus of control and cancer screening attitudes revealed that women with fewer years of eduction had higher scores for belief in chance and belief in powerful others12. These patients did not value early diagnosis of cancer and had low self-perceived cancer susceptibility.
'Hope' has been identified as a very important aspect for oncology patients. Hope has been shown to be related to effective coping strategies in oncology patients and has been the subject of several publications13,14. However little information is available in the current literature that explores the relationship between rurality and levels of hope.
This study is a forerunner to a planned larger project to explore the research question: do oncology patients from rural backgrounds differ in their health attitudes and hope levels compared with those from urban backgrounds? The aim of this study was to determine the utility of the selected validated instruments, newly diagnosed patients' willingness and ability to complete the composite survey instrument, and to identify likely issues for inclusion and/or greater focus in the larger project. As pilot studies are also used to develop or refine research questions and hypotheses, this article also considers some research questions for the planned large scale study.
Method
New patients referred to the Medical Oncology Department at The Townsville Cancer Centre were approached over a 5 month period to participate in the study. One of the authors (AH) attempted to personally inform patients about the study and invite their participation. When this was not possible, a member of the clinical staff undertook the recruitment process. Patients were excluded if they were not fluent in English, had significant cognitive impairment, or at the request of the treating oncologist. After consenting to the study, patients completed a questionnaire which included a series of previously validated scales and also collected demographic data.
In order to compare groups, a measure of quality of life and symptom issues was necessary. The EORTC QLQ-C30 is a well-established tool developed to evaluate quality of life in cancer patients. The current version has 30 items and explores functional domains, symptom issues and global health score via a 4-point Likert-type scale. The questionnaire and has good validity and reliability scores15. This questionnaire has undergone qualitative and quantitative assessment of patient and observers' responses with good agreement scores16.
Social support status is known to significantly influence patients' health attitudes and level of hope. The Duke-UNS Functional Social Support Questionnaire was used. This is a short instrument that measures quality and quantity of support17. In the present study, an additional question 'help when I need transportation' was included as this is often a major issue for rural patients. A 5-point semantic interval scale is used ranging from 'as much as I would like' to 'much less than I would like'. The scale has previously had reasonable re-test reliability with a correlation co-efficient of 0.6618.
Multi-dimensional health locus of control (MDHLC) evaluates health and sick role attitudes by three broad categories: internal, chance and powerful others19. Patients scoring highly on the internal section are more likely to engage in positive health role behaviours. However, health beliefs should only predict health related behaviour under high health value conditions. The scales use a 6 point Likert-type scale and have moderate reliability (Cronbach alpha score 0.60 - 0.70) and retest stability. Scores for multi-dimensional health locus of control range from 6 to 36 for each belief studied i.e. powerful others (individuals who the respondent believes can positively influence health, including spiritual, clinical, emotional others), chance (no individual can influence outcome) or internal (the individual can positively influence their health outcome). A score of 21 is neutral, with scores above this indicating increasing conviction of belief. This scale has been validated in the Australian population20.
The Herth Hope Index is a brief instrument developed from the longer Herth Hope Scale in 1992 and found to be as reliable21. The scale has 12 items using a Likert-type ordinal scale of 1 to 4. The index has been validated using acutely ill, chronically ill and terminally ill patients. The alpha co-efficient was 0.97 with a two-week re-test reliability of 0.91. Previous studies have found a significant relationship between levels of coping and hope scores in oncology patients14,22.
Information was also obtained regarding the patients' address, place of birth and whether they considered themselves a rural person. Each patient's current town of residence was used to assign a 'rurality' score using the Rural, Remote and Metropolitan Areas classification (RRMA)23. The intent of the current treatment - either curative or palliative - was sought from patients' charts and was assumed to have been conveyed to patients by their consultant clinician.
Data were collated and analysed using SPSS (vers. 12; SPSS Inc; Chicago, IL, USA). Uni-variate analyses were conducted because small numbers precluded multi-variate analysis. Non-parametric Mann-Whitney U and Kruskal-Wallis tests were used where data were skewed, or categorical. Monte-Carlo estimations of p-values were generated, because exact p-values were not able to be generated. The study was approved by The Townsville Hospital Ethics Committee, protocol number 22/04.
Results
Forty-seven of 66 eligible patients completed the questionnaire (71% participation rate). Patients did not appear to have difficulty completing the questionnaire because only four failed to answer all questions. Demographic information is set out in Table 1. There was little difference between participants and non-participants with respect to any of the variables, except that the female participation rate was higher. That said, females accounted for 27% of eligible patients.
Table 1: Demographics and cancer diagnosis of study participants and non-participants
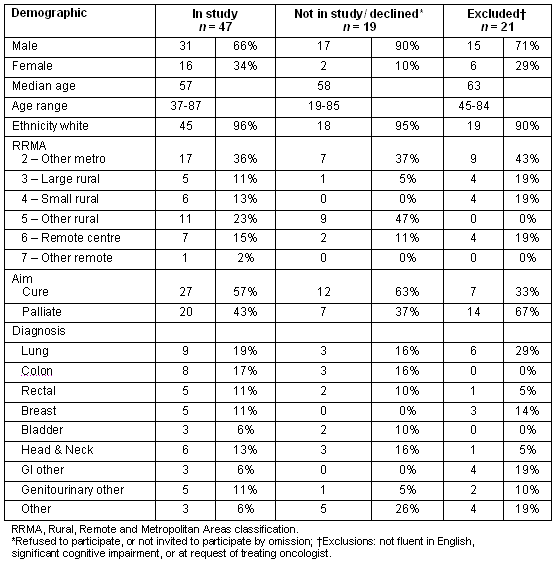
There were discrepancies between RRMA classification and patients' perceptions of rurality in eight cases. Four patients lived in a metropolitan area but identified themselves as 'rural'; three of these had retired to live in town after working in rural environments, while the fourth lived in a small community on the outskirts of the metropolitan area. These four patients were considered rural for purposes of analysis. Conversely, four patients identified themselves as 'not rural' despite scoring between 4 and 7 on the RRMA scale (ie, living in rural or remote areas). Reasons for this were less clear. These patients were considered urban in the analysis.
Most patients in this study felt well-supported with high ratings in the Duke Support Scale. There was not statistically significant difference by rurality and treatment intent (Kruskal-Wallis Test, 4 independent groups, p = 0. 53). Hope scores were generally high with a median Herth Hope Index score of 39 (Table 2). However, urban palliation patients demonstrated median scores (36.0) appreciably lower than urban curative (39.0) and rural curative (41.0) and rural palliation (40.5) participants, although this was not statistically significant (Kruskal-Wallis test, 4 independent groups, p = 0. 54).
Table 2: Median Scores for Herth Hope Index, Duke Social Support Scale and Multi-dimensional Health Locus of Control by Rural or Urban Background
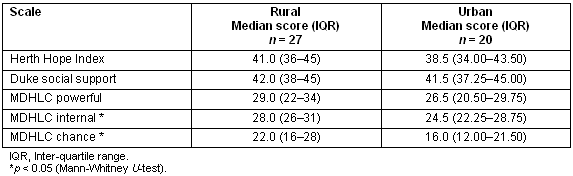
Results for multi-dimensional health locus of control showed internal belief was statistically significantly higher in rural patients, as a whole, compared with urban patients as a whole (Mann-Whitney U-test, p = 0.02), and approached significance when treatment intent was considered (Kruskal-Wallis Test, four independent groups, p = 0.07) (Table 3). Rural patients also had statistically significantly higher belief in chance than urban patients (Mann-Whitney U-test, p = 0.02), although this did not hold when treatment intent was included (Kruskal-Wallis test, four independent groups, p = 0.10). There was no statistically significant difference in belief in powerful others between rural and urban patients, either as a whole or when treatment intent was included.
Table 3: Median scores and Inter-quartile Ranges(Kruskal-Wallis test) for Herth Hope Index, Multi-dimensional Health Locus of Control, Duke Total Score and EORTC Global Health Status by Rurality and Treatment Intent
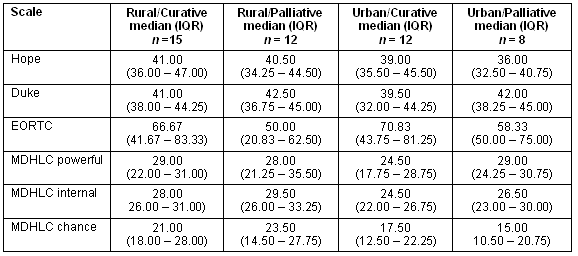
One interesting pattern to emerge was a gender difference with men having higher belief in chance than women in this study population (Mann-Whitney U-test, p = 0.02). There were no other statistically significant gender-related differences in attitudes or beliefs. The distribution of men and women across rurality/ treatment intent categories was not significantly different (χ2 = 3.02, df = 3, p = 0.41).
Discussion
Because approximately 50% of north Queensland's population lives outside major urban centres, the urban-rural mix in this study is likely representative. Of note was that approximately 17% of patients identified as rural or urban, counter to their current town of residence. A subsequent open-ended question in the questionnaire demonstrated that recent relocation from a rural to an urban setting accounted for half these cases. The discrepancies for the remainder were less clear. The results suggest the need to retain the open-ended explanatory question if reasonable accuracy in terms of likely rural 'identity' or 'connectedness' is to be obtained. The planned large scale study will enable deeper investigation of the connection between perceived 'rurality', or 'rural connectedness', and individuals' attitudes and beliefs. It may be, for example, that 'rural connectedness' (seeing oneself as rural, even though an urban resident and vice versa - 'rural disconnectedness') is just as important in understanding some people's attitudes and beliefs, as actual location in a rural setting. Thus, a research question for the larger study could be 'do people with "rural connectedness" (regardless of actual residence) display similar attitudes and beliefs as people from rural settings?' A similar question could be asked about rural residents who display 'rural disconnectedness' (ie, identify as urban).
The high levels of hope found in this study of oncology patients are generally in line with other work13,22,24,25. The findings of this study are also in keeping with previous literature in showing no relationship between hope and age, level of education, stage of disease or quality of life14,22,24-26. However, it is noteworthy that no other studies appear to have compared rural and urban people in terms of hope. That no statistically significant differences were found would suggest that, when faced with a potentially terminal disease, most people hold great hope. It is also interesting to note that people, from both rural and urban settings, receiving palliative treatment had lower scores than their respective counterparts receiving curative treatment (who held very similar levels of hope). The larger study may enable determination of the significance of this pattern.
The statistically significantly higher belief in chance for rural patients compared with urban patients, and regardless of treatment intent, is consistent with perceptions that rural people are more fatalistic, perhaps because of the impact of their environment8,10 The association between rural stoicism and delayed health-seeking has been demonstrated previously and attributed to the general patterns of later diagnosis, more advanced disease and poorer prognoses for people from rural areas3-6,8,20. Previous work in this area has also demonstrated higher belief in chance in lower socio-economic groups11,27,31, older people11,27 and lower level of education27; groups who characterise much of rural Australia1.
The statistically significantly higher internal belief in rural patients compared with urban patients is in line with the oft-noted 'independence' of rural people10. Although not statistically significant in this study, a pattern worthy of further investigation in the larger study is that participants receiving palliative treatment in rural and urban settings had higher internal scores than their respective counterparts receiving curative treatment.
There was no statistically significant difference in belief in powerful others between rural and urban patients. Studies of patients with chronic disease (eg diabetes, hypertension) have shown higher belief in powerful others than healthy populations28. Previous studies have also shown rural residents to have low levels of positive health-related behaviours (eg cancer screening, which could be argued reflect belief in the influence that others can have on health)2,5. The lack of implementation of these (powerful others) practices may reflect different health values among rural residents compared with urban residents, or a greater focus on functionality rather than comfort or cosmetic quality8. Previous studies have shown higher belief in powerful others in lower socio-economic groups11,27, in older people27,29-30 and lower level of education12,31. Although these are characteristic of rural populations generally, the overall pattern ties in with those of the two other elements of the MDHLC, and is consistent with the notion of 'independence'. Further focused investigation of the influence of 'independence' on healthcare attitudes, beliefs and behaviours is needed. One issue, in particular, worthy of further investigation is the relationship between hope, beliefs and attitudes and subsequent health care behaviours: that is, are people with lower levels of hope less compliant in terms of treatment and follow up?
The developers of the MDHLC stated that there were no significant gender differences in interpretation of the questions or scales32. Thus our finding of higher belief in chance amongst men might reflect the fact that women generally take better care of themselves (internal) and also are more willing and accustomed to seeking professional care (powerful others) than men. As women were under-represented in this study, the issue will be explored intensively in the large study.
There were several potential limitations to this study. The participation rate reflected the inability of the first author to approach all patients personally to invite them to participate. This highlights the importance of having someone whose principal task is to recruit and assist patients in a survey of this type. The sample was not representative in terms of diagnosis (eg breast was particularly under-represented) and therefore probably not representative of the demographic mix of patients referred to The Townsville Cancer Centre, particularly in terms of gender. The small numbers of patients involved with the study limited statistical analysis, although Monto-Carlo estimations of p-values were used to increase confidence in the results. The classification between rural and urban patients was not always clear cut; however, there was a reasonable relationship between patients' perception and RRMA classification, as discussed above. The study was carried out in a large urban centre but not a capital city. It may be that including patients from larger urban areas would produce more significant differences between rural and urban participants.
Conclusions
Little work has previously examined the influence of rural background on health attitudes. This study demonstrated and discussed some interesting patterns worthy of further investigation and raised some additional areas for consideration. The findings support the veracity of the planned larger scale project to investigate health attitudes and beliefs in north Queensland oncology patients.
This study demonstrated the utility of the selected scales in terms of completion rate and consistency with other studies in terms of results. Additionally, the demographic questions demonstrated reasonable consistency. With respect to the identification with location questions, there is a need to include an open 'explanatory' question. The participation rate demonstrated the need for a consistent patient information and recruitment process, with dedicated recruitment staff.
Certain patterns, particularly with respect to rurality and treatment intent, were evident in the results that suggest further investigation is warranted and thus support the need for a large scale study. Larger numbers within each rurality/intent category should determine the veracity of the patterns described here. A number of potential research questions have arisen from this study which will be explored in the large scale study.
References
1. Australian Institute of Health and Welfare. Rural, regional and remote health: a study on mortality (summary of findings). AIHW cat. No. PHE 49 (Rural Health Series no. 3). Canberra: AIHW, 2003.
2. Cooney M, Baade P. Urban-rural differences in prostate cancer mortality, radical prostatectomy and prostate antigen testing in Australia. Medical Journal of Australia 2005; 182: 112-115.
3. Liff J, Chow W, Greenberg R. Rural-urban differences in stage at diagnosis. Cancer 1991; 67: 1454-1459.
4. Jong K, Smith D, Yu X, O'Connell L, Goldstein D, Armstrong B. Remoteness of residence and survival from cancer in New South Wales. Medical Journal of Australia 2004; 180: 618-622.
5. Hill D, White V, Giles G, Collins J, Kitchen P. Changes in the Investigation and Management of Primary Operable Breast Cancer in Victoria. Medical Journal of Australia 1994; 161: 110-112.
6. Bain N, Campbell N. Treating patients with colorectal cancer in rural and urban areas: a qualitative study of the patients' perspective. Family Practice 2000; 17: 475-479.
7. Bain N, Campbell N, Ritchie L, Cassidy J. Striking the right balance in colorectal cancer care - a qualitative study of rural and urban patients. Family Practice 2002; 19: 369-374.
8. Elliott-Schmidt R, Strong J .The concept of well being in a rural setting: understanding health and illness. Australian Journal of Rural Health 1997; 5: 59-63.
9. Burman M, Weinert C. Rural dwellers' cancer fears and perceptions of cancer treatment. Public Health Nursing 1997; 14: 272-279.
10. Kellehear A. Country health: another side of the rural crisis. Regional Journal of Social Issues 1988; 22: 1-8.
11. Marks G, Richardson JL, Graham J, Levine A. Role of health locus of control beliefs and expectations of treatment efficacy in adjustment to cancer. Journal Personal Social Psychology 1986; 51: 443-450.
12. Sugarek N, Deyo R, Holmes B. Locus of control and beliefs about cancer in a multi-ethnic clinic population. Oncology Nursing Forum 1998; 15: 481-486.
13. Nowotny M. Assessment of hope in patients: development of an instrument. Oncology Nursing Forum 1989; 16: 57-61.
14. Herth K .The relationship between level of hope and level of coping response and other variables in patients with cancer. Oncology Nursing Forum 1989; 16: 67-72.
15. Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ et al. The European Organisation for Research and Treatment of Cancer QLQ-C30: A quality of life instrument for use in international clinical trials in oncology. Journal of the National Cancer Institute 1993; 85: 365-376.
16. Groenvold M, Klee M, Sprangers M, Aaronson N. Validation of the QLQ-C30 Quality of Life Questionnaire through combined qualitative and quantitative assessment of patient-observer agreement. Journal of Clinical Epidemiology 1997; 50: 441-450.
17. Broadbent WE, Gehlbach SH, de Gruy FV, Kaplan BH. The Duke-UNC Functional Social Support Questionnaire. Measurement of social support in family medicine patients. Medical Care 1988; 26: 709-723.
18. McDowell I, Newell C, Measuring Health, Second edn. Oxford: Oxford University Press, 1996; 140-142.
19. Wallston K, Wallston B, DeVellis R. Development of the Multidimensional Health Locus of Control Scales. Health Education Monographs 1978; 6(2): 160-171.
20. Veitch PC. An Investigation of the influence of distance on rural and remote area residents' health care behaviour. PhD Thesis, The University of Queensland, Brisbane, Australia; 1995.
21. Herth K. Abbreviated instrument to measure hope; development and psychometric evaluation. Journal of Advanced Nursing 1992; 17: 1251-1259.
22. Felder B. Hope and coping in patients with cancer diagnoses. Cancer Nursing 2004; 27: 320-324.
23. Australian Government. Rural, Remote and Metropolitan Areas Classification, 1991 Census Edition. Canberra, Australian Government Publishing Service.
24. Rustoen T, Wiklund I. Hope in newly diagnosed patients with cancer. Cancer Nursing 2000; 23: 214-219.
25. Ballard A, Green T, Mccaa A, Logson M. A comparison of the level of hope in patients with newly diagnosed and recurrent cancer. Oncology Nursing Forum 1997; 24: 899-904.
26. Herth K. Fostering hope in terminally-ill people. Journal of Advanced Nursing 1990; 15: 1250-1259.
27. Paxton S, Sculthorpe A. Weight and health locus of control beliefs in an australian community sample. Psychology and Health 1999; 14: 417-431.
28. Nagy VT, Wolfe G. Chronic illness and health locus of control beliefs. Journal of Social Psychology 1983; 1(1): 58-65.
29. Buckelew S, Shotty M, Hewett J, Landon T, Morrow K, Frank R. Health locus of control, gender differences and adjustment to persistent pain. Pain 1990; 42: 287-294.
30. Fleming M, Barry K. Health locus of control in a primary care sample of alcoholics and non-alcoholics. Behavioural Medicine 1991; Spring: 25-30.
31. Galanos A, Strauss R, Pieper C. Sociodemographic correlates of health beliefs among black and white community dwelling elderly individuals. International Journal of Aging and Human Development 1994; 38: 339-350.
32. Wallston K, Wallston B. Health Locus of Control scales, research with the locus of control construct (Vol 1). New York: Academic Press, 1981; 189-243.
Abstract
Introduction: Rural patients have poor outcomes in cancer management. Previous studies have shown different health beliefs and values in rural populations with high levels of stoicism and fatalism, leading to later presentation and diagnosis, with subsequent poorer prognosis and survival. This study explores the relationship between urban or rural background and health attitudes of newly diagnosed oncology patients, attending one oncology unit in north Queensland, during a 5 month period. This study is a forerunner to a planned larger project to explore the research question: do oncology patients from rural backgrounds differ in their health attitudes and hope levels compared with those from urban backgrounds? The aim of this study was to determine the utility of the selected validated instruments, newly diagnosed patients' willingness and ability to complete the composite survey instrument, and to identify likely issues for inclusion and/or greater focus in the larger project. As pilot studies are also used to develop or refine research questions and hypotheses, this article also considers some research questions for the planned large scale study.
Methods: Self-administered questionnaire survey of 47 patients newly referred to the Medical Oncology Department in The Townsville Cancer Centre. Scales used were: the EORTC QLQ-C30 to assess symptom burden and quality of life; the Duke UNS Functional Social Support Questionnaire to assess social support; the Herth Hope Index to assess hope; and the Multi-Dimensional Health Locus of Control to assess health beliefs. Data were collated and transformed according to the various scales' scoring manuals. Rurality was ascertained using the RRMA classification and patient self-assessment. Uni-variate analyses were conducted as small numbers precluded multi-variate analysis. Non-parametric Mann-Whitney U and Kruskal-Wallis tests were used where data were skewed, or categorical. Monte-Carlo estimations of p-values were generated.
Results: In all, 28 of 47 patients classified as rural, 27 were suitable for curative treatment, and 31 were male. Median age was 56 years. Some respondents (17%) identified as 'rural', although they had an urban residence, and vice versa. Health attitude scores were not affected by global health scores or by intent of treatment (palliative/curative). Males scored significantly higher for belief in chance. Rural patients scored significantly higher for internal belief and belief in chance. No statistically significant differences were evident between rural/urban patients by gender, nor social support scores. Hope levels were generally high with no significant difference between urban and rural patients, regardless of treatment intent.
Discussion: The study does reveal differences in health attitudes between urban and rural populations; however, there are several confounding factors which may contribute to this, especially gender. In this study women were under-represented. People with fatalistic beliefs (high belief in chance) tend to have poor initiative in health matters which may cause delay in seeking treatment, or poor compliance with treatment. Analysis is limited by small numbers of patients. This study is a pilot to a larger project to investigate health attitudes and decisions by oncology patients in northern Queensland. The questionnaire was well received by patients, but the need for a dedicated recruitment person was evident. There is a need to determine how patients identify in terms of rurality over and above their actual place of residence.
Keywords: cancer, health attitudes, hope.
You might also be interested in:
2016 - Food-related practices and beliefs of rural US elementary and middle school teachers
2008 - Measuring primary care: the standard primary care year
